Astronaut Don Pettit posted this image of a Jupiter-like water globe he created on the International Space Station. In microgravity, surface tension reigns as the water’s supreme force, pulling the mixture of water and food coloring into a perfect sphere. It will be interesting to see a video version of this experiment, so that we can tell what tools Pettit used to swirl the droplet into the eddies we see. Is the full droplet rotating (as a planet would), or are we just seeing the remains of a wire passed through the drop? We’ll have to stay tuned to Pettit’s experiments to find out. (Image credit: NASA/D. Pettit; via space.com; submitted by J. Shoer)
Search results for: “water droplet”

Where to Follow FYFD Online
Hi, folks! As the social media landscape fractured, I’ve been dragging my feet about making some needed changes. But no longer. As of November 2024, I am no longer updating FYFD’s X/Twitter account. Here are the places you can currently follow FYFD online:
- Bluesky
- Mastodon/Fediverse: @[email protected]
- Tumblr
- YouTube
Most of those services get autoposts rather than regular check-ups, so I rarely see messages on Instagram/Tumblr/YouTube. Fediverse replies autopost as comments to the blog, so I do see those, and I will probably hang around on Bluesky some, but email is your best bet these days if you want me to see your message.
And, if you just want FYFD in your inbox every other week, you can subscribe to the newsletter!
(Image credit: P. Czerwinski)

Non-Newtonian Raindrops
Fluids like air and water are called Newtonian because their viscosity does not vary with the force that’s applied to them. But many common fluids — almost everything in your fridge or bathroom drawer, for example — are non-Newtonian, meaning that their viscosity changes depending on how they’re deformed.
Non-Newtonian droplets can behave very differently than Newtonian ones, as this video demonstrates. Here, their fluid of choice is water with varying amounts of silica particles added. Depending on how many silica particles are in the water, the behavior of an impacting drop varies from liquid-like to completely solid and everything in between. Why such a great variation? It all has to do with how quickly the droplet tries to deform and whether the particles within it can move in that amount of time. Whenever they can’t, they jam together and behave like a solid. (Image, video, and research credit: S. Arora and M. Driscoll)

Marangoni Blossoms
When surface tension varies along an interface, fluids move from regions of low surface tension to higher surface tension, a behavior known as the Marangoni effect. Here, a drop of (dyed) water is placed on glycerol. The two fluids are miscible, but water has much a lower viscosity and density yet a higher surface tension. The drop’s interface quickly becomes unstable; viscous fingers form along the edge as the less viscous water pushes into the more viscous glycerol. Eventually, the surface-tension-driven Marangoni flow breaks those fingers off into lip-like daughter drops. The researchers also show how the interplay between viscosity and surface tension affects the size of fingers that form by varying the water/glycerol concentration. (Image and video credit: A. Hooshanginejad et al.)

When Fires Make Rain
The intense heat from wildfires fuels updrafts, lifting smoke and vapor into the atmosphere. As the plume rises, water vapor cools and condenses around particles (including ash particles) to form cloud droplets. Eventually, that creates the billowing clouds we see atop the smoke. These pyrocumulus clouds, like this one over California’s Line fire in early September 2024, can develop further into full thunderstorms, known in this case as pyrocumulonimbus. The storm from this cloud included rain, strong winds, lightning, and hail. Unfortunately, storms like these can generate thousands of lightning strikes, feeding into the wildfire rather than countering it. (Image credit: L. Dauphin; via NASA Earth Observatory)

Billowing Ouzo
Pour the Greek liquor ouzo into water, and your glass will billow with a milky, white cloud, formed from tiny oil droplets. The drink’s unusual dynamics come from the interactions of three ingredients: water, oil, and ethanol. Ethanol is able to dissolve in both water and oil, but water and oil themselves do not mix.
In this video, researchers explore the turbulent effects of pouring ouzo into water. In particular, pouring from the top creates a fountain-like effect, due to a tug-of-war between the ouzo’s momentum and its buoyancy. Momentum wants the ouzo to push down into the water, and buoyancy tries to lift it back up. For an extra neat effect, they also show what happens when the ouzo is confined to a 2D plane and what happens when momentum and buoyancy act together instead of oppositely. (Image and video credit: Y. Lee et al.)

