For a little Friday fun, consider this microgravity experiment conducted aboard the infamous Vomit Comet: what happens when you combine Diet Coke and Mentos in microgravity? For the record, I think this is the answer. (Via jshoer, who will be flying on the Vomit Comet in 2 weeks – but sadly not with a fluids experiment)
Search results for: “microgravity”
Microgravity Boiling
Boiling a liquid is a common enough phenomenon that we are all familiar with it. But, as with many aspects of fluid mechanics, removing gravity drastically changes the situation.

Combustion in Microgravity
‘Hot air rises.’ It’s common knowledge. But we usually forget that this is only true thanks to Earth’s gravity. On Earth, a candle flame’s distinctive pointed shape is due to hot air rising. Without gravity, there is no buoyant convection; hot air has no reason to rise (and no definition of what up is either!). This makes flames in microgravity spherical, as in the video above from a drop tower on earth. See also: astronaut explains fire in microgravity.

Water Balloons in Microgravity
Sometimes you need microgravity in order to observe the neat effects of surface tension on a fluid. Also, I hear it’s a good excuse for popping water balloons on the Vomit Comet. #

A Mini Jupiter
Astronaut Don Pettit posted this image of a Jupiter-like water globe he created on the International Space Station. In microgravity, surface tension reigns as the water’s supreme force, pulling the mixture of water and food coloring into a perfect sphere. It will be interesting to see a video version of this experiment, so that we can tell what tools Pettit used to swirl the droplet into the eddies we see. Is the full droplet rotating (as a planet would), or are we just seeing the remains of a wire passed through the drop? We’ll have to stay tuned to Pettit’s experiments to find out. (Image credit: NASA/D. Pettit; via space.com; submitted by J. Shoer)

Ice Without Gravity
Astronaut Don Pettit is back in space, and that means lots of awesome microgravity experiments. Here, he grew thin wafers of ice in microgravity in a -95 degree Celsius freezer. Then he took the ice wafers and photographed them between crossed polarizers, creating this colorful image. The colors highlight different crystal orientations within the ice and give us a hint about how the freezing front formed and expanded. I can’t wait to see more examples! (Image credit: D. Pettit/NASA; via Ars Technica; submitted by J. Shoer)

Farewell, Saffire!
After eight years and six flight tests, NASA said a fiery farewell to the Spacecraft Fire Safety Experiment, or Saffire, mission. Each Saffire test took place on an uncrewed Cygnus supply vehicle after undocking from the space station. Cygnus craft burn up during atmospheric re-entry, so using them as a platform guaranteed safety for the station’s crew.
A Plexiglass sample burns as part of Saffire-V’s experiments. In this experiment, researchers found that flames grew and spread faster on thin ribs of Plexiglass (left) than on thicker samples (right). Saffire itself used a small wind tunnel to push air past its burning materials. The tests included materials like plexiglass, cotton, Nomex, and other fabrics that might be found on a spacecraft or its occupants. The goal, of course, is to understand how fires grow and spread in a spacecraft in order to protect the crew. To that end, Saffire experiments recorded not only what went on inside their test unit, but also what the conditions were in the spacecraft as Saffire burned. (Image and video credit: NASA; via Gizmodo and NASA Glenn)

Droplet Medusa
Vibration is one method for breaking a drop into smaller droplets, a process known as atomization. Here, researchers simulate this break-up process for a drop in microgravity. Waves crisscrossing the surface create localized craters and jets, making the drop resemble the Greek mythological figure of Medusa. With enough vibrational amplitude, the jets stretch to point of breaking, releasing daughter droplets. (Image and research credit: D. Panda et al.)

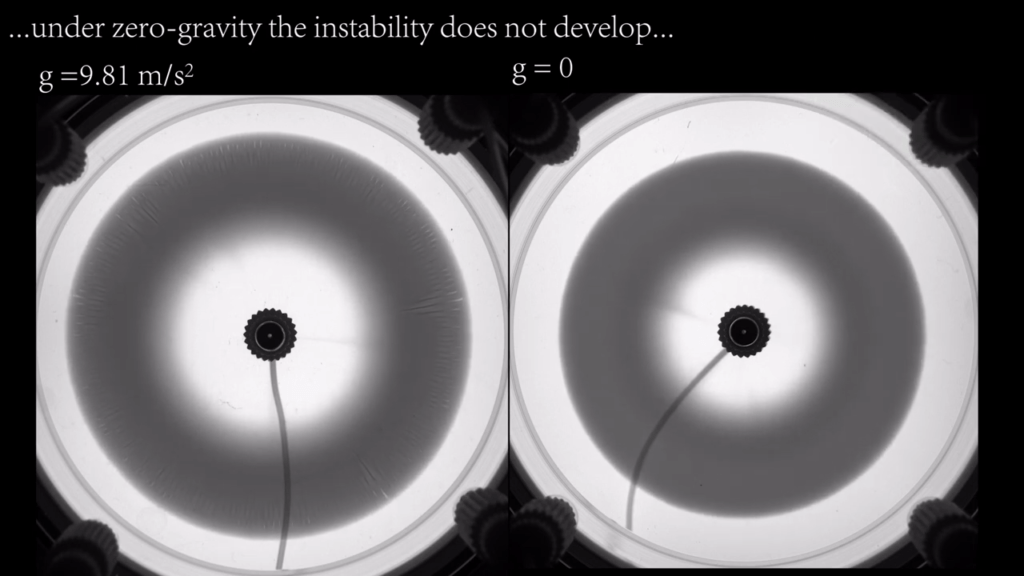
Chemical Flowers
These “flowers” blossom as two injected chemicals react in the narrow space between two transparent plates. The chemical reaction produces a darker ring that develops a streaky outer edge due to competition between convection and chemical diffusion.
To show how gravity affects the instability, the researchers repeated the experiment on a parabolic flight. In microgravity conditions, no instability formed. That’s exactly what we’d expect if convection (i.e. flow due to density differences) is a major cause. No gravity = no convection. In contrast, under hypergravity conditions, the instability was initially spotty before developing streaks. (Image and video credit: Y. Stergiou et al.)

Free Contact Lines
How a simple drop of water sits on a surface is a strangely complicated question. The answer depends on the droplet’s size, its chemistry, the roughness of the surface, and what kind of material it’s sitting on. Vetting the mathematical models that describe these behaviors is especially difficult since droplets often get stuck, or “pinned,” along their contact line where water, air, and surface meet.
To get around this issue, researchers sent their experiment to the International Space Station, asking astronauts to run the tests for them. Without gravity‘s influence squishing drops, the astronauts could use much larger droplets than they could on Earth. Larger drops are less likely to get pinned by a stray surface defect, so on the space station, astronauts could place droplets on a vibrating platform and observe their contact line freely moving as the drop changed shape. Under these conditions, the experiment tested many surfaces with different wetting characteristics, thereby gathering data to test models we cannot easily confirm on Earth. (Image and research credit: J. McCraney et al.; via APS Physics)