For decades, biologists have focused on genetics as the key determiner for biological processes, but genetic signals alone do not explain every process. Instead, researchers are beginning to see an interplay between genetics and mechanics as key to what goes on in living bodies.
For example, scientists have long tried to unravel how an undifferentiated blob of cells develops a clear head-to-tail axis that then defines the growing organism. Researchers have found that, rather than being guided purely by genetic signals, this stage relies on mechanical forces–specifically, the Marangoni effect.
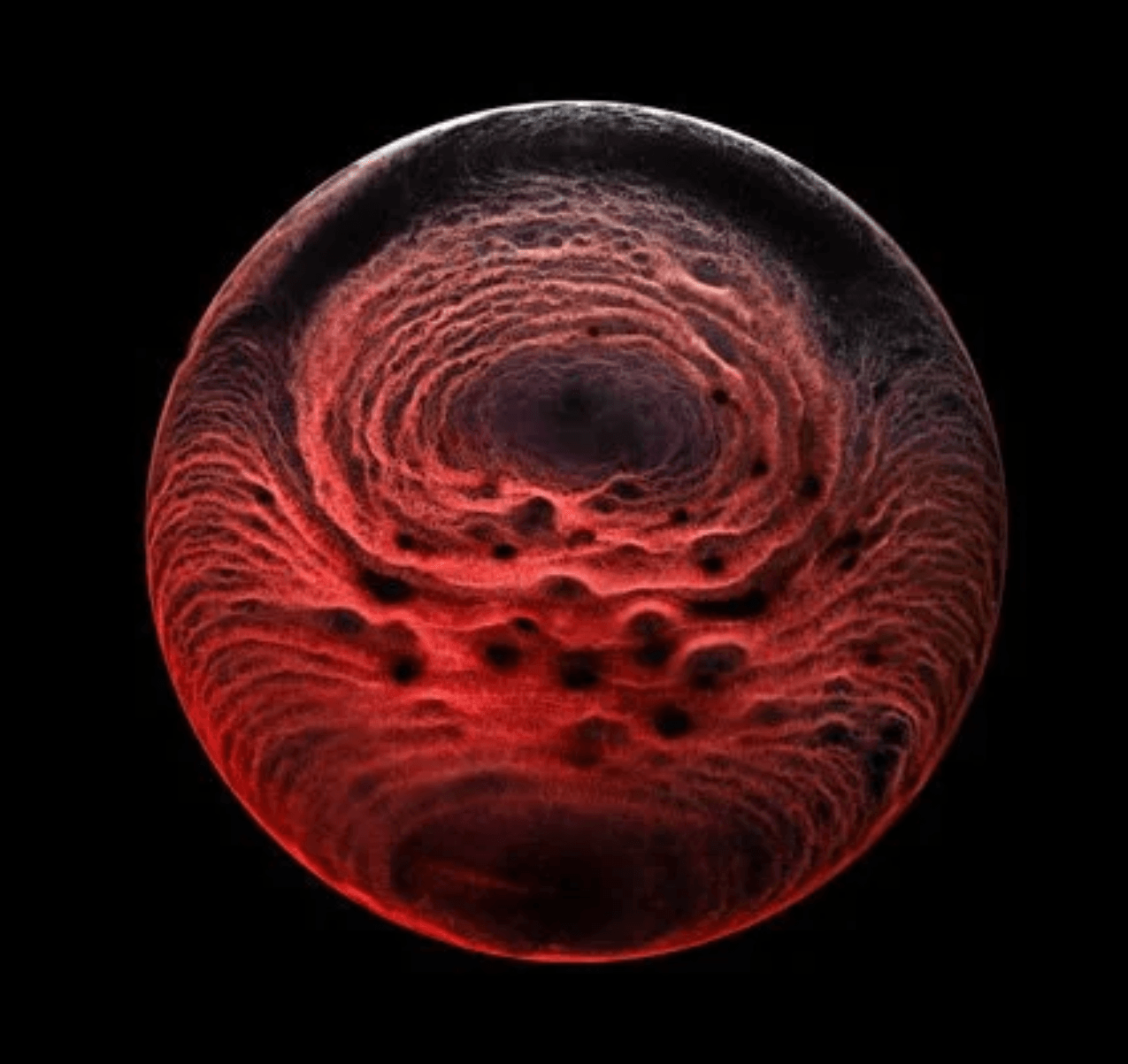
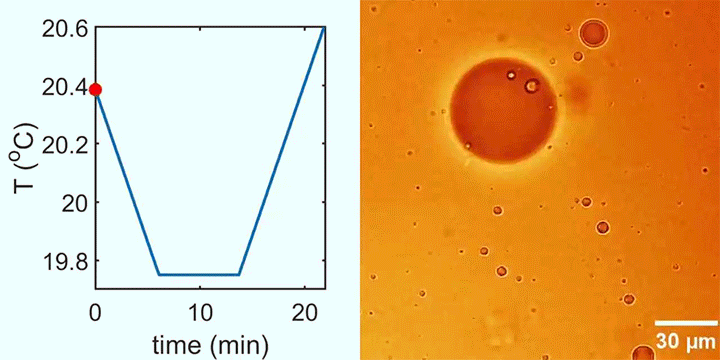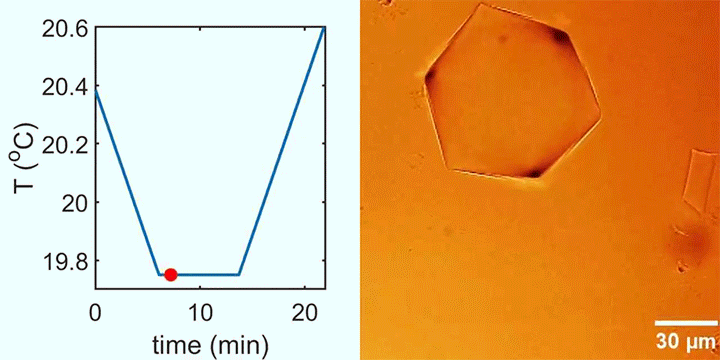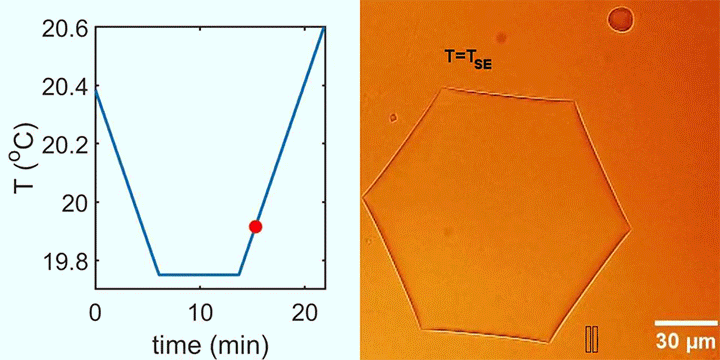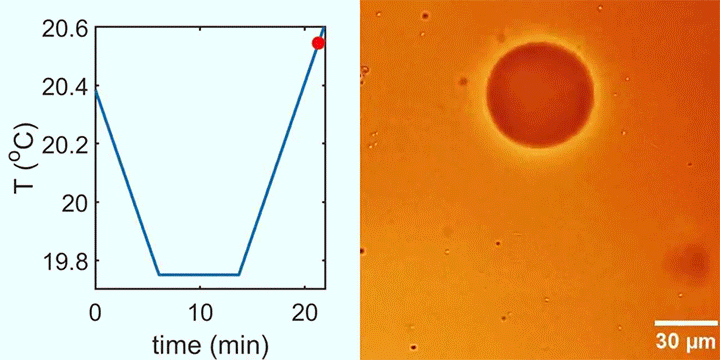
The image above shows a mouse gastruloid, a bundle of stem cells that mimic embryo growth. As they develop, cells flow up the sides of the gastruloid, with a returning downward flow down the center. This is the same flow that happens in a droplet with higher surface tension in one region; the Marangoni effect pulls fluid from the lower surface tension region to the higher one, with a returning flow that completes the recirculation circuit.
The same thing, it turns out, happens in the gastruloid. Genes in the cells trigger a higher concentration of proteins in one region of the bundle, creating a lower surface tension that causes tissue to flow away, helping define the head-to-tail axis. (Image credit: S. Tlili/CNRS; research credit: S. Gsell et al.; via Wired)














![Black and white image of a film pulled outward and breaking into droplets. Text reads, "The [0.05%] surfactant renders the ejected droplets prone to 'popping'." Black and white image of a film pulled outward and breaking into droplets. Text reads, "The [0.05%] surfactant renders the ejected droplets prone to 'popping'."](https://fyfluiddynamics.com/wp-content/uploads/surfburst2-1024x576.png)
![Black and white image of a film pulled outward and spreading in unevenly. Text reads, "When surfactant concentration is further increased [to 1%], drop spreading resumes." Black and white image of a film pulled outward and spreading in unevenly. Text reads, "When surfactant concentration is further increased [to 1%], drop spreading resumes."](https://fyfluiddynamics.com/wp-content/uploads/surfburst3-1024x576.png)