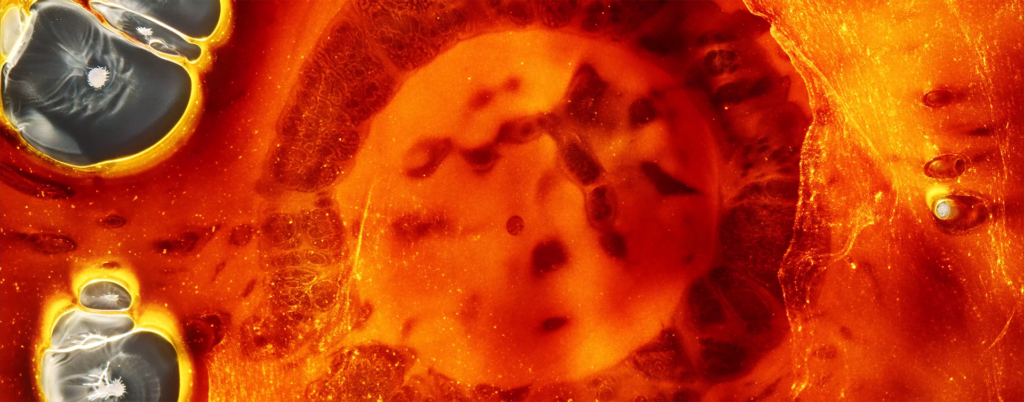
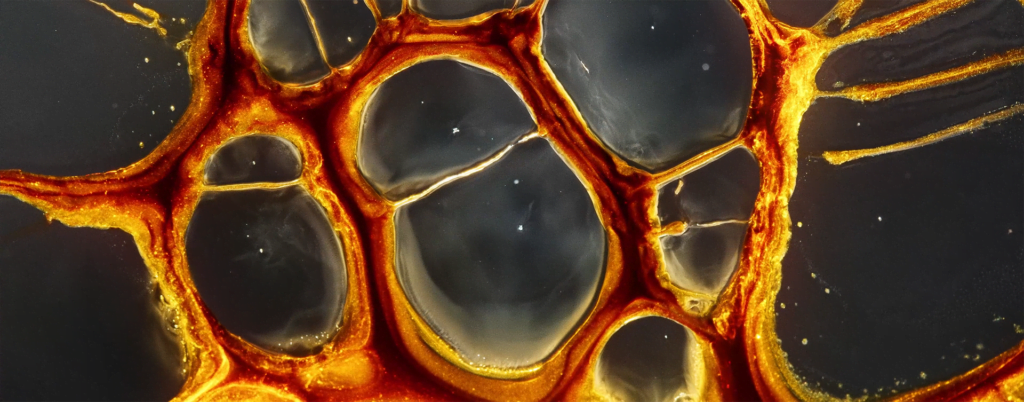
When a drop of ethanol lands on a pool of water, surface tension forces draw it into a fast-spreading film. Evenly-spaced plumes form at the edges of the film, then the film stops spreading and instead retracts. All of this takes place in about 0.6 seconds. But, as the image above shows, there’s more that goes on beneath the surface. A vortex ring forms and spreads under the film, driven by the shear layer under the edge of the plumes. Here, the vortex ring is visible in the swirling particles near the water surface. (Image and research credit: A. Pant and B. Puthenveettil)
Search results for: “surface tension”

