Photographer Jan Erik Waider is a master of capturing incredible landscape imagery. In these videos, he uses a drone to film waves in the Baltic Sea gently undulating polygonal slabs of ice on the ocean surface. The interplay of light, color, and motion looks almost surreal, but nature is better than we credit at making imagery too good to look away from. (Video and image credit: J. Waider/NorthLandscapes; via Colossal)
Tag: freezing

A Fungus That Freezes Water
Although water can freeze below 0 degrees Celsius, it requires a little help–in the form of a nucleation site–to do so. Often temperatures must dip well below 0 degrees Celsius for droplets to become ice. But a new study shows that at least one fungus forms proteins that help the process along.
The proteins come from the Mortierellaceae fungal family, by way of a bacterial species some hundreds of thousands of years ago or more. In experiments, adding the fungal protein helped water freeze 10 or more degrees Celsius sooner than it otherwise would.
The authors note that there are many possible applications for this freezing additive; it could help preserve food or cells without requiring lower freezing temperatures that could damage delicate tissues. It could also serve as a cloud seeding chemical in place of toxic silver iodide particles. (Image and research credit: R. Eufemio et al.; via Gizmodo; see also V. Tech)

“Quiet Pulse” and “Another World”
Light shines dimly through the wall of an ice cave in this photograph by Marie-Line Dentler. Shaped by melting, pressure, freezing, and fracture, these structures are dynamic and ethereal. (Image credit: M. Dentler; via Colossal)



Aging Salty Ice
When ice forms in salty water, it starts out mushy and porous. Salt does not freeze neatly into ice’s crystalline structure, so the forming ice has pores and gaps where salty brine gathers. As the ice ages, more brine is pushed out and gradually convects downward, due to its greater density. Over time, this makes the ice layer thinner but more solid, with fewer pores. You can see a timelapse of the process in a laboratory experiment below. (Image credit: sea ice – C. Matias, experiment – F. Wang et al.; research credit: F. Wang et al.)


Milano Cortina 2026: Cortina Sliding Center
This year’s sliding events–bobsleigh, luge, and skeleton–will take place at the brand-new Cortina Sliding Center. Built on the site of a historic sliding track, this new venue came together in only the last couple of years. It features a state-of-the-art refrigeration system that pumps a mixture of water and ethylene glycol beneath the track surface to keep the ice properly chilled. Each section of the track is continuously monitored to optimize the flow rate, temperature, and pressure of the refrigerant to keep the track at maximum performance while minimizing environmental impact.
According to the designers, it’s the first competition track to use a glycol-based refrigeration system, which should be more sustainable than the ammonia-based systems used elsewhere. For a sense of what a run is like, check out this skeleton driver POV run from the facility’s shakedown competition last year. (Image credit: LMSteel; video credit: tuff sledding)

“Moment of Creation”
Bubbles caught in ice resemble the growth of a cellular organism in this photograph of Tatiewa Lake in Japan, taken by Soichiro Moriyama. When water freezes, gases dissolved in it come out of solution, but depending on the speed and direction of freezing, these bubbles do not always escape before ice forms around them, freezing pockets of gas within the ice’s structure. (Image credit: S. Moriyama; via ILPOTY)

“Melting Snowflake”
It’s hard to preserve something as ephemeral as a snowflake, as seen in this microphotograph by Michael Robert Peres. Despite the old adage, it is possible to make identical snowflakes, but it requires mirroring the freezing conditions exactly, including both temperature and humidity. Here, the snowflake’s crystalline structure survives as a ghost in a melting droplet. (Image credit: M. Peres; via Ars Technica)

How to Keep Water From Freezing
When supercooled, water can remain a liquid even below its freezing point. As explained in this Minute Physics video, this happens because of a tug-of-war between effects in the water. Generally speaking, having impurities in the water or smacking the bottle will shift that battle enough for freezing to win out. But it’s possible–theoretically, at least–to create a situation where supercooled water can never freeze. (Video and image credit: Minute Physics)

Frosted
Frost forms hexagonal columns on a wooden rail in this microphotograph by Gregory B. Murray. Like in snowflakes, when water molecules freeze they position themselves to form six-sided crystals. From this perspective, it looks like a miniature version of the Giant’s Causeway. (Image credit: G. Murray; via Ars Technica)

Salt and Sea Ice Aging
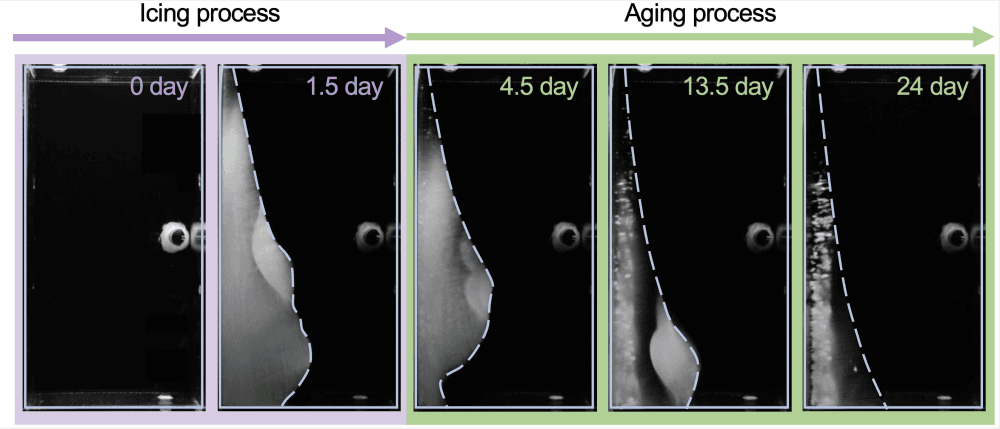
Sea ice’s high reflectivity allows it to bounce solar rays away rather than absorb them, but melting ice exposes open waters, which are better at absorbing heat and thus lead to even more melting. To understand how changing sea ice affects climate, researchers need to tease out the mechanisms that affect sea ice over its lifetime. A new study does just that, showing that sea ice loses salt as it ages, in a process that makes it less porous.
Researchers built a tank that mimicked sea ice by holding one wall at a temperature below freezing and the opposite wall at a constant, above-freezing temperature. Over the first three days, ice formed rapidly on the cold wall. But it did not simply sit there, once formed. Instead, the researchers noticed the ice changing shape while maintaining the same average thickness. The ice got more transparent over time, too, indicating that it was losing its pores.

Looking closer, the team realized that the aging ice was slowly losing its salt. As the water froze, it pushed salt into liquid-filled pores in the ice. One wall of the pore was always colder than the others, causing ice to continue freezing there, while the opposite wall melted. Over time, this meant that every pore slowly migrated toward the warm side of the ice. Once the pore reached the surface, the briny liquid inside was released into the water and the ice left behind had one fewer pores. Repeated over and over, the ice eventually lost all its pores. (Image credit: T. Haaja; research credit and illustration: Y. Du et al.; via APS)