The upside-down jellyfish Cassiopea lives along the sea bottom in coastal regions. As its name suggests, the jellyfish rests upside-down with its bell against the sea floor and its frilly oral arms pointed upward. This jellyfish is a filter feeder, and it draws water up and through its arms by pulsing its bell. The video above visualizes this flow using dye. Each pulse propels fluid up through the arms and draws in fresh water from the surroundings. The frilly arms break up any large vortices from the pulsed flow and diffuse the filtered water as it moves upward. (Video credit: Applied Fluid Mechanics Laboratory at Oklahoma State University)
Tag: science

Reader Question: Lift
everyonelikespotatissallad asks:
so, how is lift actually generated? i’ve been going through Anderson’s Introduction to Flight (6th Ed.) and while it offers the derivation of various equations very thoroughly, it barely touches on why lift is generated, or how camber contributes to the increase of C(L)
This is a really good question to ask. There are a lot of different explanations for lift out there (and some of the common ones are incorrect). The main thing to know is that a difference in pressure across the wing–low pressure over the top and higher pressure below–creates the net upward force we call lift. It’s when you ask why there’s a pressure difference across the wing that explanations tend to start diverging. To be clear, aerodynamicists don’t disagree about what produces lift – we just tend to argue about which physical explanation (as opposed to just doing the math) makes the most sense. So here are a couple of options:
Newton’s third law states that for every action there is an equal and opposite reaction. If you look at flow over an airfoil, air approaching the airfoil is angled upward, and the air leaving the aifoil is angled downward. In order to change the direction of the air’s flow, the airfoil must have exerted a downward force on the air. By Newton’s third law, this means the air also exerted an upward force–lift–on the airfoil.
The downward force a wing exerts on the air becomes especially obvious when you actually watch the air after a plane passes:

This one can be harder to understand. Circulation is a quantity related to vorticity, and it has to do with how the direction of velocity changes around a closed curve. Circulation creates lift (which I discuss in some more detail here.) How does an airfoil create circulation, though? When an airfoil starts at rest, there is no vorticity and no circulation. As you see in the video above, as soon as the airfoil moves, it generates a starting vortex. In order for the total circulation to remain zero, this means that the airfoil must carry with it a second, oppositely rotating vortex. For an airfoil moving right to left, that carried vortex will spin clockwise, imparting a larger velocity to air flowing over the top of the wing and slowing down the air that moves under the wing. From Bernoulli’s principle, we know that faster moving air has a lower pressure, so this explains why the air pressure is lower over the top of the wing.
Asymmetric Flow and Bernoulli’s Principle

There are two basic types of airfoils – symmetric ones (like the one in the first picture above) and asymmetric, or cambered, airfoils (like the one in the image immediately above this). Symmetric airfoils only generate lift when at an angle of attack. Otherwise, the flow around them is symmetric and there’s no pressure difference and no lift. Cambered airfoils, by virtue of their asymmetry, can generate lift at zero angle of attack. Their variations in curvature cause air flowing around them to experience different forces, which in turn causes differing pressures along the top and the bottom of the airfoil surface. A fluid particle that travels over the upper surface encounters a large radius of curvature, which strongly accelerates the fluid and creates fast, low-pressure flow. Air moving across the bottom surface experiences a lesser curvature, does not accelerate as much, and, therefore, remains slower and at a higher pressure compared to the upper surface.
(Image credit: M. Belisle/Wikimedia; National Geographic/BBC2; O. Cleynen/Wikimedia; video credit: J. Capecelatro et al.)

