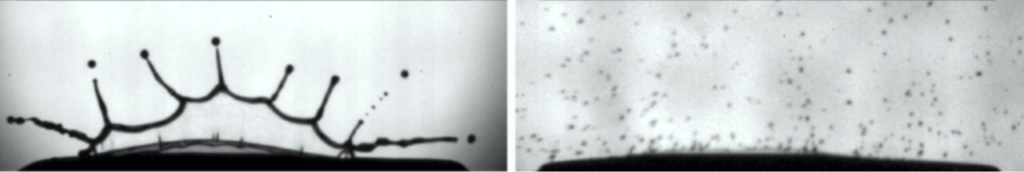
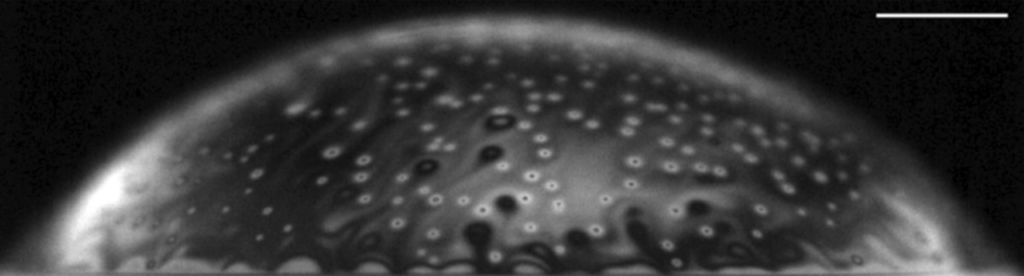
Soft systems like this bubble raft can retain memory of how they reached their current configuration. Because the bubbles are different sizes, they cannot pack into a crystalline structure, and because they’re too close together to move easily, they cannot reconfigure into their most efficient packing. This leaves the system out of equilibrium, which is key to its memory.
By shearing the bubbles between a spinning inner ring (left in image) and a stationary outer one (not shown) several times, researchers found they they could coax the bubbles into a configuration that was unresponsive to further shearing at that amplitude.
Once the bubbles were configured, the scientists could sweep through many shear amplitudes and look for the one with the smallest response. This was always the “remembered” shear amplitude. Effectively, the system can record and read out values similar to the way a computer bit does. Bubbles are no replacement for silicon, though. In this case, scientists are more interested in what memory in these systems can teach us about other, similar mechanical systems and how they respond to forces. (Image and research credit: S. Mukherji et al.; via Physics Today; submitted by Kam-Yung Soh)