Surface tension tries to minimize a bubble‘s surface area, which is why bubbles assume a spherical shape. But when many bubbles clump together, a curved interface is not always the most energy efficient one. In this case, bubbles can take on many shapes and sizes while still minimizing the overall surface energy. Take a close look at this image and see what shapes you discover! (Image credit: M. Adil)
Tag: bubbles

Stabilizing Foams
Bubbles in a pure liquid don’t last long, but with added surfactants or multiple miscible liquids, bubbles can form long-lasting foams. In soapy foams, surfactants provide the surface tension gradients necessary to keep the thin liquid layers between bubbles from popping. But what stabilizes a surfactant-free foam?
New work finds that foams in mixtures of two miscible fluids only form when the surface tension depends nonlinearly on the concentration of the component liquids. When this is true, thinning the wall between bubbles creates changes in surface tension that stabilize the barrier and keep it from popping.
In mixtures without this nonlinearity, foams just won’t form. The new results are valuable for manufacturing, where companies can avoid unintentional foams simply by careful selection of their fluids. (Image credit: G. Trovato; research credit: H. Tran et al.; via APS Physics; see also Ars Technica, submitted by Kam-Yung Soh)

Oil in Water
In the decade since the Deepwater Horizons oil spill, scientists have been working hard to understand the intricacies of how liquid and gaseous hydrocarbons behave underwater. The high pressures, low temperatures, and varying density of the surrounding ocean water all complicate the situation.
Released hydrocarbons form a plume made up of oil drops and gas bubbles of many sizes. Large drops and bubbles rise relatively quickly due to their buoyancy, so they remain confined to a relatively small area around the leak. Smaller drops are slower to rise and can instead get picked up by ocean currents, allowing them to spread. The smallest micro-droplets of oil hardly rise at all; instead they remained trapped in the water column, where currents can move them tens to hundreds of kilometers from their point of release. (Image and research credit: M. Boufadel et al.; via AGU Eos; submitted by Kam-Yung Soh)

Wrinkles on Bubble Collapse
A viscous bubble wrinkles when it collapses, and scientists long assumed this behavior was caused by gravity. But a new experiment shows that the buckling is, instead, driven by surface tension.
To test gravity’s influence on bubble collapse, the researchers popped bubbles in three orientations: the (normal) upright orientation (Images 1 and 2), upside-down (Image 3), and sideways (Image 4). In all cases, the bubble’s thin film wrinkled as it collapsed, indicating that gravity had little influence on the process. Instead the authors concluded that surface-tension-driven collapse causes the dynamic buckling of the film. (Image and research credit: A. Oratis et al.; submitted by Zander B.)

Simulating Better Breaking Waves
In the ocean, breaking waves trap air into bubbles that then cluster into foam, but conventional simulations don’t capture this foaminess. For bubbles to cluster into foam, there has to be a force preventing — or at least delaying — their coalescence. Typically, this is caused by impurities in the water that help lower the surface tension and thereby lengthen the bubbles’ lifespans. When these features get added to simulation models, bubbles begin to cluster and breaking waves become foamy. (Image and video credit: P. Karnakov et al.)

Droplets From Jets
On the ocean, countless crashing waves are creating bubbles. When they burst, those bubbles generate jets and droplets that spray into the sky, carrying sea salt, dust, and biological material into the atmosphere. Researchers know these droplets and their evaporation are important for understanding environmental processes, but figuring out how to capture that importance in models continues to be a challenge.
In a new study, researchers concentrated on a simplified problem: the bursting of a single bubble in pure water. By studying a wide range of conditions, the team found that jets from these bubbles could eject as many as 14 droplets apiece. And though existing models have mostly ignored all but the first droplet, their work showed that all of the droplets should be accounted for in any evaporation models. (Image credit: C. Couto; research credit: A. Berny et al.)

Measuring Contaminants in Drops and Bubbles
Rising bubbles and droplets are common in many chemical and industrial applications. But just a tiny concentration of contaminants on their surface can completely alter their behavior, disrupting coalescence and slowing down chemical reactions.
Historically, it’s been hard to measure the level of contamination in these some drops and bubbles, but a new study outlines a way to measure these small concentrations by perturbing the drops and watching how they deform. By analyzing how the drop shimmies and shakes, they’re able to measure its surface tension and, ultimately, the concentration of contaminants. (Image credit: S. Sørensen; research credit: B. Lalanne et al.; via APS Physics)

