Champagne owes much of its allure to its tiny bubbles. Unlike other wines, champagne undergoes a secondary fermentation in the bottle, during which the yeasts in the wine consume sugars and produce carbon dioxide, which dissolves into the wine. When opened, the carbon dioxide can begin to escape. Bubbles form in the glass around imperfections, either due to intentional etching of the glass or impurities left behind by cleaning. Once formed, trails of bubbles rise to the surface, swelling as more dissolved carbon dioxide is absorbed into each bubble. The bubbles then cluster near the surface of the champagne, occasionally popping and creating a flower-like distortion of the surrounding bubbles. The gases within the bubbles contains higher concentrations of aromatic chemicals than the surrounding wine, and the bursting of each bubble propels tiny droplets of these aromatics upwards, carrying the scent of the champagne to the drinker. For more beautiful champagne photos, I recommend this LuxeryCulture article; for more on the science of champagne, see Chemistry World’s coverage. Happy 2014! (Image credits: G. Liger-Belair et al.)
Tag: bubbles

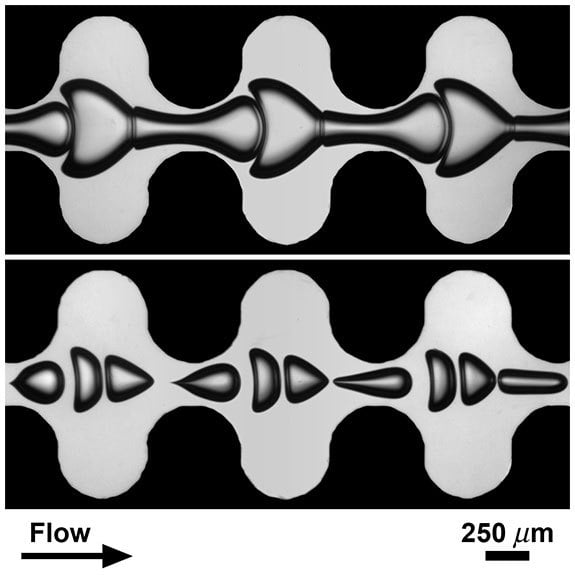
Bubbles Through Constrictions
Surface tension usually constrains bubbles to the smallest area for a given volume – a sphere – but sometimes other forces generate more complicated geometries. The images above show bubbles flowing through microfluidic channels filled with a highly viscous carrier fluid. The bubble size and packing affects the shapes they assume, but so does the geometry of the channel. The narrow constrictions accelerate the flow, elongating the bubbles, whereas the wider channel regions slow the carrier fluid and squish the bubbles together. (Image credit: M. Sauzade and T. Cubaud (Stony Brook University))

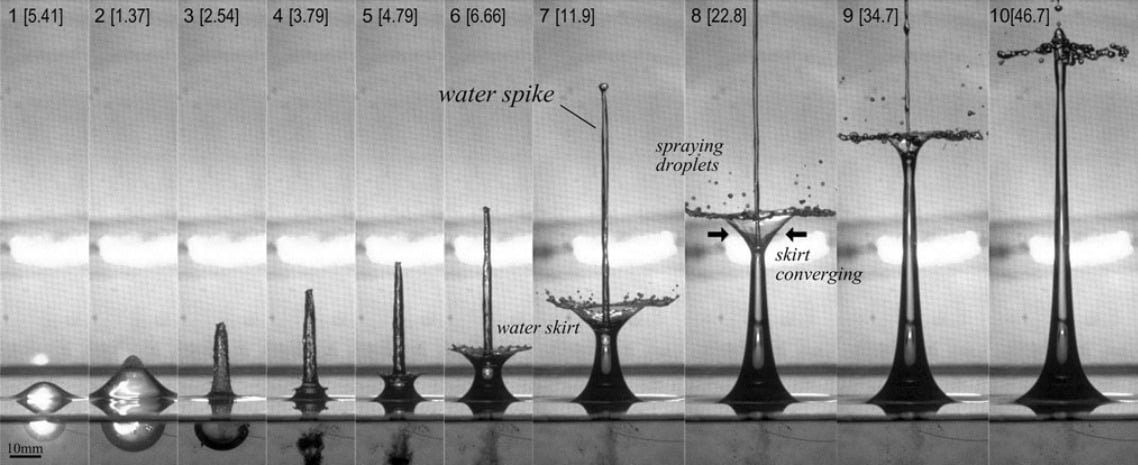
Fluid Sculptures From Bursting Bubbles
A bubble initiated near a free surface–like the air-water interface here–can generate some spectacular dynamics. Beginning at the far left, the expanding subsurface bubble causes a dome at the surface that sharpens into a spike. By Frame 3, the bubble is collapsing but overshoots and rebounds, which introduces the tiny instability in Frame 4 that grows in subsequent time steps to form the water skirt that surrounds the spike. Although generated entirely differently, the end result is reminiscent of the water sculptures made by artists like Marcus Reugels, Corrie White, Jack Long, and others. (Image credit: A. M. Zhang et al.)

