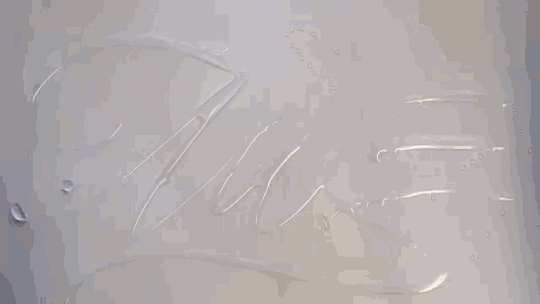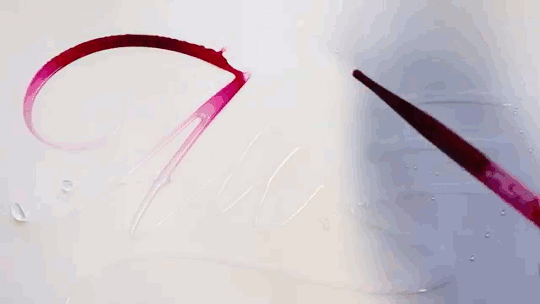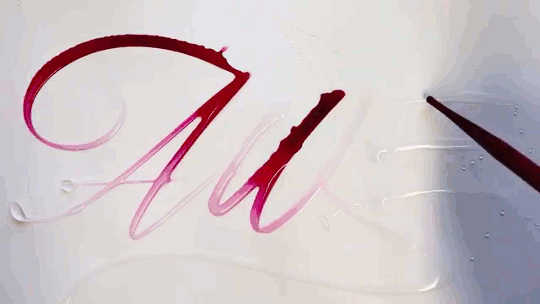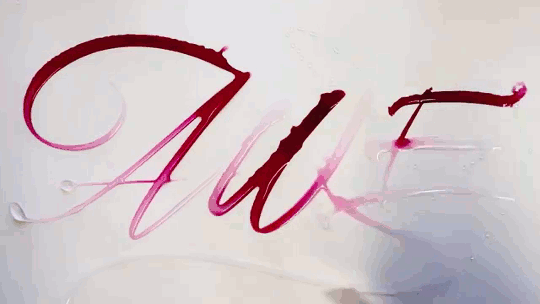Pay attention after a rainfall, and you may notice beads of water gathering in the corners of a spider’s web or along the leaves of a cypress tree (bottom right). Look closely and you’ll notice that the largest droplets don’t form along a straight fiber. Instead they nestle into the corners of a bent fiber (top image). Researchers recently characterized this corner mechanism and found that the angle at which the largest droplets form is about 36 degrees. This angle provides the optimal conditions for capillary action and surface tension to hold large drops in place. At smaller angles, a growing droplet’s weight pulls it down until the thin film holding the droplet near the top ruptures and the droplet falls. At larger angles, a heavy droplet will slowly detach from one side of its fiber and shift toward the other side until its weight is too great for the wetted length of fiber to hold. Then it detaches completely and falls. (Research and image credit: Z. Pan et al.; via T. Truscott)
Tag: surface tension

Wrapping Droplets
Future efforts for targeted drug delivery may require encapsulating droplets before transporting them to their final location. One method for encapsulation is wrapping a drop in a thin, solid sheet. Previously, we saw that drops can wrap themselves with a little outside assistance, but here the drops achieve that same feat on their own, using the energy of droplet impact to wrap liquids.
Here’s how it works: float a thin sheet on a bath of a liquid like water, then let an oil drop fall into the bath. Its impact deforms the air-water interface and, with a sufficiently energetic impact, causes the oil droplet to pinch off. The flexible sheet wraps around the droplet, and the encapsulated droplet sinks due to gravity. The shape of the final drop depends on the sheet’s initial geometry. The researchers have successfully used circular, triangular, and cross-shaped sheets to wrap droplets. Check out the original paper or the video below for more. (Image and research credit: D. Kumar et al.; video credit: Science)

Paint Balloons
The Slow Mo Guys have a history of personal sacrifice in the name of cool high-speed footage, and their Super Slow Show is no exception. In a recent segment, both Dan and Gav were knocked flat by giant swinging balloons of paint, and, as you might expect, the splashes are spectacular. The speed is just right for some of the paint to form nice sheets before momentum pulls them into long ligaments. Eventually, that momentum overcomes surface tension’s ability to keep the paint together, and the paint separates into droplets, which, as you see below, rain down on the hapless victims. (Video and image credit: The Slow Mo Guys)

Water Calligraphy
Artist Seb Lester creates calligraphy using ink and water, but not in the way you might expect. After writing in water, the artist applies ink a drop at a time, allowing fluid forces to spread it. There are a few effects at play here. Molecular diffusion – the random motion of molecules – can help two fluids mix, but it’s an extremely slow process. The fast, dramatic spread of ink seen in the video is more likely a Marangoni effect. The water and ink have different surface tensions, creating a gradient in surface tension that depends on the relative concentration of the two fluids. Gradients in surface tension create flow, which is why the ink spreads most quickly when it’s applied in an area that’s pure water. For similar physics, check out maze-solving soaps and the title sequence for “Marco Polo”. (Video and image credit: S. Lester, source; via Gizmodo)

Swimming Microdroplets
Simple systems can sometimes have surprisingly complex behaviors. In this video, the Lutetium Project outlines a scheme for swimming microdroplets. Most of the droplets shown are just water, but they’re released into a chamber filled with a mixture of oil and surfactants. All flow through the chamber is shut off, but the droplets swim around in complicated, disordered patterns anyway. To see why, we have to zoom way in. The surfactant molecules in the oil cluster around the droplets, orienting so that their hydrophobic parts are in the oil and their hydrophilic parts point toward the water. They actually draw some of the water out of the droplets. This creates a variation in surface tension that causes Marangoni flow, making the droplets swim. Over time, the droplets shrink and slow down as the surfactants pull away more and more of their water and the variations in surface tension get smaller. (Image and video credit: The Lutetium Project; research credit: Z. Izri et al.)


