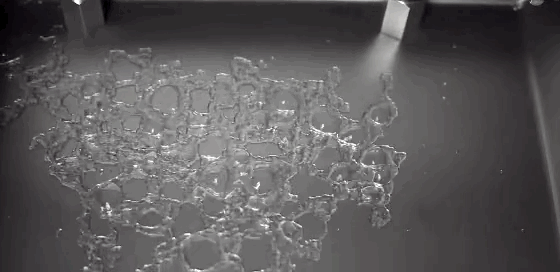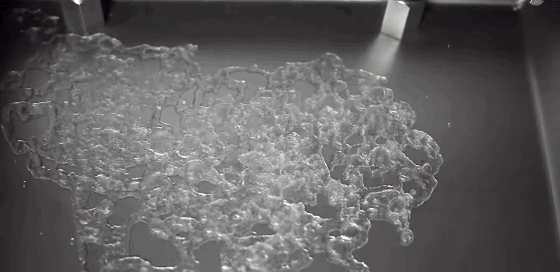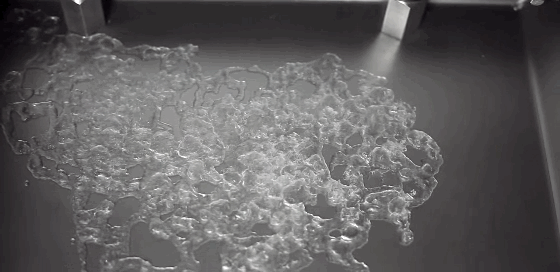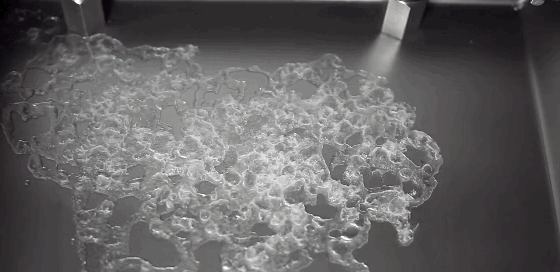Watch closely as a droplet freezes on a cold surface, and you’ll observe something surprising. First, a freeze front will appear, traveling upward from the substrate. It curves slightly near the edges, leaving a liquid cap atop the frozen drop. But, as we’ve all discovered, water expands as it freezes. We can watch the drop freezing and see that the water isn’t expanding radially. Instead, the water expands vertically, forming a sharp tip or cusp just as the drop freezes completely. Remarkably, the geometry of the final tip doesn’t depend on the temperature of the substrate or on the wetting contact angle. (Video credit: L. Posada)
Tag: freezing

Growing Icicles
For those from colder climates, icicles are a familiar part of winter. They come in a multitude of shapes and sizes, many of which have been captured and cataloged in the Icicle Atlas project. The site contains images, videos, and measurements of more than 230 icicles grown in the lab over the course of four years of research into how and why icicles form the way they do. One interesting result of the work was the discovery that the ripples commonly found on icicles are directly related to impurities. Icicles grown with pure water remain smooth, and only those with ionic impurities, like salt, develop ripples. Check out more images and icicle research at the Atlas. (Video credit: A. Chen and S. Morris/The Icicle Atlas; via Discover)

Grow Your Own Snowflakes
If your Christmas holiday was a little too green (like mine was), Science Friday has just the activity for you – grow your own snowflakes! With a few materials you probably already have and some dry ice from the store, you can grow and observe ice crystals at home. Although these crystals form from water vapor instead of water droplets like proper snowflakes, they do exhibit different structures depending on temperature and humidity, just the way natural snowflakes do. (Video credit: Science Friday/F. Lichtman)

Growing Snowflakes
It’s easy to miss the beauty of a snowflake if you don’t take a close look. These tiny crystals form when water freezes onto a dust particle or other nucleation site, and they grow as water vapor freezes on to the nucleus. The structured appearance of a snowflake comes from the bonds formed between water molecules, but the exact type and shape of crystal formed–not all snowflakes are six-sided!–depends on the local temperature and humidity during freezing. This microscopic timelapse video by Vyacheslav Ivanov lets you watch the process in action. (Video credit: V. Ivanov; via io9)

