On Earth, it’s easy for the effects of surface tension and capillary action to get masked by gravity’s effects. This makes microgravity experiments, like those performed with drop towers or onboard the ISS, excellent proving grounds for exploring fluid dynamics unhindered by gravity. The video above looks at how colliding jets of liquid water behave in microgravity. At low flow rates, opposed jets form droplets that bounce off one another. Increasing the flow rate first causes the droplets to coalesce and then makes the jets themselves coalesce. Similar effects are seen in obliquely positioned jets. Perhaps the most interesting clip, though, is at the end. It shows two jets separated by a very small angle. Under Earth gravity, the jets bounce off one another before breaking up. (The jets are likely separated by a thin film of air that gets entrained along the water surface.) In microgravity, though, the jets display much greater waviness and break down much quicker. This seems to indicate a significant gravitational effect to the Plateau-Rayleigh instability that governs the jet’s breakup into droplets. (Video credit: F. Sunol and R. Gonzalez-Cinca)
Search results for: “microgravity”

Fire in Microgravity
In the movie “Gravity” Sandra Bullock’s character battles a fire aboard the International Space Station. Combustion is a huge concern in space habitats. Microgravity fires are challenging to detect and fight because they behave very differently in the absence of buoyancy. On Earth, buoyancy makes hot air rise from a flame while cooler air is pulled in near the base. This feeds fresh oxygen to the teardrop-shaped flame. In space, there is no buoyancy and flames are spherical. They also burn at lower temperatures and lower oxygen concentrations–so low, in fact, that the oxygen depletion necessary to extinguish a fire is lower than what humans require to survive.
No buoyancy makes it harder for fires to spread, but it also makes them harder to detect since smoke doesn’t rise toward a detector on the ceiling. Instead, fire detectors aboard the Space Station are housed in the ventilation system that moves air through the modules constantly. In the event of a fire, astronauts use a three-step fire suppression system. First, they shut off the ventilation system to delay the fire’s spread. Then they shut off power to the affected unit, and, finally, they use fire extinguishers on the flames. The Russian module is equipped with a foam extinguisher and the others use CO2 units. (Image credit: Warner Brothers)

Microgravity Water Balloons
When a water balloon pops in microgravity, waves propagate from the initial point of contact and the final point of contact (where the balloon skin peels away). As these waves come inward toward one another, the water is compressed from its original potato-like shape into a pancake-like one. In most cases, surface tension will provide a damping force on this oscillatory motion, eventually making the water into a sphere. On Earth, in contrast, a water balloon seems to hold its shape after popping. This is because the effect of gravity on the water is much larger than the effect of the propagating waves. This is one reason that it is useful to have a laboratory in space! Without a microgravity environment, it is much harder to study and observe secondary and tertiary-order forces on a physical event. (Video credit: Don Pettit, Science Off The Sphere)

Microgravity Cornstarch
We’ve seen the effects of vibration on shear-thickening non-Newtonian fluids here on Earth before in the form of “oobleck fingers” and “cornstarch monsters”, but, to my knowledge, this is the first such video looking at the behavior in space. The vibrations of the speaker cause shear forces on the cornstarch mixture, which causes the viscosity of the fluid to increase. This is what makes it react like a solid to sudden impacts while still flowing like a liquid when left unperturbed. In microgravity there is one less force working against the rise of the cornstarch fingers, so the formations we see in this video are subtly different from those on Earth.

Microgravity Combustion
This collage of three combustion images reveals the beautiful symmetry of flames in microgravity. In the absence of gravity, flames are spherical, and, in the confines of a spacecraft, any combustion is extremely dangerous. Thus, most microgravity combustion experiments occur in drop towers. From NASA:
Each image is of flame spread over cellulose paper in a spacecraft ventilation flow in microgravity. The different colors represent different chemical reactions within the flame. The blue areas are caused by chemiluminescence (light produced by a chemical reaction.) The white, yellow and orange regions are due to glowing soot within the flame zone. #

Microgravity Water Spheres
Here astronaut Don Pettit demonstrates the effects of rotation on a sphere of water in microgravity. Bubbles, being less dense than water, congregate in the middle of the sphere along its axis of rotation. Tea leaves, which are denser than the water, are thrown to the outside; this is the same concept used in a centrifuge for separating samples.
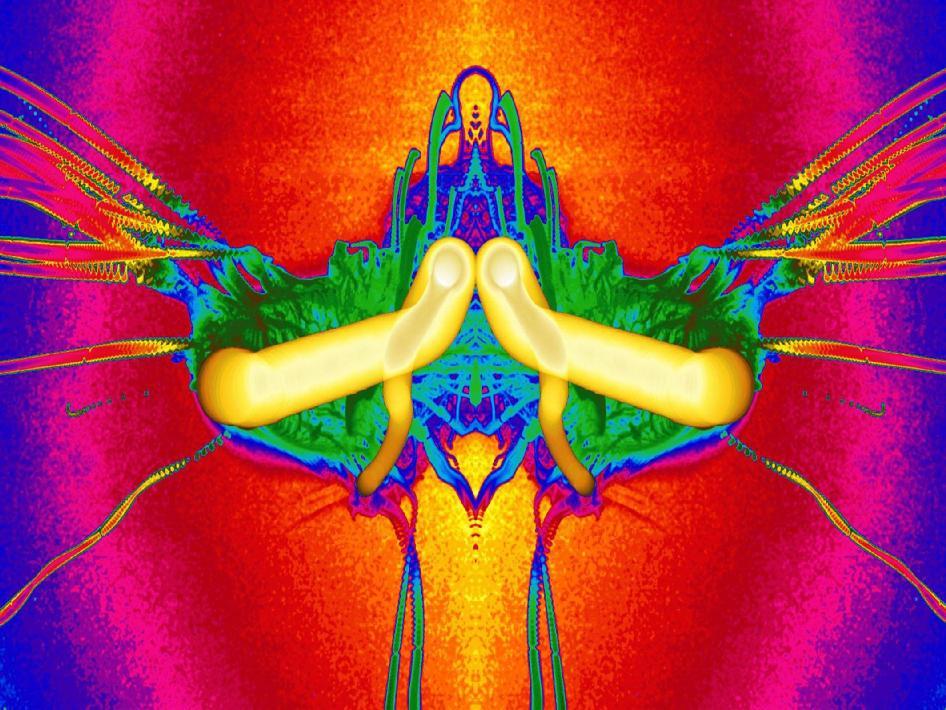
Microgravity Combustion
Combustion in microgravity is markedly different than that on earth, due to a lack of buoyant convection. The combustion of a droplet of heptane is shown here as a composite image. The bright yellow structure shows the path of the droplet, which gets smaller as it burns. The green structures show the initial development of soot, which eventually streams outward as long streaks. # (submitted by jshoer)

