Surface tension is the result of an imbalance between intermolecular forces near an interface. Imagine a water molecule far from the surface; it is surrounded on all sides by other water molecules and feels each of those pulling on it. Since all the nearby molecules are water, the tugs from every direction balance and there is no net force. Now imagine that water molecule near the air interface. Instead of being influenced on all sides by water, our molecule now feels water in some directions and air molecules in another. The water molecules tug harder on it than air, leaving a net force that pulls along the interface. This is surface tension, and, for a liquid-gas interface, it behaves somewhat like an elastic sheet. Surface tension is even strong enough to let a jet of soap solution bounce repeatedly off a soap film. Each bounce deforms the interface, like a trampoline dimpling when someone jumps on it, but surface tension keeps the interface taut enough for the jet to skip off without breaking it. (Image credit: C. Kalelkar and S. Phansalkar, source)
Tag: surface tension

Detergency
Have you ever wondered just how detergents are able to get grease and oil off a surface? This simple example demonstrates one method. In the top image, a drop of oil sits attached to a solid surface; both are immersed in water. An eyedropper injects a surfactant chemical near the oil drop. This lowers the surface tension of the surrounding water and allows the mixture to better wet the solid. That eats away at the oil drop’s contact with the surface. It takes awhile – the middle animation is drastically sped up – but the oil droplet maintains less and less contact with the surface as the surfactant works. Eventually, in the bottom image, most of the oil drop detaches from the surface and floats away. (Image credits: C. Kalelkar and A. Sahni, source)

Convection
Blue paint in alcohol forms an array of polygonal convection cells. We’re accustomed to associating convection with temperature differences; patterns like the one above are seen in hot cooking oil, cocoa, and even on Pluto. In all of those cases, temperature differences are a defining feature, but they are not the fundamental driver of the fluid behavior. The most important factors – both in those cases and the present one – are density and surface tension variations. Changing temperature affects both of these factors, which is why its so often seen in Benard-Marangoni convection.
For the paint-in-alcohol, density and surface tension differences are inherent to the two fluids. Because alcohol is volatile and evaporates quickly, its concentration is constantly changing, which in turn changes the local surface tension. Areas of higher surface tension pull on those of lower surface tension; this draws fluid from the center of each cell toward the perimeter. At the same time, alcohol evaporating at the surface changes the density of the fluid. As it loses alcohol and becomes denser, it sinks at the edges of the cell. Below the surface, it will absorb more alcohol, become lighter, and eventually rise at the cell center, continuing the convective process. (Image credit: Beauty of Science, source)

Elastic Bounces
A rigid ball accelerated by a moving surface can only ever move as fast as the surface propelling it. But that’s not true for squishy objects like a water droplet. The composite image above shows the trajectory of a water droplet launched from a moving superhydrophobic surface. As the surface starts rising, it squishes the droplet like a pancake, triggering a deformation cycle where the droplet will squish and extend repeatedly. How quickly the drop changes shape depends on factors like its size and surface tension. The researchers found that a droplet’s launch was strongly affected by the ratio of the droplet’s shape-changing frequency and the frequency of the plate’s motion. When the drop’s shape changed three times faster than the surface’s motion, it would catapult off the surface with 250% of the kinetic energy of a rigid ball!
Launching elastic balls works the exact same way as droplets, indicating that the phenomenon depends on the way the projectiles deform. The process is similar to jumping on a trampoline. If a trampolinist times her jump just right, she’ll get more energy from the trampoline and fly higher. The droplet does the same when its deformation is properly tuned to its catapult. (Image credit: C. Raufaste et al.; via APS Physics; submitted by Kam-Yung Soh)

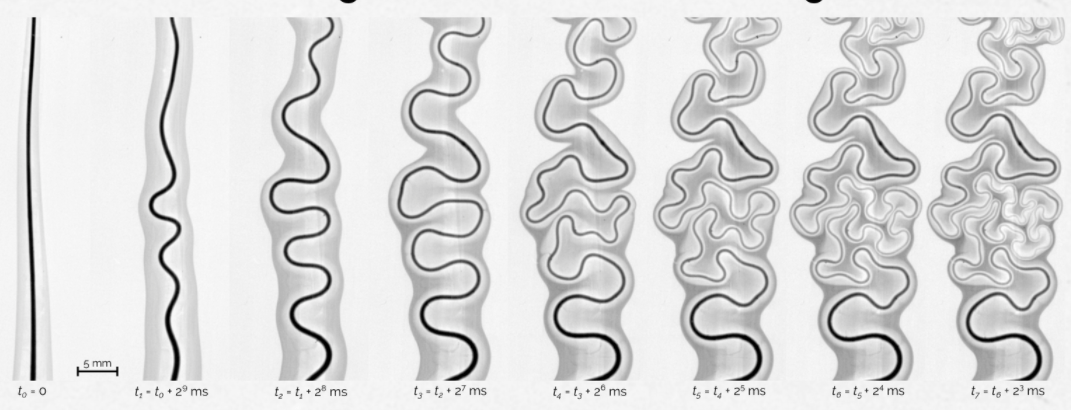
Wriggling Threads
A thread of mineral oil laid across a pool of water twists and turns like a river run wild. Because the oil has a lower surface tension than the water, Marangoni forces spread it outward (far left). Small variations in the thread make the areas of highest oil concentration start to bend just a bit. Inside the bends, the gradient of surface tension – the difference between the lowest and highest surface tensions – is very high, which pulls at these regions more than others. So bends beget more bends, causing the entire thread to wrinkle. Although the behavior is driven by a completely different process than the one that causes rivers to meander, the end result looks remarkably similar; this is because, in both cases, forces act to make each bend increasingly sinuous. (Image credit: B. Néel et al., source)
Editor’s note: Starting tomorrow I’ll be on a trip that takes me out of range of the Internet until next week. Regular posts are queued up and should post as usual, but we’ll all have to trust Tumblr to handle everything because I won’t be able to check. Thanks!

