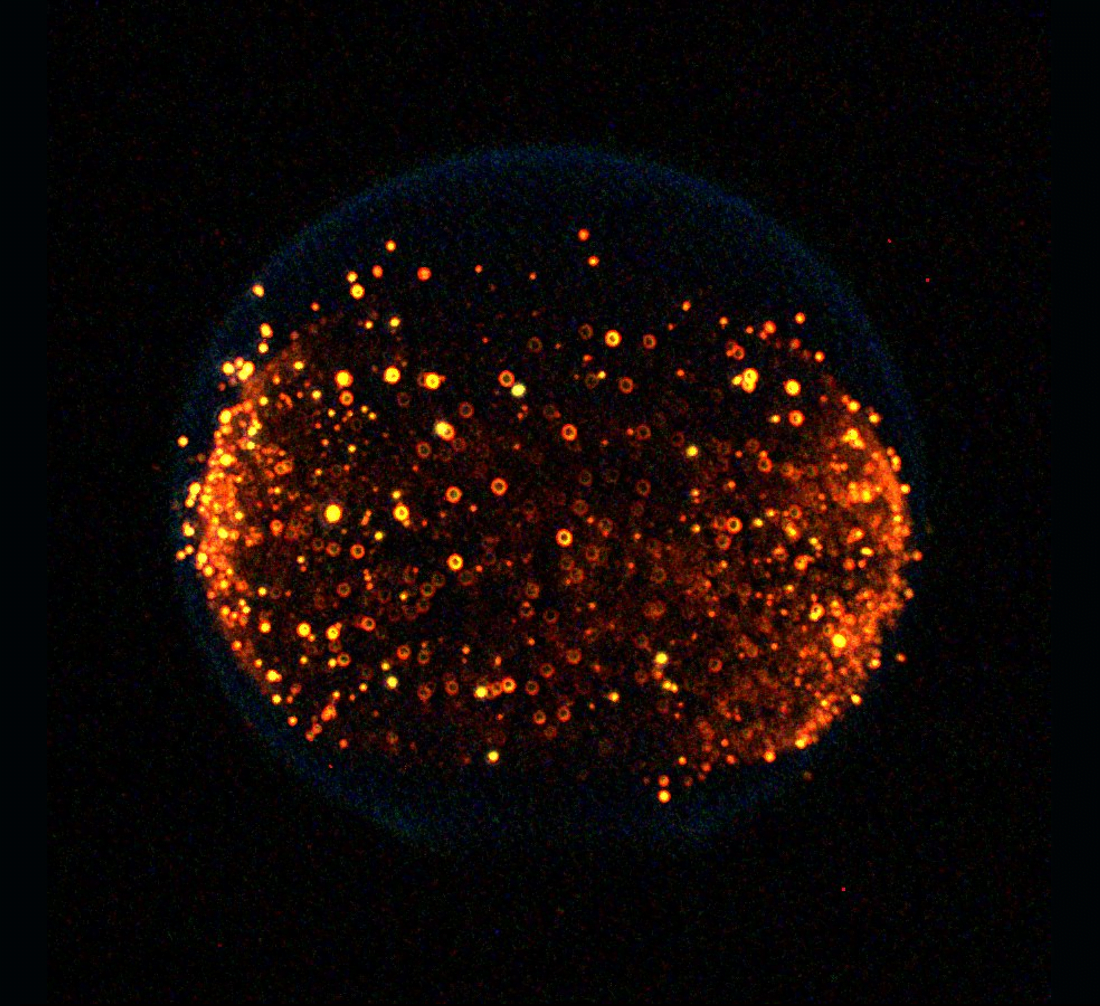
In space, flames behave quite differently than we’re used to on Earth. Without gravity, flames are spherical; there are no hot gases rising to create a teardrop-shaped, flickering flame. In many ways, removing gravity makes combustion simpler to study and allows scientists to focus on fundamental behaviors. It’s no surprise, then, that combustion experiments are a long-standing feature on the International Space Station.
In the photo above, we see a flame in microgravity studded with bright yellow spots of soot. Soot is a by-product of incomplete combustion; it’s essentially unburned leftovers from the chemical reaction between fuel and oxygen. In this experiment, researchers were studying how much soot is produced under different burning conditions, work that will help design flames that burn more cleanly in the future. (Image and video credit: NASA; submitted by @LordDewi)