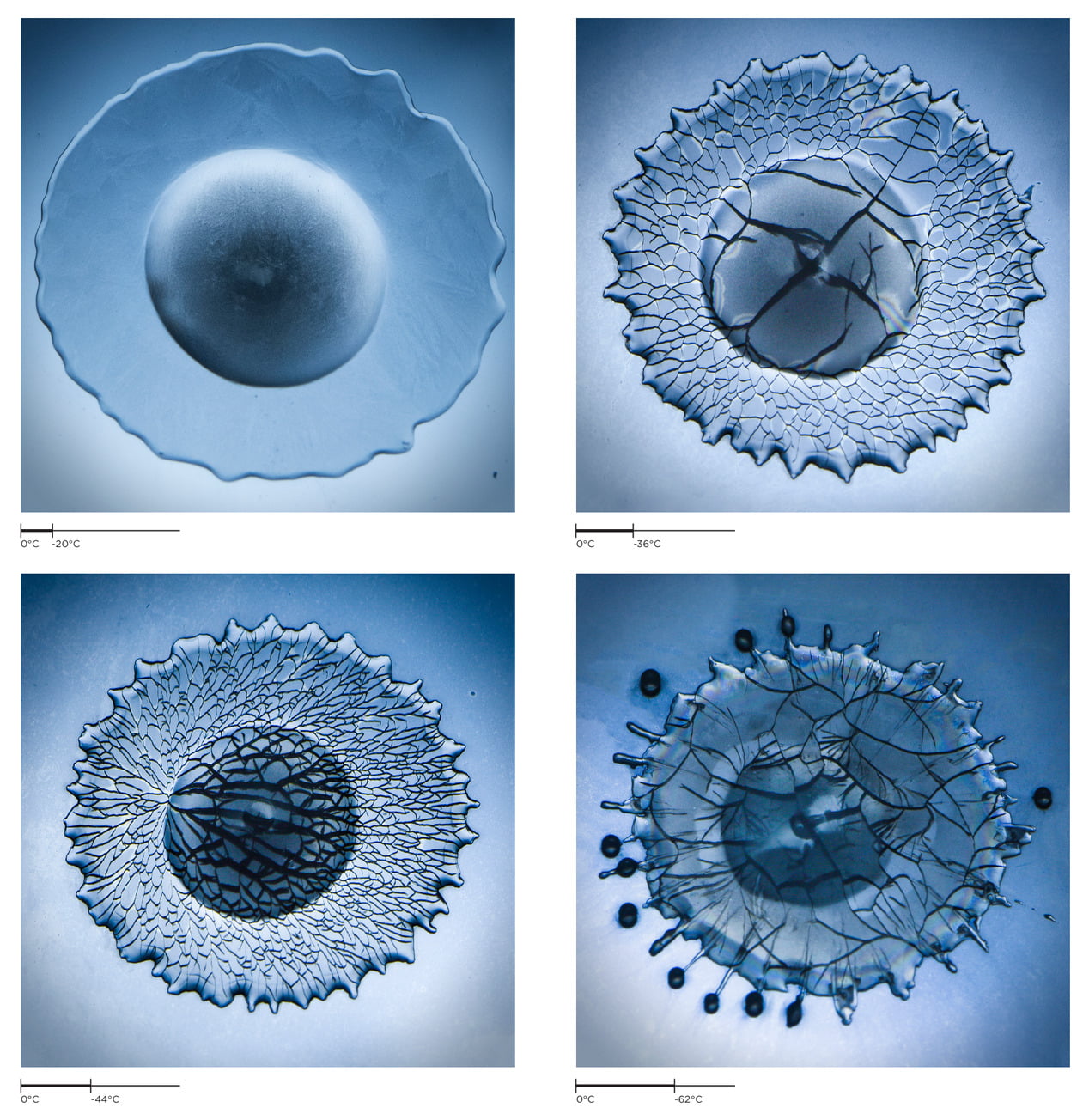
Water is one of those strange materials that expands when it freezes, which raises an interesting question: what happens to a water drop that freezes from the outside in? A freezing water droplet quickly forms an ice shell (top image) that expands inward, squeezing the water inside. As the pressure rises, the droplet develops a spicule – a lance-like projection that helps relieve some of the pressure.
Eventually the spicule stops growing and pressure rises inside the freezing drop. Cracks split the shell, and, as they pull open, the cracks cause a sudden drop in pressure for the water inside (middle image). If the droplet is large enough, the pressure drop is enough for cavitation bubbles to form. You can see them in the middle image just as the cracks appear.
After an extended cycle of cracking and healing, the elastic energy released from a crack can finally overcome surface energy’s ability to hold the drop together and it will explode spectacularly (bottom image). This only happens for drops larger than a millimeter, though. Smaller drops – like those found in clouds – won’t explode thanks to the added effects of surface tension. (Image credit: S. Wildeman et al., source)
ETA: A previous version of this post erroneously said this was freezing from the “inside out” instead of “outside in”.