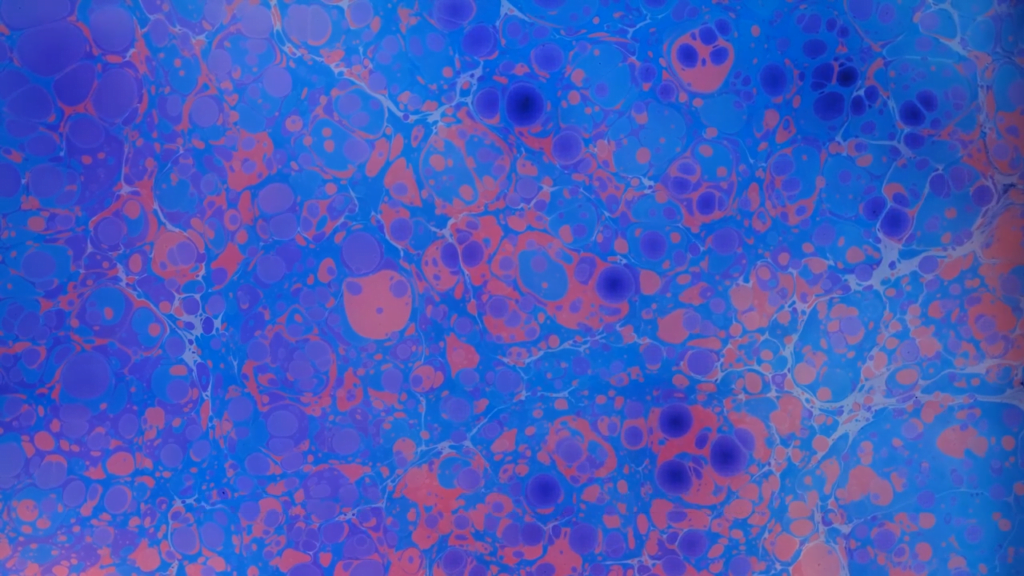
“Lucid”
Artist Roman Hill made this official music video to go with Thomas Vanz’s “Lucid.” The imagery, formed from ink and other fluids, warps our sense of scale. Though the camera focuses on an extremely small area, to our eyes the results shift from nebulas to oceans and back again. There are likely a whole host of phenomena going on here, but without knowing more about Hill’s ingredients, I can only speculate that there are Marangoni flows driven by variations in surface tension and maybe some density instabilities going on between fluid layers. I’m also fairly confident that Hill has played with time reversal in the video editing. Regardless of the secrets in its making, the film is captivating and gorgeous. (Image and video credit: R. Hill)
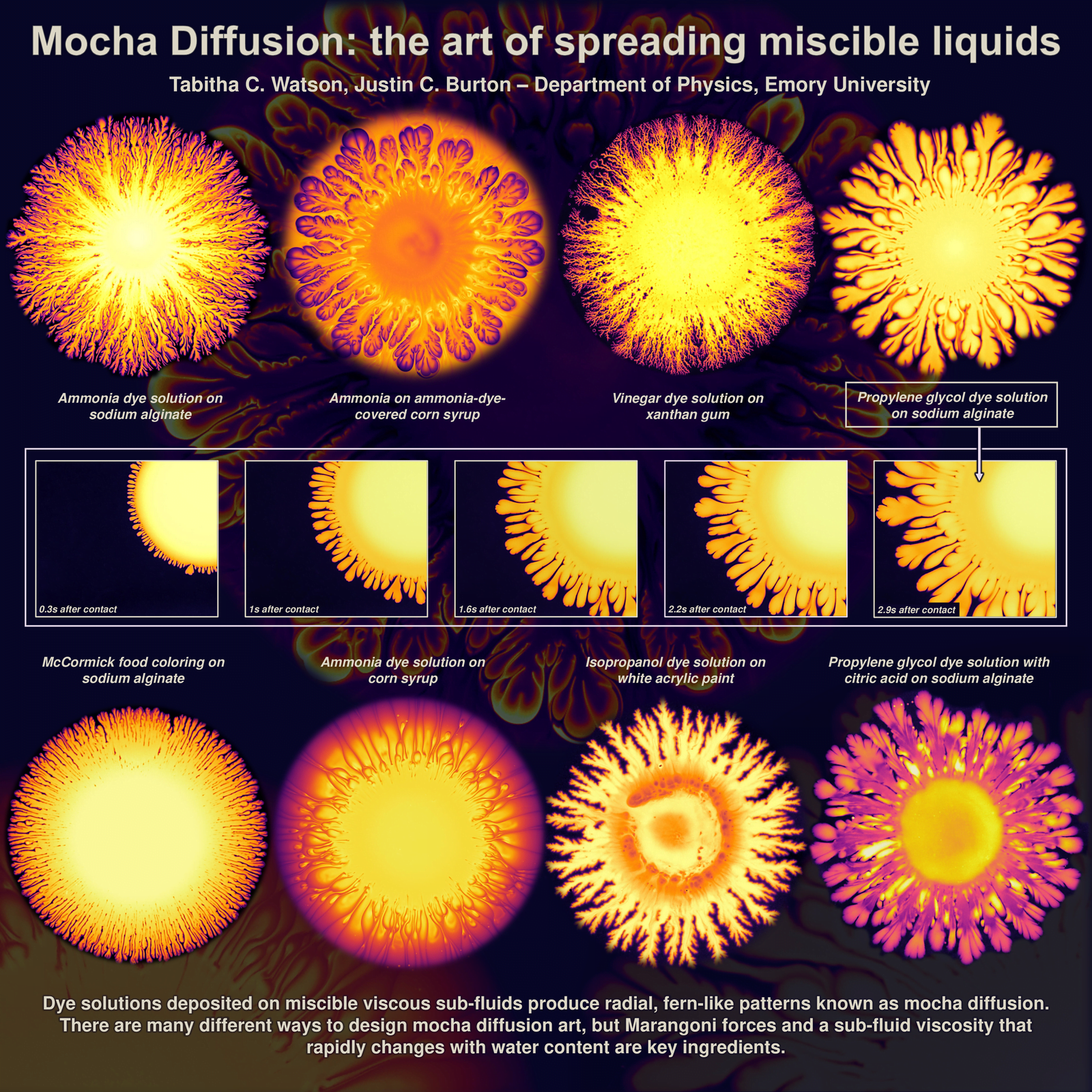
Mocha Diffusion
These firework-like patterns spread when dyes are added atop a viscous but miscible lower fluid layer. Here, researchers use lower layers like corn syrup and xanthan gum; then they spread dye mixtures including ammonia and vinegar atop those layers. Because the upper and lower layers of fluid are miscible and can diffuse into one another, they together form elaborate patterns. The mixing of the two layers creates gradients in surface tension that can drive the flow and create these mocha diffusion patterns. (Image credit: T. Watson and J. Burton)
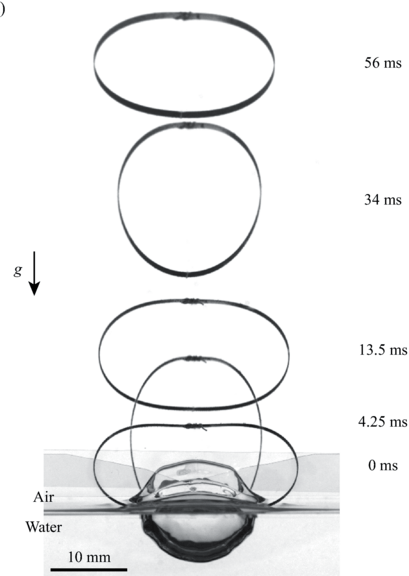
Water Jumping Hoops