Overflowing Foam
Hitting a glass bottle full of a non-carbonated drink can shatter the bottle due to cavitation, but doing the same with a carbonated beverage can make the bottle overflow with foam. The video above breaks down the physics of this bar prank. It all begins with nucleation and the tiny bubbles of carbon dioxide that form in the liquid. Striking the top of the bottle generates a compression wave that travels through the liquid, shrinking bubbles as it passes. When it hits the bottom of the bottle, it gets reflected as an expansion wave that expands the bubbles. This reflection happens several times between the free surface of the liquid and the bottom of the bottle. The rapid collapse-and-expansion of the bubbles makes them implode into a cloud of tinier bubbles that expands until the local supply of carbon dioxide is used up. At this point, the buoyancy of the bubbles carries them upward in plumes, creating more bubbles with the dissolved carbon dioxide nearby. And, all of a sudden, you’ve got foam everywhere. Like all of this week’s videos, this video is an entry in the 2013 Gallery of Fluid Motion. (Video credit: J. Rodriguez-Rodriguez et al.)

Bubbles With Tails
In water and other Newtonian fluids, a rising bubble is typically spherical, but for non-Newtonian fluids things are a different story. In non-Newtonian fluids the viscosity–the fluid’s resistance to deformation–is dependent on the shear rate and history–how and how much deformation is being applied. For rising bubbles, this can mean a teardrop shape or even a long tail that breaks up into fishbone-like ligaments. The patterns shown here vary with the bubble’s volume, which affects the velocity at which it rises (due to buoyancy) and thus the shear force the bubble and surrounding non-Newtonian fluid experience. (Video credit: E. Soto, R. Zenit, and O. Manero)

Drop-Tower Droplets
A microgravity environment can cause some nonintuitive behaviors in fluids. Many of the effects that dominate fluid dynamics in space are masked by gravity’s effects here on Earth. As a result, it can be very difficult to predict how seemingly straightforward technologies like heat exchangers, refrigeration units, and fuel tanks will behave. The photos above show two bubble jets–created by injecting a liquid-gas mixture into a liquid–colliding in microgravity. This particular experiment was conducted in a drop tower rather than on-orbit, which produced some side effects like the large bubbles seen in the images. These were created by the coalescence of smaller bubbles that congregated near the top of the tank shortly before the experiment attained free-fall. (Photo credit: F. Sunol and R. Gonzalez-Cinca)

Dancing Jets
Vibrating a gas-liquid interface produces some exciting instability behaviors. The photo above shows air and silicone oil vibrated vertically within a prism. For the right frequencies and amplitudes, the vibrations produce liquid jets that shoot up and eject droplets as well as gas cavities and bubble transport below the interface. To see a similar experiment in action, check out this post. (Photo credit: T. J. O’Hern et al./Sandia National Laboratories)

The Colors of Soap
The brilliant and beautiful colors of a bubble are directly related the the thickness of the soap film surrounding it. When light shines on the soap film, some rays are reflected from the upper surface of the film, while others are refracted through the film and reflect off its lower surface. These reflected rays have different phase shifts and their interference is what causes the colors we observe. The color patterns themselves reveal the interior flow of the soap film, in which gravity tries to thin the film and surface tension tries to distribute the film evenly. (Photo credit: R. Kelly, A. Fish, D. Schwichtenberg, N. Travers, G. Seese)

Fluids Round-up – 9 June 2013
It’s time for some more fluidsy fun around the Internet! Here are some fun links I’ve come across since our last round-up.
- NPR reviews how dolphins and others play with vortex rings.
- Lawrence Berkeley National Laboratory/UC Berkeley offer some insight into simulating bubbles popping. (Hint: it requires supercomputers.)
- FlowViz shares some awesome accidental Rayleigh-Taylor instabilities you can replicate at home.
- PhysicsBuzz brings us a podcast on tornado physics.
- Reader Cedric Vella sent in his fluids-featuring trailer.
- io9 pointed out some great cymatics footage that shows off how granular materials and vibration creates beautiful patterns.
- And finally: what happens when you drop hot charcoal into liquid oxygen? The Periodic Table of Videos shows us, in high speed! (via Flow Visualization)
(Photo credit: L. L. A. Adams et al., multi-fluid double emulsions)

Turning Sound into Light
Sonoluminescence – the creation of light from sound – was discovered in the 1930s, and, due to the difficulty of obtaining direct measurements, the exact mechanism remains highly debated even today. The phenomenon typically takes place within a tiny cavitation bubble inside a liquid. When bombarded with ultrasonic sound, such a bubble will repeatedly expand and collapse. Once a bubble is established, the cycle can be kicked off by increasing the driving acoustic pressure. This will collapse the bubble, drastically increasing its pressure and temperature (up to thousands of degrees Kelvin) and causing the bubble to emit a pulse of light before the pressure imbalance causes it to expand again. Several theories exist as to how the light is generated, the leading one being that the high temperatures in the bubble ionize the noble gases within and that those free electrons emit light via thermal bremstrahlung radiation. Sonoluminescence happens outside the lab, too. Both the previously discussed pistol shrimp and the mantis shrimp generate such light-emitting bubbles when hunting. (Video credit: The Point Studios; suggested by Bobby E.)