This simulation shows 2D Rayleigh-Benard convection in which a fluid of uniform initial temperature is heated from below and cooled from above. This is roughly analogous to the situation of placing a pot of water on a hot stovetop. (In the case of the water on the stove, the upper boundary is the water-air interface, while, in the simulation, the upper boundary is modeled as a no-slip (i.e. solid) interface.) The simulation shows contours of temperature (black = cool, white = hot). In general, the hot fluid rises and the cold fluid sinks due to differences in density, but, as the simulation shows, the actual mixing that occurs is far more complex than that simple axiom indicates.
Search results for: “convection”

Solutal Convection
Solutal convection, rather than relying on temperature gradients, can occur due to gradients in concentration or in surface tension. While less spectacular than this previously posted video, this video contains a nice simplified explanation of the mechanism. And, as noted in the video, this is a demo you can do yourself at home.

DIY Solutal Convection
In this video, the HouseholdHacker heads to the kitchen and uses milk, food coloring, and dish soap to create some solutal convection much like this one with cream and liqueur. The food coloring serves as a tracer for the fluid motion; it’s really the interaction of the milk and the dish soap that drives the motion. The dish soap lowers the surface tension of the milk, causing motion via the Marangoni effect. That motion redistributes some of the dish soap as well, causing further motion in the form of a surface tension instability. As with the cream and liqueur experiment, the fat in the milk is important; you won’t see much (if any) effect with fat free milk because its surface tension properties aren’t as dissimilar to dish soap. (via misterhonk.de)

Marangoni Convection in Space
In this Saturday Morning Science video, astronaut Don Pettit demonstrates Marangoni convection in microgravity using a water film with tracer particles, a soldering iron, and a flashlight. This same effect occurs on earth but is masked behind the much stronger effect of buoyant convection.

Thermal Convection
This video turbulent convection in a vertical channel. Buoyancy and the density variations caused by small differences in temperature are what drive the behavior.

Convection in Cream and Liqueur
We are used to associating convection with differences in temperature, but what’s actually necessary for a Rayleigh-Taylor-type instability is a density variation (and a gravitational field). The solutal convection seen above when mixing liqueur with cream is caused by the interaction of density and surface tension. When the alcohol of the liqueur mixes with the cream, it forms a less dense alcohol-cream that tries to rise to the surface. The alcohol also breaks the surface tension of the cream, causing it to contract and open cells where the alcohol surfaces. As the alcohol evaporates, the alcohol-cream mixture gets denser and sinks back down where it can pick up more alcohol and start the process again. (via jshoer and io9)

Inside Cepheid Variable Stars
Cepheid variable stars pulsate in brightness over regular periods. That’s one reason astronomers use them as a standard candle to judge distances–even for stars well outside our galaxy. In this image, researchers display a simulation of convection inside a Cepheid eight times more massive than our sun. The colors represent vorticity, with zero vorticity in white.(Image credit: M. Stuck and J. Pratt)