Bounce or Freeze?
Icing is a major problem for aircraft. When ice builds up on the leading edge of a wing it creates major disruptions in flow around the wing and can lead to a loss of flight control. One of the important factors in predicting and controlling ice building up is knowing when and where water droplets will freeze. The video above shows how surface conditions on the wing affect how an impacting droplet freezes. On a subzero hydrophilic surface, a falling droplet spreads and freezes over a wide area, which would hasten ice buildup. A hydrophobic surface is slightly better, with the droplet freezing over a smaller area, whereas a superhydrophobic surface shows no ice buildup. Unfortunately, at present superhydrophobic surfaces and surface treatments are extremely delicate, making them unsuitable for use on aircraft leading edges. (Video credit: G. Finlay)

Does Liquid in a Vacuum Boil or Freeze?
What happens to a liquid in a cold vacuum? Does it boil or freeze? These animations of liquid nitrogen (LN2) in a vacuum chamber demonstrate the answer: first one, then the other! The top image shows an overview of the process. At standard conditions, liquid nitrogen has a boiling point of 77 Kelvin, about 200 degrees C below room temperature; as a result, LN2 boils at room temperature. As pressure is lowered in the vacuum chamber, LN2’s boiling point also decreases. In response, the boiling becomes more vigorous, as seen in the second row of images. This increased boiling hastens the evaporation of the nitrogen, causing the temperature of the remaining LN2 to drop, the same way sweat evaporating cools our bodies. When the temperature drops low enough, the nitrogen freezes, as seen in the third row of images. This freezing happens so quickly that the nitrogen molecules do not form a crystalline lattice. Instead they are an amorphous solid, like glass. As the residual heat of the metal surface warms the solid nitrogen, the molecules realign into a crystalline lattice, causing the snow-like flakes and transition seen in the last image. Water can also form an amorphous ice if frozen quickly enough. In fact, scientists suspect this to be the most common form of water ice in the interstellar medium. (GIF credit: scientificvisuals; original source: Chef Steps, video; h/t to freshphotons)

Pointed Drops
When water droplets sit on a cold substrate, they freeze into a shape with a pointed tip. At first glance, this behavior seems very odd since surface tension usually acts to prevent such sharp protrusions. The shape is, however, a result of water’s expansion as it freezes. The droplet freezes from the substrate upward, with a concave shape to the solidification front. The angle of the point does not depend on the substrate temperature or the wetting angle between the water and surface. Instead, it turns out that this concave front shape and water’s expansion are the key factors that determine the pointed cusp’s angle, and that the final geometry of the cusp is essentially universal. (Video credit: M. Nauenberg; additional research credit: A. Marin et al.)

Frozen Bubbles

Snowflakes aren’t the only frozen crystals to play with outside in the winter. Photographer Angela Kelly recently posted a series of frozen soap bubbles made by her and her son. In temperatures well below freezing, the thin film of the soap bubble does not survive long before it begins to freeze. The bubbles do not freeze all at once; instead the frost creeps gradually across it. For bubbles sitting on a surface, the ice front expands upward, much the same as with a freezing water drop. Once frozen, the bubbles crack or rip when touched instead of melting and popping. (Photo credit: A. Kelly; via BoredPanda; submitted by jshoer)

Holiday Fluids: Snowflakes
Just about everyone wishes for a White Christmas, but even when that happens, it’s rare to get a good look at the beauty of individual snowflakes. Alexey Kljatov’s macro photography of snowflakes is simply stunning and highlights the incredible variety of forms snowflakes take. A snowflake forms when a water droplet freezes onto dust or other particles and grows as more water vapor freezes onto the initial crystal. The symmetry of the snowflakes, as with any crystal, comes from the internal order of its water molecules. The shape and features that form vary due to the local temperature and humidity level while vapor is freezing onto the crystal. Check out this handy graph showing which shapes form for various situations. Since snowflakes can encounter wildly different conditions on their path to the ground, it’s rare or next-to-impossible to find any two alike. Join us all this week at FYFD as we look at holiday-themed fluid dynamics. (Photo credit: A. Kljatov)

The Evolution of Icicles
The time-lapse video above shows the growth of icicles of various compositions under laboratory conditions. Many icicles in nature exhibit a rippling effect in their shape, which some theories attribute to an effect of lower surface tension in some liquids. Here researchers show the icicle growth of three liquids: pure distilled water, and water with two concentrations of dissolved salt. They found that lowering the surface tension of the freezing liquid with non-ionic surfactants (i.e. not salt) did not produce ripples, but that dissolved ionic impurities like salt strongly affected the growth of ripples. They posit that this may be due to constitutional supercooling, in which growth of the solid-liquid interface is destabilized by the preferential concentration of impurities near the interface. (Video credit: A. S. Chen and S. Morris)