Boiling in Microgravity
This week’s edition of the ISS research blog focuses on the Boiling Experiment Facility (BXF) and the goals of unlocking the secrets of boiling in microgravity. Without gravity to provide buoyant convection, boiling in space tends to produce one giant bubble instead of the hundreds of tiny ones we’re accustomed to seeing on our stoves. According to Dr. Tara Ruttley:
TheBoiling Experiment Facility or BXF, which launched on STS-133 in February 2010, will enable scientists to perform in-depth studies of the complexities involved in bubble formation as a result of heat transfer. For instance, what roles do surface tension and evaporation play during nucleate boiling when buoyancy and convection are not in the equation? What about the variations in the properties of the heating surface? By controlling for gravity while on the International Space Station, scientists can investigate the various elements of boiling, thus potentially driving improved cooling system designs. Improved efficiency in cooling technology can lead to positive impacts on the global economy and environment; two hot topics that have much to gain from boiling in space.
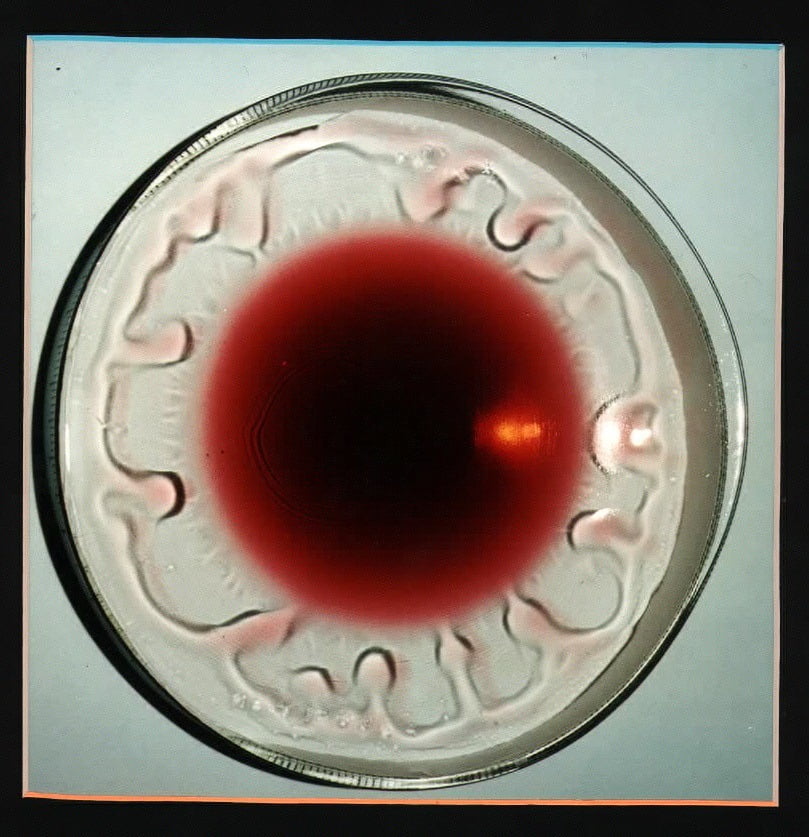
Microgravity Marangoni
Astronauts are preparing an experiment on the Marangoni effect, in which a variation in surface tension can cause mass flow, for flight aboard the International Space Station. The effect, also responsible for causing tears of wine, will benefit from study in microgravity because competing effects like gravity-induced sedimentation and buoyant convection will be negligible. Astronaut Ron Garan reports more on the upcoming experiment on the Fragile Oasis blog.

Microgravity Water Films
In this video astronaut Don Pettit demonstrates some interesting laminar flow effects using a water film in microgravity. By using a film, fluid motion is essentially confined to two dimensions. This is important because it prohibits the development of turbulence, which is a purely three-dimensional phenomenon. Doing the experiment in microgravity allows Pettit to leave the experiment for a long period of time without buoyant effects or similar disturbances. When he first stirs the film, the tracer particles show some signs of what looks like turbulent mixing, but soon the film rotates uniformly with streaks of gray caused by different concentrations of tracer particles. Pettit notes that he allowed the film to rotate overnight and it eventually all turned milky white. This is the effect of molecular diffusion of the tracer particles; without turbulence, the only way for mixing to occur is through the random motion of molecules. See more of Pettit’s Saturday Morning Science videos for additional microgravity fluid mechanics.