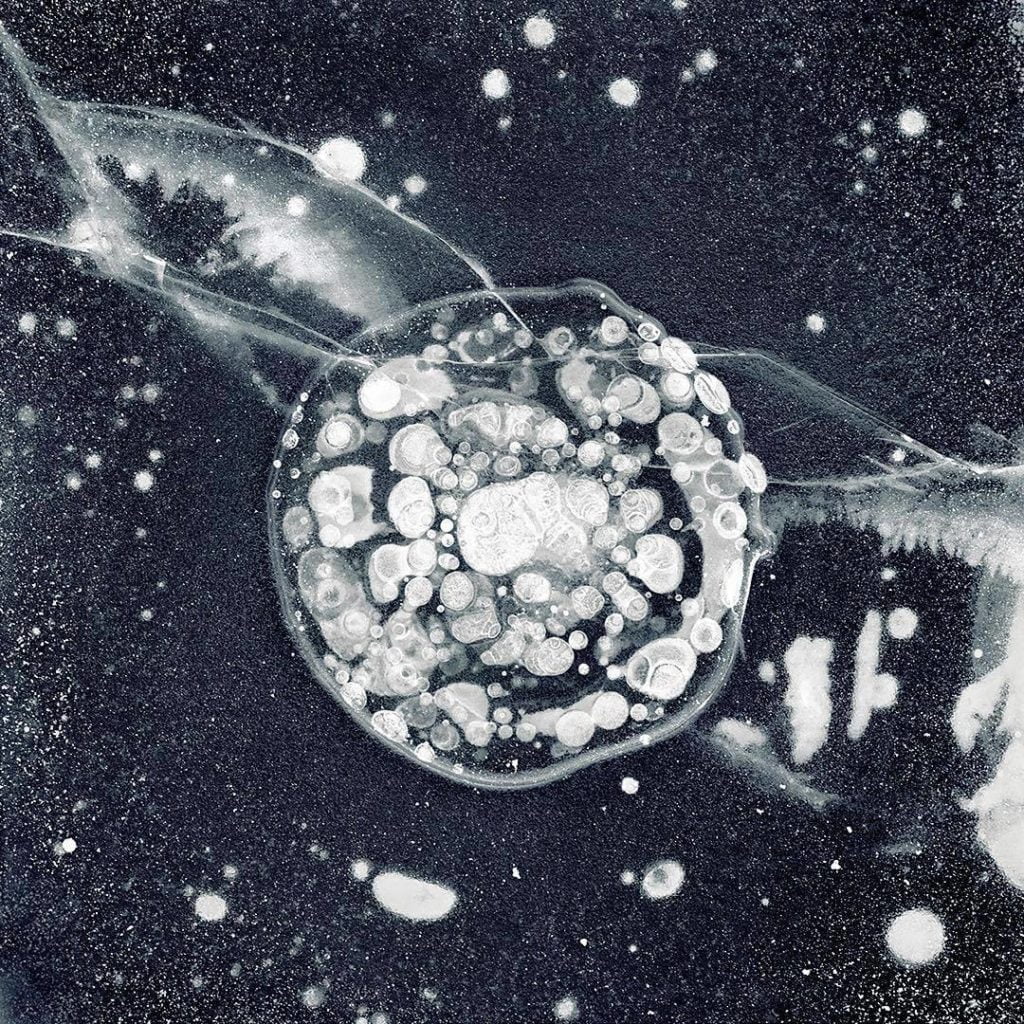
Leidenfrost drops – liquid drops that levitate on a layer of their own vapor over a hot surface – have been all the rage in recent years. We’ve seen how they can be guided, trapped, and self-propelled. What you see here is a bit different. This is a droplet of room-temperature ethanol deposited on a bath of liquid nitrogen. What levitates the droplet in this case is vaporous nitrogen evaporating from the bath.
The droplet is quickly cooling down; it freezes after its second or third bounce off the side walls of the beaker. What causes the droplet to self-propel is an asymmetry of the thin vapor layer beneath the droplet. As soon as some instability causes a slight difference in the thickness of the vapor layer, that triggers the propulsion, which the drop maintains even after freezing. (Image and research credit: A. Gauthier et al.)