Small creatures like springtails and spiders can jump off the air-water interface using surface tension. But larger creatures can water-jump, too, using drag. Here, researchers study drag-based water jumping with a simple elastic hoop. Initially, two sides of the hoop are pulled closer by a string, deforming the hoop. Then, with the hoop sitting upright on the air-water interface, a laser burns the string, releasing the energy stored in the hoop. The hoop’s bottom pushes into the water, generating drag. That resistance provides a reaction force strong enough to launch the hoop.
Compared to the hoop’s jumps off land, it’s slower to take-off from water, and it’s less efficient at jumping. Lighter hoops, however, jump better off water than heavier ones — a wrinkle that isn’t seen in ground jumpers. That suggests that weight reduction is more important for aquatic jumpers than for their terrestrial counterparts. (Image and research credit: H. Jeong et al.)
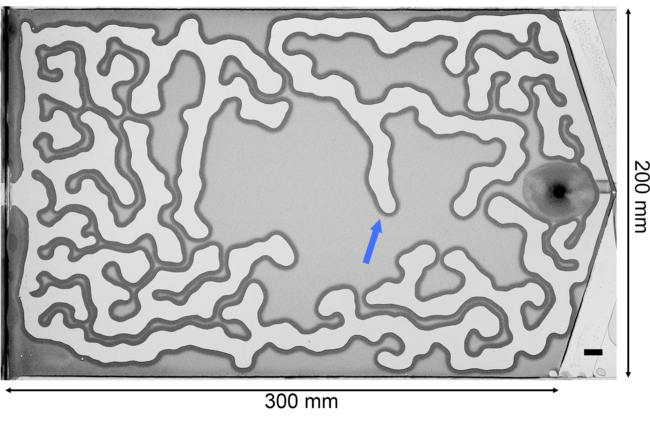
Frictional Fingers
Air pushes into a thin gap filled with water and granular particles in the labyrinth-like image above. The encroaching air pushes grains like a bulldozer’s blade, building up a compacted wall. The invasion continues until the pressure of the air is countered by the combined capillary and frictional forces of the wet grains. Researchers built an analytical model that explains how these frictional fingers form and grow. Unlike Saffman-Taylor fingering patterns, which depend on long-range viscous forces, these patterns depend entirely on short-range forces from surface tension and friction. (Image and research credit: E. Flekkøy et al.)

