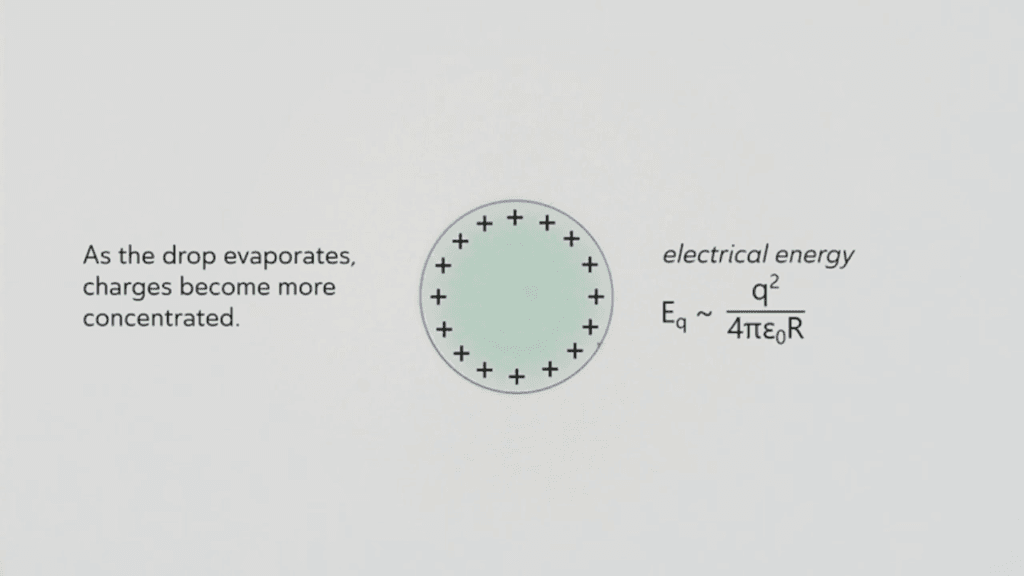
Dropping water from a plastic pipette onto a pool of oil electrically charges the drop. Then, as it evaporates, it shrinks and concentrates the charges closer and closer. Eventually, the strength of the electrical charge overcomes surface tension, making the drop form a cone-shaped edge that jets out tiny, highly-charged microdrops. Afterward, the drop returns to its spherical shape… until shrinkage builds up the charge density again. This microjetting behavior can carry on for hours! (Video and image credit: M. Lin et al.; research preprint: M. Lin et al.)
Search results for: “surface tension”

The Mystery of the Binary Droplet
What goes on inside an evaporating droplet made up of more than one fluid? This is a perennially fascinating question with lots of permutations. In this one, researchers observed water-poor spots forming around the edges of an evaporating drop, almost as if the two chemicals within the drop are physically separating from one another (scientifically speaking, “undergoing phase separation“). To find out if this was really the case, they put particles into the drop and observed their behavior as the drop evaporated. What they found is that this is a flow behavior, not a phase one. The high concentration of hexanediol near the edge of the drop changes the value of surface tension between the center and edge of the drop. And that change is non-monotonic, meaning that there’s a minimum in the surface tension partway along the drop’s radius. That surface tension minimum is what creates the separated regions of flow. (Video and image credit: P. Dekker et al.; research pre-print: C. Diddens et al.)

Within a Drop
In this macro video, various chemical reactions swirl inside a single dangling droplet. Despite its tiny size, quite a lot can go on in a drop like this. Both the injection of chemicals and the chemical reactions themselves can cause the flows we see here. Surface tension variations and capillary waves on the exterior of the drop can play a role, too. Just because a flow is tiny doesn’t mean it’s simple. (Video and image credit: B. Pleyer; via Nikon Small World in Motion)

Chemical reactions swirl within a single, hanging droplet. 
Active Cheerios Self-Propel
The interface where air and water meet is a special world of surface-tension-mediated interactions. Cereal lovers are well-aware of the Cheerios effect, where lightweight O’s tend to attract one another, courtesy of their matching menisci. And those who have played with soap boats know that a gradient in surface tension causes flow. Today’s pre-print study combines these two effects to create self-propelling particle assemblies.
The team 3D-printed particles that are a couple centimeters across and resemble a cone stuck atop a hockey puck. The lower disk area is hollow, trapping air to make the particle buoyant. The cone serves as a fuel tank, which the researchers filled with ethanol (and, in some cases, some fluorescent dye to visualize the flow). Like soap, ethanol’s lower surface tension disrupts the water’s interface and triggers a flow that pulls the particle toward areas with higher surface tension. But, unlike soap, ethanol evaporates, effectively restoring the interface’s higher surface tension over time.
With multiple self-propelling particles on the interface, the researchers observed a rich series of interactions. Without their fuel, the Cheerios effect attracted particles to each other. But with ethanol slowly leaking out their sides, the particles repelled each other. As the ethanol ran out and evaporated, the particles would again attract. By tweaking the number and position of fuel outlets on a particle, the researchers found they could tune the particles’ attractions and motility. In addition to helping robots move and organize, their findings also make for a fun educational project. There’s a lot of room for students to play with different 3D-printed designs and fuel concentrations to make their own self-propelled particles. (Research and image credit: J. Wilt et al.; via Ars Technica)