Shaped Splashes
When a raindrop hits a leaf, it spreads out into a rimmed sheet that breaks up into droplets. These tiny drops can carry dust, spores, and even pathogens as they fly off. But many leaves aren’t smooth-edged; instead they have serrations or teeth. How does that affect a splash? That’s the question at the heart of today’s study.

A water drop hits a star-shaped pillar and breaks up. To simplify from a leaf’s shape, the team studied water dropping onto star-shaped pillars. As seen above and below, the pillar’s edge shaped the splash sheet, with the sheet extending further in the edge’s troughs. This asymmetry extends into the rim also, concentrating the liquid — and the subsequent spray of droplets — along lines that extend from the edge’s troughs and peaks.

A viscous water-glycerol drop hits a star-shaped pillar, spreads, and breaks into droplets. The team found that, in addition to sending drops along a preferred direction, the shaped edge made the droplets larger and faster than a smooth edge did. (Image and research credit: T. Bauer and T. Gilet)

“Starlit”
In “Starlit,” filmmaker Roman de Giuli leverages paint, ink, water, and oil to create astronomical views. Colorful droplets spin past like neon exoplanets. Shards of glitter form comets. Satellite droplets become moons about their larger sibling. (Video and image credit: R. de Giuli)

Making Reconfigurable Liquid Circuits
Microfluidic circuits are key to “labs on a chip” used in medical diagnostics, inkjet printing, and basic research. Typically, channels in these circuits are printed or etched onto solid surfaces, making it difficult to reconfigure them. A group in China developed an alternative design, inspired by reconfigurable toys like Lego blocks. Their set-up, shown above, uses a pillared surface immersed in oil. To create the channels, they pipette water — one droplet at a time — into the space between pillars. The combination of oil and pillars traps the drop. With multiple drops linked together, they get channels, like the ones above that mix two fluids. When the time comes to reconfigure the channels, they just pipette the water out and cut the channel with a sheet of coated paper. (Image and research credit: Y. Zeng et al.; via Physics Today)

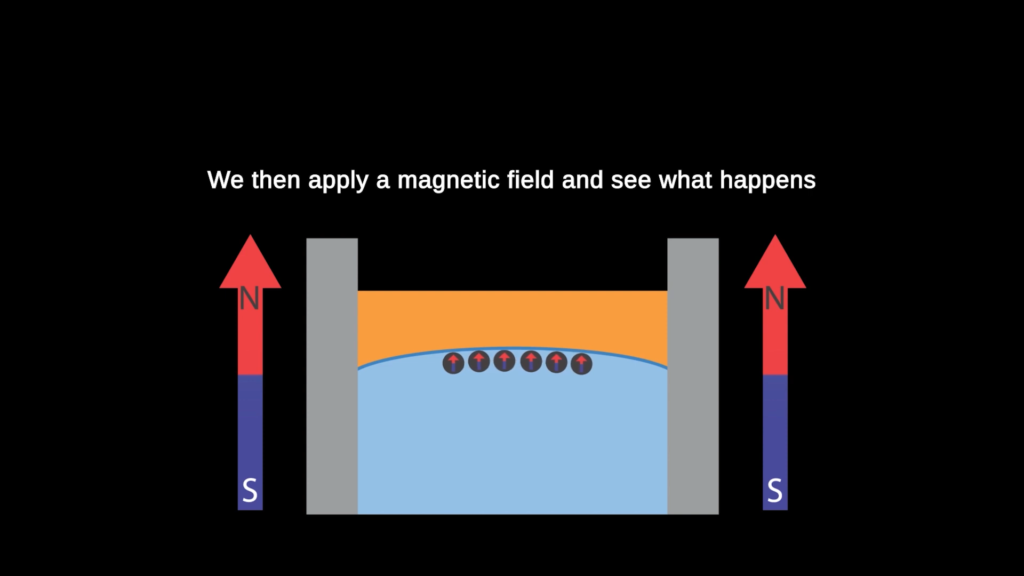
Making Magnetic Crystals From Ferrofluids
Ferrofluids are a great platform for exploring liquids and magnetism. Here, researchers trap ferrofluid droplets along an oil-water meniscus and then apply a magnetic field that makes the drops repel one another. The results are crystalline patterns formed from magnetic droplets. For a given patch of drops, increasing the magnetic field’s strength pushes drops further apart. But changing the drops’ size and levels of self-attraction also shifts the patterns. Check out the video to see the crystals in action. (Video and image credit: H. Khattak et al.)