Coriolis
There’s an infamous supposition about drains swirling one way in the Northern Hemisphere and the other way in the Southern Hemisphere. Destin from Smarter Every Day and Derek from Veritasium have put the claim to the test with experiments on either side of the globe. First, go here and watch their synchronized videos side-by-side. (To synchronize, start the left video and pause it at the sync point. Then start the second video and unpause the first video when the second video hits the sync point.) I’ll wait here.
…
That was awesome, right?! The demonstration doesn’t work with toilets because they’re driven by the placement of jets around the circumference. And your bathtub doesn’t usually work either because any residual vorticity in the tub gets magnified by conservation of angular momentum as it drains. It’s like a spinning ice skater pulling their arms in; the rotation speeds up. So, to get around that problem, Destin and Derek let their pools sit for a day to damp out any motion before draining. At that point, the Coriolis effect is strong enough to cause the pools to rotate in opposite directions when drained. You may wonder why the effect is so slight for the pools when it’s pretty stark with hurricanes and cyclones. The answer is a matter of scale. The pools are perhaps 2 meters wide, which means that the difference in latitude across the the pool is very slight and therefore, the differential speed imparted by the Earth’s rotation is also very small. Because hurricanes and cyclones are much larger, they experience stronger influence from the Coriolis effect. (Image credits: Smarter Every Day/Veritasium; via It’s Okay To Be Smart)

Convection from a Heat Source
Convection is a major driver in many flows in nature. In this film, the UCLA Spinlab demonstrates buoyant convection caused by a local heat source. They deposit dye on a submerged, continuously heated plate, then observe as the dye slowly rises with the heated (lower density) fluid. The surface forms a cap for the rising dye, which then spreads horizontally. Qualitatively similar flows can be seen in nature over volcanic eruptions or in thunderstorms when clouds reach the troposphere or a capping inversion. Be sure to check out the rest of the Spinlab’s videos. (Video credit: UCLA Spinlab; submitted by Jon B.)