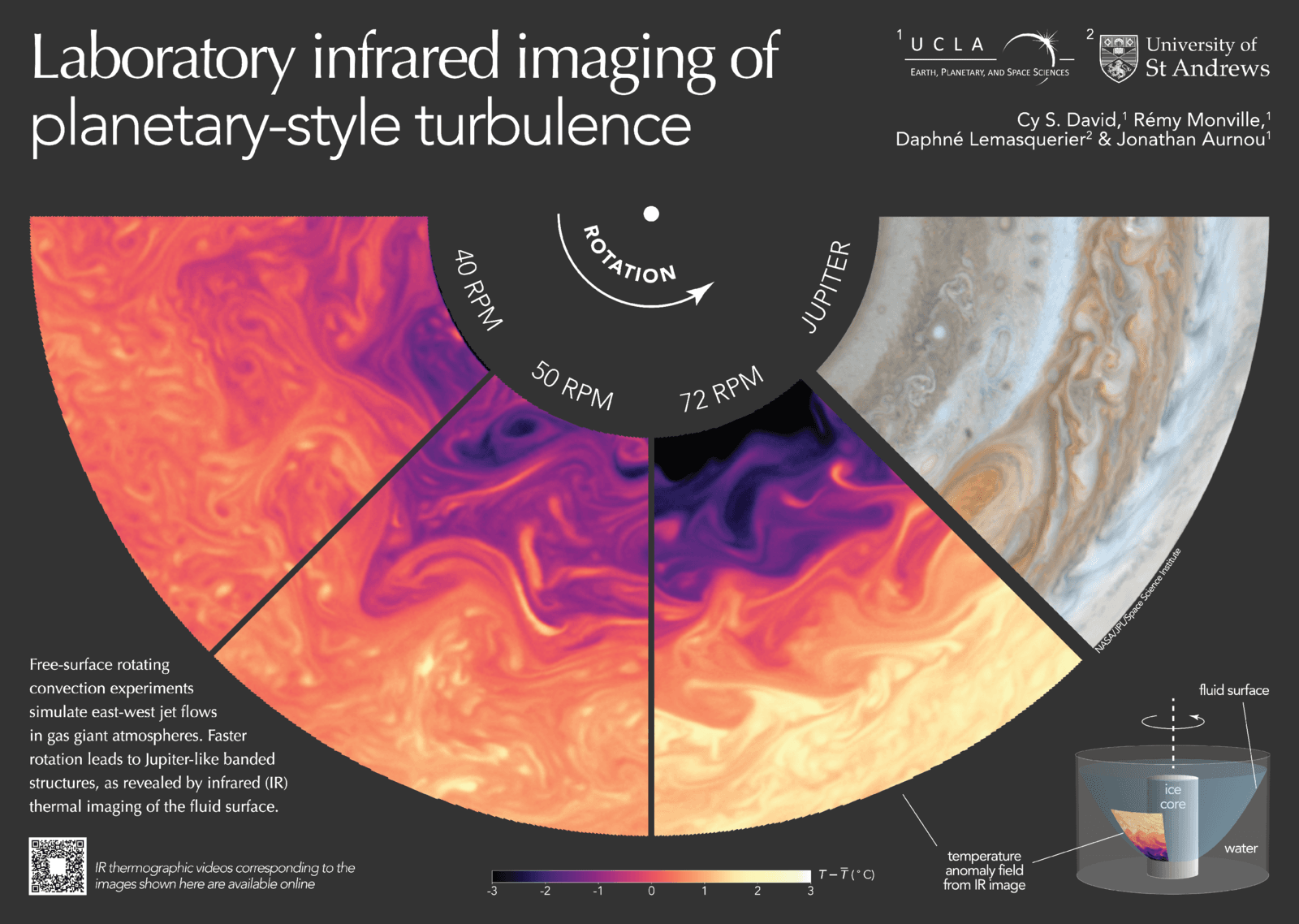
Jupiter in a Lab
The vivid bands of a gas giant like Jupiter come from the planet’s combination of rotation and convection. It’s possible to create the same effect in a lab by rapidly spinning a tank of water around a central ice core. That’s the physical set-up behind this research poster–note the illustration in the lower right corner. The central snapshots show how temperature gradients on the water surface change the faster the tank rotates. At higher rotational speeds, the parabolic water surface gets ever steeper and Jupiter-like temperature bands form. (Image credit: C. David et al.)
The Balvenie
Photographer Ernie Button explores the stains left behind when various liquors evaporate. This one comes from a single malt scotch whisky by The Balvenie. The stain itself is made up of particles left behind when the alcohol and water in the whisky evaporate. The pattern itself depends on a careful interplay between surface tension, evaporation, pinning forces, and internal convection as the whisky puddle dries out. (Image credit: E. Button/CUPOTY; via Colossal)

“Vorticity 6”
It’s time for another storm-chasing timelapse from photographer Mike Olbinski! “Vorticity 6” focuses on supercell thunderstorms and their tornadoes. There’s billowing turbulent convection, undulating asperitas, bulging mammatus, microbursts, and more. There’s nothing like timelapse to highlight the growth, rotation, and shear involved in these storms. (Video and image credit: M. Olbinski)