A Mini Jupiter
Astronaut Don Pettit posted this image of a Jupiter-like water globe he created on the International Space Station. In microgravity, surface tension reigns as the water’s supreme force, pulling the mixture of water and food coloring into a perfect sphere. It will be interesting to see a video version of this experiment, so that we can tell what tools Pettit used to swirl the droplet into the eddies we see. Is the full droplet rotating (as a planet would), or are we just seeing the remains of a wire passed through the drop? We’ll have to stay tuned to Pettit’s experiments to find out. (Image credit: NASA/D. Pettit; via space.com; submitted by J. Shoer)

Predicting Droplet Sizes
Squeeze a bottle of cleaning spray, and the nozzle transforms a liquid jet into a spray of droplets. These droplets come in many sizes, and predicting them is difficult because the droplets’ size distribution depends on the details of how their parent liquid broke up. Shown above is a simplified experimental version of this, beginning with a jet of air striking a spherical water droplet on the far left. In less than 3 milliseconds, the droplet has flattened into a pancake shape. In another 4 milliseconds, the pancake has ballooned into a shape called a bag, made up of a thin, curved water sheet surrounded by a thicker rim. A mere 10 milliseconds after the jet and drop first meet, the liquid is now a spray of smaller droplets.
Researchers have found that the sizes of these final droplets depend on the balance between the airflow and the drop’s surface tension; these two factors determine how the drop breaks up, whether that’s rim first, bag first, or due to a collision between the bag and rim. (Image credit: I. Jackiw et al.; via APS Physics)

Marangoni Blossoms
When surface tension varies along an interface, fluids move from regions of low surface tension to higher surface tension, a behavior known as the Marangoni effect. Here, a drop of (dyed) water is placed on glycerol. The two fluids are miscible, but water has much a lower viscosity and density yet a higher surface tension. The drop’s interface quickly becomes unstable; viscous fingers form along the edge as the less viscous water pushes into the more viscous glycerol. Eventually, the surface-tension-driven Marangoni flow breaks those fingers off into lip-like daughter drops. The researchers also show how the interplay between viscosity and surface tension affects the size of fingers that form by varying the water/glycerol concentration. (Image and video credit: A. Hooshanginejad et al.)

Bubblegum Sculptures
Like soap bubbles, bubbles blown in gum are ephemeral, lasting only seconds. Their break-up mechanism is quite different, though. Where surface tension rips a bubble apart once it is pierced, bubblegum instead deflates and wrinkles around a hole that does not grow, thanks to the elasticity of the gum. This photographic series by Suzanne Saroff features a rainbow of gum sculptures, all frozen in the moments of their disintegration. (Image credit: S. Saroff; via Colossal)

“Color Show”
Brightly colored paints and inks mix and flow in artist Roman De Giuli’s “Color Show.” De Giuli typically creates this fluid art in thin layers atop paper. He’s a master of the form, manipulating surface tension gradients to create streaming flows, dendritic patterns, and feathery wisps. If this kind of art is your jam, he offers an app full of live wallpapers* for Android phones. See more of his work on his website and on Instagram. (Video and image credit: R. De Giuli)
*Not sponsored, I just like his art!
Lasers and Soap Films
Soap films are a great system for visualizing fluid flows. Researchers use them to look at flags, fish schooling and drafting, and even wind turbines. In this work, researchers explore the soap film’s reaction to lasers. When surfactant concentrations in the soap film are low, laser pulses create shock waves (above) in the film that resemble those seen in aerodynamics. The laser raises the temperature at its point of impact, lowering the local surface tension. That temperature difference triggers a Marangoni flow that draws the heated fluid outward. The low surfactant concentration gives the soap film relatively high elasticity, and that allows the shock waves to form.
In contrast, a soap film with a high concentration of surfactants has relatively little elasticity. In these films (below), the laser creates a mark that stays visible on the flowing soap film. This “engraving” technique could be used to visualize flow in the soap film without using tracer particles. (Image and research credit: Y. Zhao and H. Xu)

When surfactant concentrations are high, a laser pulse “engraves” spots onto a flowing soap film. Shown in terms of interference (left) and Schlieren (right) imaging.