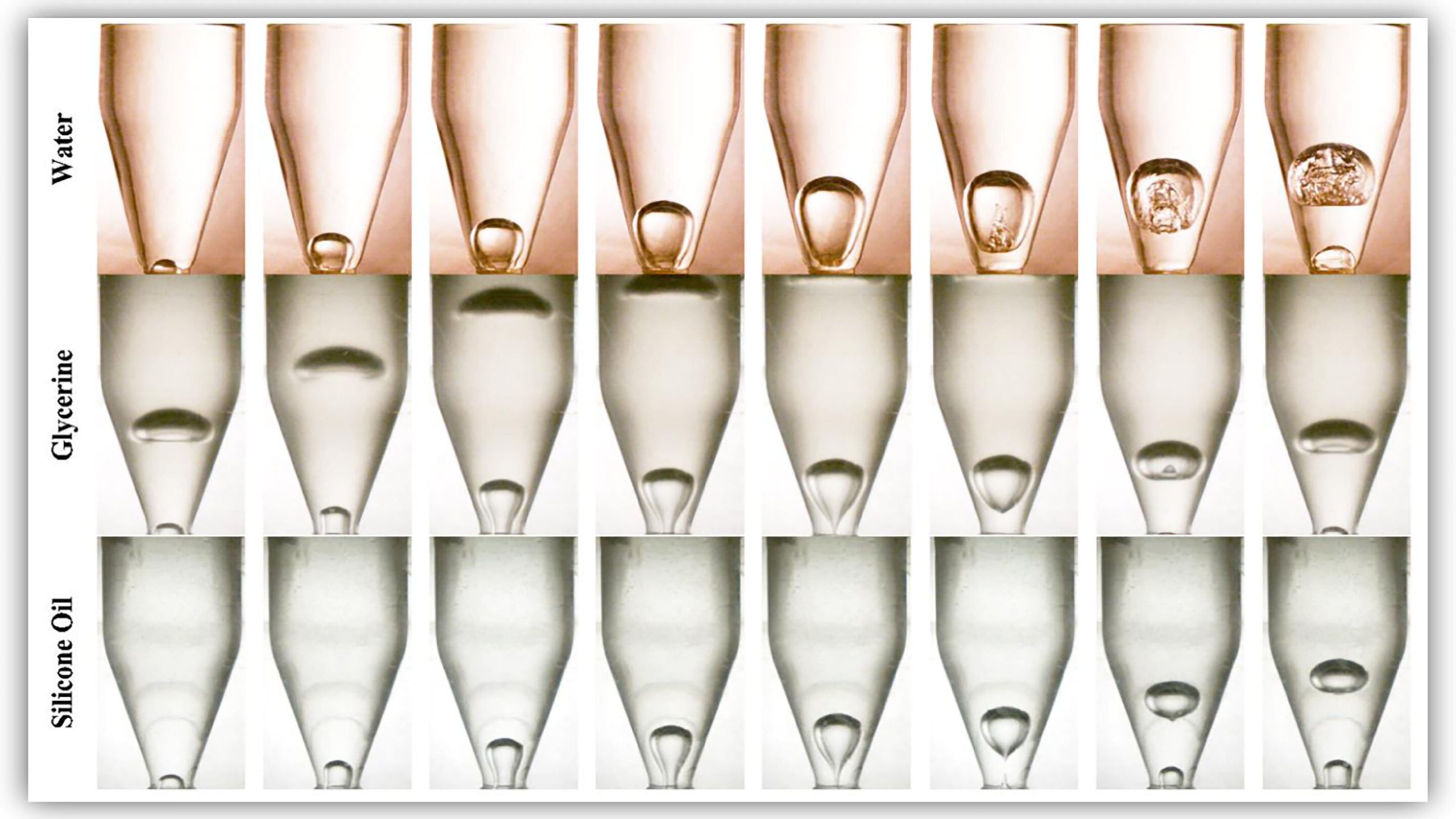
Bubble Dynamics Govern Faster Pouring
We’re all familiar with the problem of pouring a liquid from a narrow-necked bottle. To a certain extent, tilting the bottle further will reduce the time it takes to empty, but if you tilt too far, your smooth pour becomes violent glugging as bubbles forming at the bottle’s mouth block liquid from exiting.
Researchers find that the time it takes to empty a bottle depends both on the qualities of the liquid — its viscosity and surface tension — and on the geometry of the bottle. In particular, they found that the shape of the bottle influences how quickly bubbles grow at the bottle’s mouth when tilted to the critical angle. Their findings suggest that higher tilt angles and faster pours can be achieved by optimizing bottle geometry. (Image and research credit: L. Rohilla and A. Das; via phys.org)

Cavitation Through Acceleration
Cavitation refers to the formation of destructive bubbles of vapor within a liquid. Traditionally, we think of it as occurring when the velocity in a flow becomes high enough for the pressure to drop below the local vapor pressure, causing bubbles to form. This is what we see around turbine blades and ship propellers.
But cavitation also occurs in situations where the overall velocity is relatively low, provided there’s a sudden acceleration. That’s the situation we see above. The impact — either of a mallet off-screen or of the tube striking the floor — causes the liquid inside suddenly accelerate upward. Notice in the second image how the liquid interface moves upward as the first bubbles form.
Each of these cavitation bubbles has such a low pressure that they’re basically a vacuum, and their collapse can cause shock waves that reverberate through the container, causing it to break. Check out that test tube in the last image. Notice that there’s no sign of cracking when the test tube hits the floor; in fact, the researchers demonstrate in their paper that an empty test tube dropped from the same height doesn’t break. Fractures only form after the cavitation bubbles do. (Image and research credit: Z. Pan et al.; submitted by A.J.F.)

Nitro Bubble Cascades

Fans of nitro beers — particularly Guinness’ stout — have probably noticed the fascinating cascade of bubbles that form as the beer settles. It’s a non-intuitive behavior — bubbles rise since they’re lighter than the surrounding fluid. So why do the bubbles appear to sink in these beers?
There are several effects at play here. Firstly, overall the bubbles in the beer are rising; even mixing nitrogen gas into a beer in place of carbon dioxide doesn’t change that. But pint glasses typically flare so that they’re wider at the top than at the bottom. Since the bubbles rise essentially straight up, this causes a bubble-less film to form near the upper walls. And as that heavier fluid sinks, it pulls some of the tiny nitrogen bubbles with it. (You don’t see this effect in typical beers because the bubbles there are larger and thus too buoyant to get pulled down by the falling fluid.)
As for the cascading waves we see in the bubbles, this, too, comes from the shape of the glass. Hydrodynamically speaking, what’s happens as the fluid film slides down the pint glass is similar to what happens when rain runs downhill. Beyond a certain angle, the flow becomes unstable and will form rolls and waves of varying thickness instead of sinking in a thin, uniform layer. As the film goes, so go the bubbles being dragged along, giving everyone at the bar a brief but entertaining fluid dynamical show. (Image credits: pints – M. d’Itri; bubble cascade – T. Watamura et al.; research credit: T. Watamura et al.)