Life at the Interface
Water striders are masters of life at the interface of water and air. Their spindly legs are skinnier than the capillary length of water, meaning that, at their size, surface tension is strong enough to overcome gravitational effects. Thus, their feet leave dimples on the interface, but the water itself holds them up. To keep from getting accidentally drenched (and thus weighed down), the striders are covered in tiny hairs that trap a layer of air that makes them hydrophobic or water-repellent. To get around, these masters of the interface use their middle legs in a manner similar to oars. They push against the dimple around their legs, which generates vortices under the surface and helps propel them. Even more impressive, the water strider can jump off the surface, a feat that requires remarkable adaptation in order to maximize the jump without breaking surface tension. (Video credit: Deep Look)

Paintball Collisions
In their latest video, the Slow Mo Guys collide paintballs in mid-air, creating some pretty great paint splashes. The high-speed video does a nice job of revealing some of the typical stages a splash goes through. Initially, the paint spreads in a liquid sheet. As it expands and (necessarily) thins, holes form and grow, driving the paint into string-like ligaments. These ligaments are also stretching and eventually break up into an spray of droplets, much like the jet dripping from your faucet does. If you’d like to see some more awesome high-speed liquid collisions, check out what happens when a solid projectile hits a falling drop and then look at when a laser pulse hits a droplet. (Image and video credit: The Slow Mo Guys; submitted by Omar M.)

Burning a Rocket Underwater
In a recent video, Warped Perception filmed a model rocket engine firing underwater. Firstly, it’s no surprise that the engine would still operate underwater (after its wax waterproofing). The solid propellant inside the engine is a mixture of fuel and oxidizer, so it has all the oxygen it needs. Fluid dynamically speaking, though, this high-speed footage is just gorgeous.
Ignition starts at about 3:22 with some cavitation as the exhaust gases start flowing. Notice how that initial bubble dimples the surface when it rises (3:48). At the same time, the expanding exhaust on the right side of the tank is forcing the water level higher on that side, triggering an overflow starting at about 3:55. At this point, the splashes start to obscure the engine somewhat, but that’s okay. Watch that sheet of liquid; it develops a thicker rim edge and starts forming ligaments around 4:10. Thanks to surface tension and the Plateau-Rayleigh instability, those ligaments start breaking into droplets (4:20). A couple seconds later, holes form in the liquid sheet, triggering a larger breakdown. By 4:45, you can see smoke-filled bubbles getting swept along by the splash, and larger holes are nucleating in that sheet.

The second set of fireworks comes around 5:42, when the parachute ejection charge triggers. That second explosive triggers a big cavitation bubble and shock wave that utterly destroys the tank. If you look closely, you can see the cavitation bubble collapse and rebound as the pressure tries to adjust, but by that point, the tank is already falling. Really spectacular stuff! (Video and image credit: Warped Perception)

Equatorial Streaming


Here you see a millimeter-sized droplet suspended in a fluid that is more electrically conductive than it. When exposed to a high DC electric field, the suspended drop begins to flatten. A thin rim of fluid extends from the drop’s midplane in an instability called “equatorial streaming”. As seen in the close-up animation, the rim breaks off the droplet into rings, which are themselves broken into micrometer-sized droplets thanks to surface tension. The result is that the original droplet is torn into a cloud of droplets a factor of a thousand smaller. This technique could be great for generating emulsions of immiscible liquids–think vinaigrette dressing but with less shaking! (Image credit: Q. Brosseau and P. Vlahovska, source)

Capillary Action in Microgravity
On Earth, gravity dominates over many fluid effects, but in microgravity a different picture emerges. This animation shows a two-channel apparatus partially filled with silicone oil being dropped. While in free-fall, the liquid experiences microgravity conditions and the height of the fluid in the two connected channels changes. The oil meniscus climbs up the walls of the tubes thanks to capillary action. This is the result of intermolecular forces between the liquid and solid walls. Capillary action is most effective in narrow tubes where surface tension and the adhesion between the liquid and solid can actually propel liquid up the walls, as seen here. On Earth we mostly ignore capillary action, except in very small spaces, but for space systems, it is a major force to reckon with in designing flows. (Image credit: NASA Glenn Research Center, source)