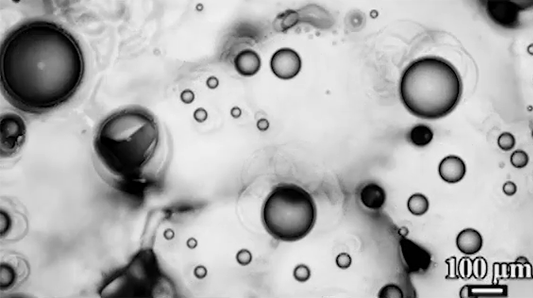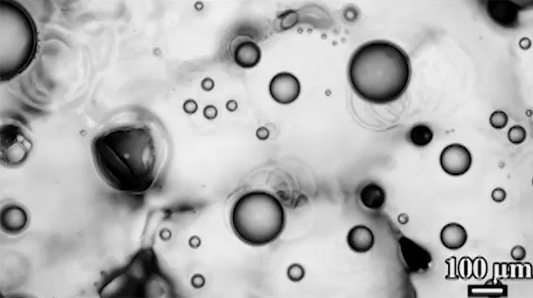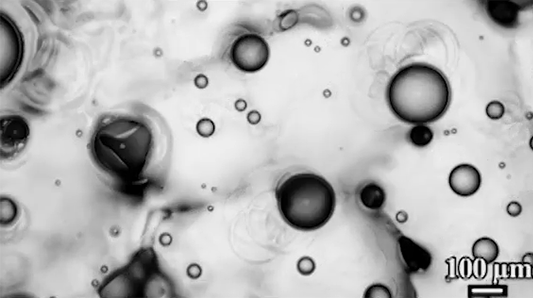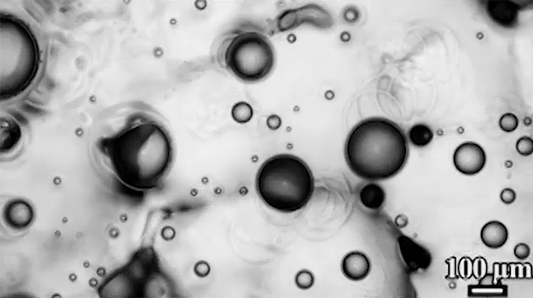
Scientists have observed distinctive differences in the way soap bubbles freeze depending on their environment. If a bubble is surrounded by room temperature air but placed on a cold surface (top), it freezes from the bottom up, with a clear freeze front that slowly creeps upward.
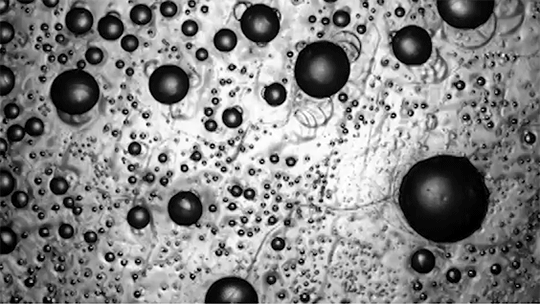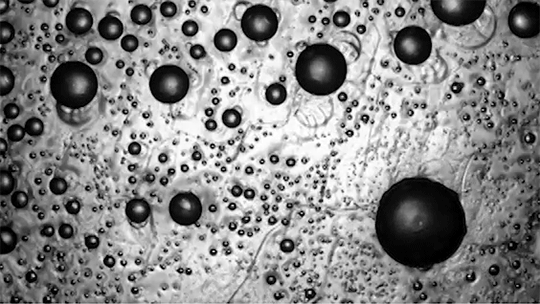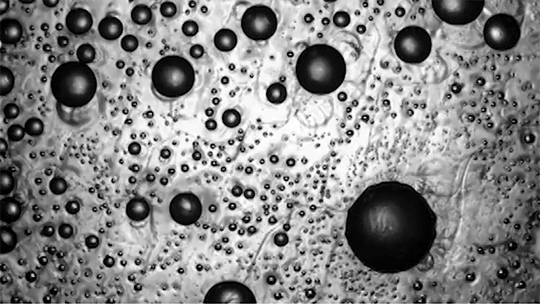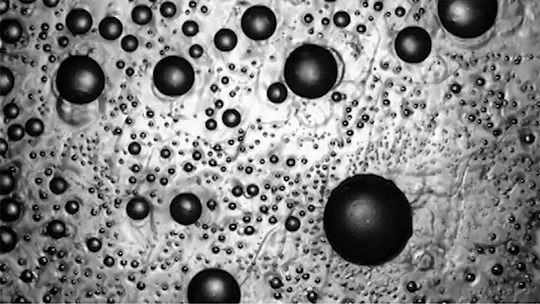
In contrast, bubbles in an isothermal environment – one where it’s equally cold everywhere – freeze with a snow-globe-like effect of ice crystals (bottom). This freezing mode is actually triggered by a Marangoni flow. As the thin bottom layer of the soap bubble begins to freeze, it releases latent heat. That local heating changes the surface tension enough to generate an upward flow. You can see the plumes form right as the bubble touches the surface. Those plumes lift up tiny ice crystals, which continue to grow, ultimately forming the snowy crystals we see take over the surface. (Image and research credit: S. Ahmadi et al.; submitted by Kam-Yung Soh)