Solving Mazes
Earlier this fall, I attempted my first corn maze. It didn’t work out very well. Early on I unknowingly cut through an area meant to be impassable and thus ended up missing the majority of the maze. Soap, as it turns out, is a much better maze-solver, taking nary a false turn as it heads inexorably to the exit. The secret to soap’s maze-solving prowess is the Marangoni effect.
Soap has a lower surface tension than the milk that makes up the maze, which causes an imbalance in the forces at the surface of the liquid. That imbalance causes a flow in the direction of higher surface tension; in other words, it tends to pull the soap molecules in the direction of the highest milk concentration. But that explains why the soap moves, not how it knows the right path to take. It turns out that there’s another factor at work. Balancing gravitational forces and surface tension forces shows that the soap tends to spread toward the path with the largest surface area ahead. That’s the maze exit, so Marangoni forces pull the soap right to the way out! (Video credit: F. Temprano-Coleto et al.; research credit: F. Peaudecerf et al.)
ETA: Based on the latest research results, gravity may play less of a role than originally thought. Instead, it seems as though the soap chooses its path in part through pre-existing background levels of surfactant. As the dye advances, it compresses the background surfactant, decreasing the local surface tension until it is in equilibrium with dyed area. Because longer paths take longer to reach that equilibrium, the dye spreads preferentially toward the largest surface area.


Blowing Bubbles in Space
Blowing bubbles in your fruit juice is a bad idea when you’re in space, as astronaut Jack Fischer demonstrates. On Earth, gravity dominates water’s behavior, except when things are very small. But in microgravity, a liquid’s other characteristics become more obvious. Adhesion between the straw and juice guides it up and onto Fischer’s face. Surface tension is strong enough to hold the expanding juice bubble together. Capillary action, the ability of fluids to climb up narrow spaces, is far more apparent in microgravity as well, although it’s not important for this demo. We sometimes forget how powerful these forces can be, but microgravity is a good reminder that fluids are more complicated than we think. (Image credit: J. Fischer, source)

Convection Without Heat
Glycerol is a sweet, highly viscous fluid that’s very good at absorbing moisture from the ambient air. That’s why a drop of pure glycerol in laboratory conditions quickly develops convection cells – even when upside-down, as shown above. This is not the picture of Bénard-Marangoni convection we’re used to. There’s no temperature or density change involved; in fact, there’s no buoyancy involved at all! This convection is driven entirely by surface tension. As glycerol at the surface absorbs moisture, its surface tension decreases. This generates flow from the center of a cell toward its exterior, where the surface tension is higher. Conservation of mass, also known as continuity, requires that fresh, undiluted glycerol get pulled up in the wake of this flow. It, too, absorbs moisture and the process continues. (Image credit: S. Shin et al., pdf)

The Cheerios Effect
You’ve probably noticed that cereal clumps together in your breakfast bowl, but you may not have given much thought as to why. This tendency for objects at an interface to attract is known as the Cheerios effect, although it happens in more than just cereal, as Joe Hanson from It’s Okay to Be Smart explains. The effect is a combination of buoyancy, gravity, and surface tension acting in concert.
When air, a liquid, and a solid meet, they form a meniscus, the curvature of which depends on characteristics of their interaction. Light, buoyant cereal and the walls of your bowl both have upward-curving menisci. Denser objects, like the tacks shown below, stay at the surface only because surface tension holds them up. Their meniscus curves downward.
Objects with a similar meniscus curvature will attract. For cereal approaching a wall, the light Cheerio is buoyant enough that there’s an upward force on it, but it’s constrained to stay at the interface. It cannot rise, but that buoyancy is enough to let it climb the meniscus at the wall. The two tacks attract one another for similar reasons, except this time their weight helps them fall into one another. Check out the full video to see more examples of this effect in nature! (Video and image credit: It’s Okay to Be Smart; research credit: D. Vella and L. Mahadevan, pdf)



Building Liquid Circuits

Building microfluidic circuits is generally a multi-day process, requiring a clean room and specialized manufacturing equipment. A new study suggests a quicker alternative using fluid walls to define the circuit instead of solid ones. The authors refer to their technique as “Freestyle Fluidics”. As seen above, the shape of the circuit is printed in the operating fluid, then covered by a layer of immiscible, transparent fluid. This outer layer help prevent evaporation. Underneath, the circuit holds its shape due to interfacial forces pinning it in place. Those same forces can be used to passively drive flow in the circuit, as shown in the lower animation, where fluid is pumped from one droplet to the other by pressure differences due to curvature. Changing the width of connecting channels can also direct flow in the circuits. This technique offers better biocompatibility than conventional microfluidic circuits, and the authors hope that this, along with simplified manufacturing, will help the technique spread. (Image and research credit: E. Walsh et al., source)