Jumps in Stratified Flows
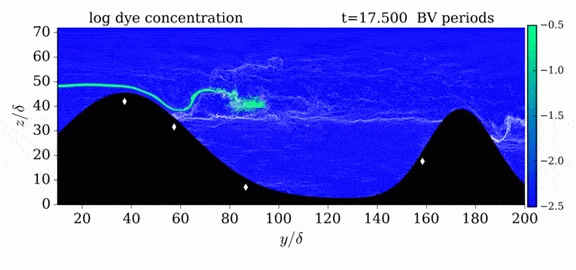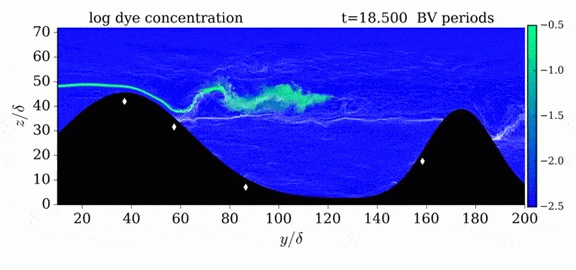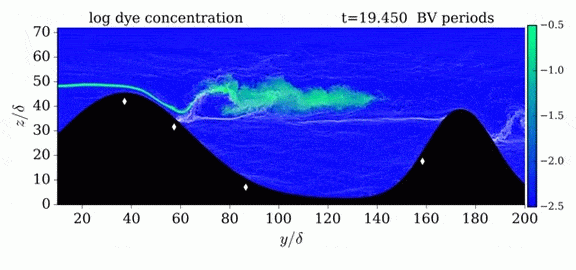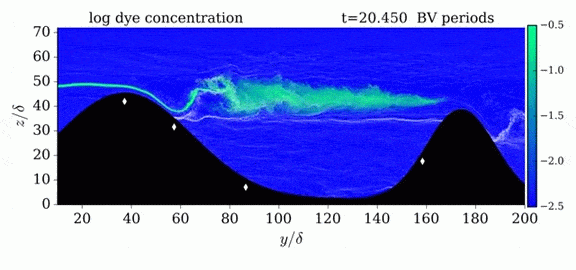
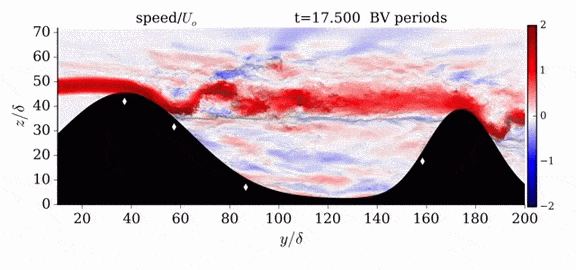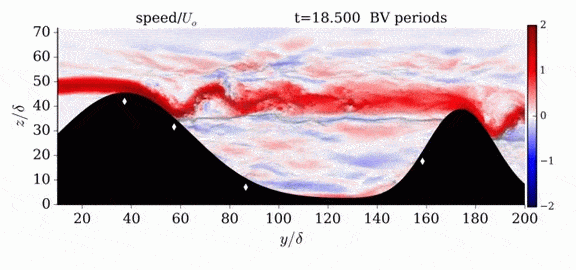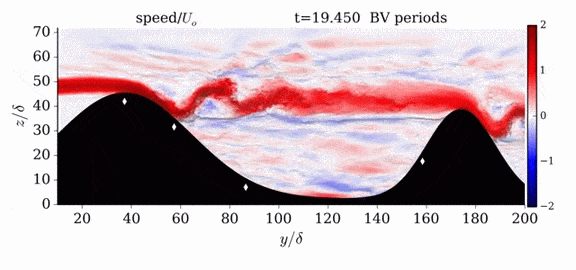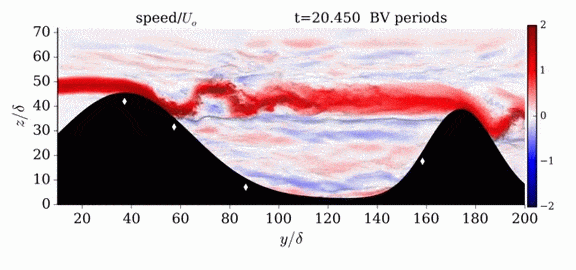
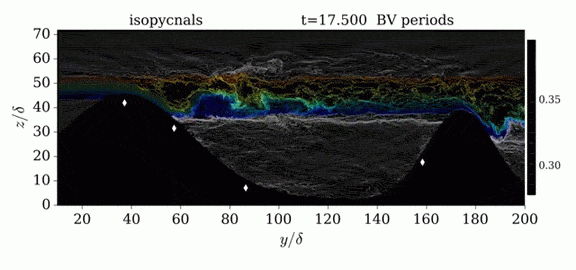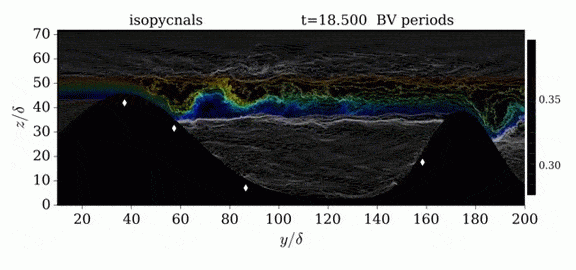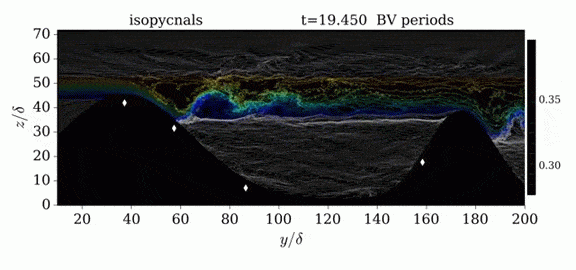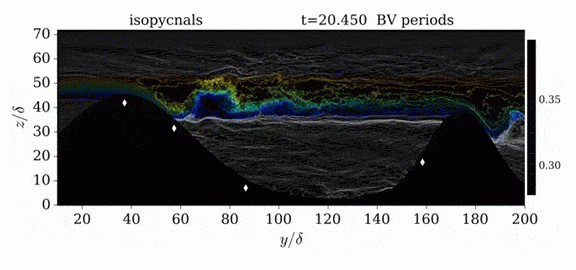
One of the factors that complicates geophysical flows is that both the atmosphere and the ocean are stratified fluids with many stacked layers of differing densities. These variations in density can generate instabilities, trap rising or sinking fluids, and transmit waves. The animations above show flow over two ridges with dye visualization (top), velocity (middle), and contours of density (bottom). The upstream influence of the left ridge creates a smooth, focused flow that quickly becomes turbulent after the crest. The jet rebounds as a turbulent hydraulic jump before slowing again upstream of the second ridge. Like the first ridge, the second ridge also generates a hydraulic jump on the lee side. Clearly both stratification and the local topography play a big role in how air moves over and between the ridges. If prevailing winds favor these kinds of flows, it can help generate local microclimates. (Image credit and submission: K. Winters, source videos)

