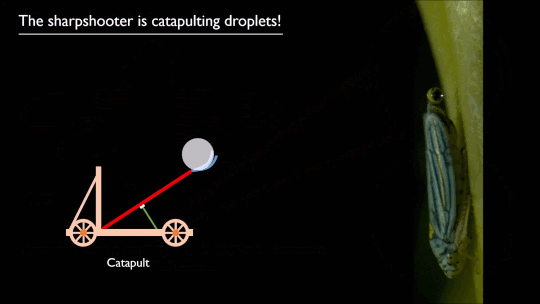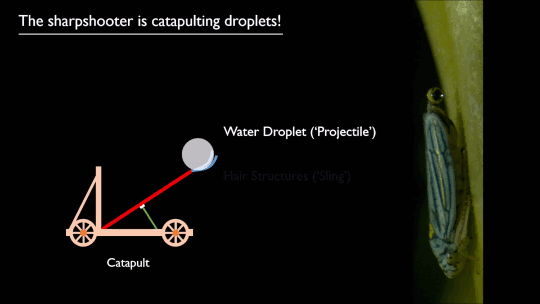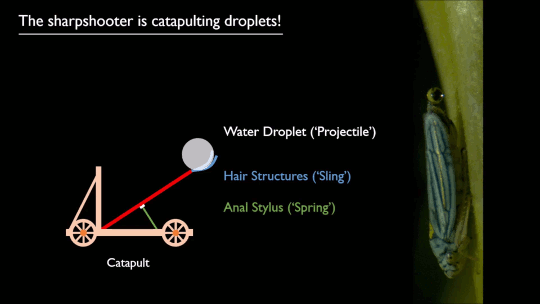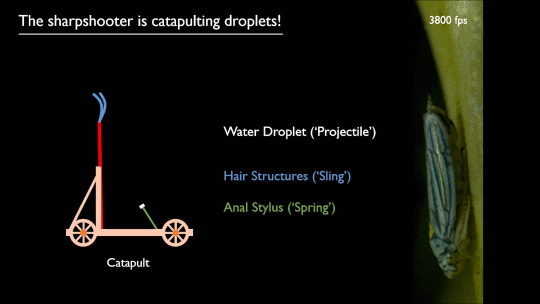
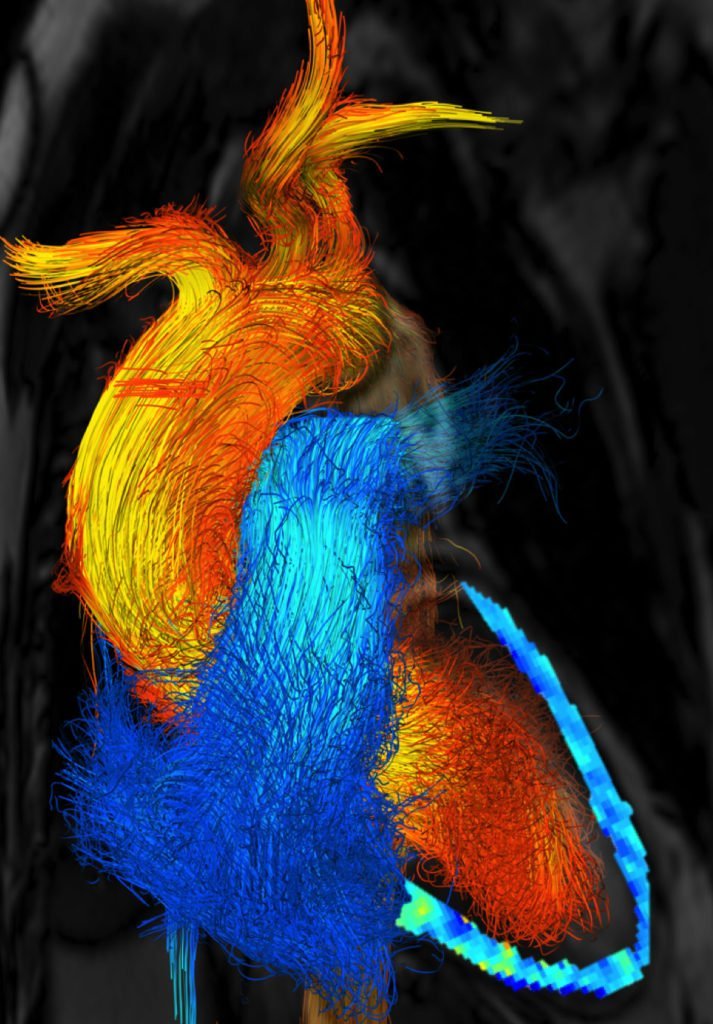
As pilots can tell you, flying near the ground (or an open expanse of water) gives one an aerodynamic boost. Essentially, the surface acts like a mirror, reflecting and dissipating the wingtip vortices that create downwash. That reduces the power necessary to fly, as long as you’re flying within about a wingspan of the surface.
Theoretically, flapping fliers like bats and birds should also benefit from this ground effect, but measurements have been hard to come by. A new study using bats trained to fly in a wind tunnel provides some of the first detailed measurements of ground effect for flapping animals. The researchers found a 29% reduction in the power necessary for flight when in ground effect compared to being out of it! That’s twice the savings predicted by modeling, meaning we still have a ways to go to accurately capture the physics of flapping flight under these circumstances.
Such a substantial savings also strengthens arguments for flight developing from the ground up. Using ground effect, surface-dwelling animals could have evolved flight gradually, taking advantage of the energy savings offered by sticking close to the surface. (Image and research credit: L. Johansson et al.; submitted by Marc A.)