Rain-spread Pathogens
Like humans, plants can spread pathogens to one another. Although scientists had observed correlations between rainfall and the spread of diseases among plants, this study is one of the first to look at the fluid dynamics of leaf and rainfall interaction. When a raindrop hits a leaf, it doesn’t simply splash as it would against an immobile surface. The impact of the drop deforms the leaf, and the plant’s rebound significantly affects the trajectory and size of the resulting droplets. Depending on factors like the leaf’s stiffness, a large drop, carrying many pathogens, may rebound and splatter onto a neighboring leaf. Other leaves tend to catapult out many smaller droplets, which may fly farther afield but carry fewer pathogens. For more, check out the press release or the original research paper. (Video credit: Massachusetts Institute of Technology; research credit: Bourouiba Research Group)

Kitchen Fluid Dynamics
The kitchen is a fantastic place to witness the everyday beauty of fluid dynamics. Daria Khoroshavina and Olga Kolesnikova capture these delectable cooking-related GIFs on their Buttery Planet Tumblr. From pouring cream to drizzling syrup, there are countless examples of fluids in daily life. Check out their site for more awesome images and be sure to keep your eyes open for great examples of fluid behavior in your day-to-day life. (Image credits: Buttery Planet; via Colossal)

Get Your Own Space Coffee Cup
A few weeks ago, we reported on the espresso machine NASA and the ESA sent to astronauts aboard ISS. The Capillary Beverage Experiment, known colloquially as the “Space Coffee Cup”, is an accompanying project that aims to use our understanding of fluid behavior in microgravity to design an open cup that simulates earthbound drinking experiences. As you can see above, astronauts are already enjoying drinks with it. The cup’s special shape is optimized so that surface tension can replace the role gravity plays in drinking on Earth. Where we pour drinks on Earth, the cup wicks liquid to the spout using surface-tension-driven capillary action. Right now there are only a handful of 3D printed cups on-orbit and here on Earth, but the company that designed them wants to manufacture glass versions for use here on the ground. So if you’d like your own space coffee cup, be sure to check out their Kickstarter campaign! (Video credit: IRPI LLC; image credit: NASA/IRPI LLC; Kickstarter project link)
ETA: To those who have been asking, that’s European astronaut Samantha Cristoforetti, who is (clearly) a Star Trek fan. I believe she’s doing a tribute to Captain Janeway’s coffee. (Black.)

Coalescence Cascade
The simple coalescence of a drop with a pool is more complicated than the human eye can capture. Fortunately, we have high-speed cameras. Here a droplet coalesces by what is known as the coalescence cascade. Because it has been dropped with very little momentum, the droplet will initially bounce, then seem to settle like a bead on the surface. A tiny film of air separates the drop and the pool at this point. When that air drains away, the drop contacts the pool and part–but not all!–of it coalesces. Surface tension snaps the remainder into a smaller droplet which follows the same pattern: bounce, settle, drain, partially coalesce. This continues until the remaining droplet is so small that it can be coalesced completely. (Image credit: Laboratory of Porous Media and Thermophysical Properties, source video)

Re-lighting a Candle
When you blow out a candle, you can re-light the wick using the smoke trail left behind. This is a topic we’ve discussed before, but I’m thrilled to finally see the process in true high-speed, thanks to the Slow Mo Guys. The plume that rises from the extinguished candle is an atomized mixture of fuel (wax) and air. When you bring a new combustion source–the match–close enough, that mixture ignites and the flame spreads downward back to the wick. (Video credit: The Slow Mo Guys)