The Hydrodynamics of Marbling
In marbling, an artist floats paints on a viscosified water bath, using various thin tools to manipulate the final image. Many cultures have developed a version of this art, but for many it will be most recognizable as a technique used to decorate book interiors. In this video, researchers consider the physics behind this beautiful practice. Surface tension helps keep the paint on the surface, even though it’s denser than the water it’s on. Variations in surface tension shape and reshape the surface as new colors are added. And then low-Reynolds-number effects help artists mix the paints without inertia or diffusion disturbing the pattern. See more examples here, here, and here. (Video credit: Y. Sun et al.)

Controlling Finger Formation
When gas is injected into thin, liquid-filled gaps, the liquid-gas interface can destabilize, forming distinctive finger-like shapes. In laboratories, this mechanism is typically investigated in the gap between two transparent plates, a setup known as a Hele-Shaw cell. In the past, researchers looking to control the instability have explored how surface tension, viscosity, and the elasticity of the gap itself affect the flows. But a new set of studies look at the compressibility of the gas being injected.
The team found that viscous fingers formed later the higher the gas’s compressibility. That provides a potential control knob for people trying to exploit the mechanism, especially geologists. For geologists trying to extract oil, viscous fingering is detrimental, but, on the flip side, viscous fingers are desirable when injecting carbon dioxide for sequestration. With these results, users can tweak their injection characteristics to match their goals. (Image credit: C. Cuttle et al.; research credit: C. Cuttle et al. and L. Morrow et al.; via APS Physics)

Dancing to Chopin
Droplets of paint whirl to Chopin’s “Nocturne Op. 9 No. 2” in this short film from artist Thomas Blanchard. The glitter particles in the paints act as seed particles that highlight the flow within and around each drop. It’s a beautiful dance of surface tension, advection, and buoyancy. (Image and video credits: T. Blanchard; via Colossal)

Viscoelasticity and Bubbles
Bursting bubbles enhance our drinks, seed our clouds, and affect our health. Because these bubbles are so small, they’re easily affected by changes at the interface, like surfactants, Marangoni effects, or, as a recent study shows, viscoelasticity.

A bubble released in pure water pops at the surface, creating a rebounding jet and a daughter droplet. In clean water, a bubble’s burst generates a rebounding jet that shoots off one or more daughter droplets, as seen in the animation above. But when researchers added proteins that modify only the water’s surface, they found something very different. As seen below, the bursting bubble no longer generated a jet, and, instead of forming droplets, it made a single, tiny daughter bubble. The difference, they found, comes from the added viscoelasticity of the surface. The long protein molecules resist getting stretched, which damps out the tiny waves that surface tension usually produces on the collapsing bubble cavity. (Image and research credit: B. Ji et al.; submission by Jie F.)

When the surface of water is viscoelastic, a bursting bubble creates no jet and a daughter bubble instead of a drop. 
Why Sea Foams
Seawater froths and foams in ways that freshwater rarely does. A new study pinpoints the ocean’s electrolytes as the reason bubbles resist merging there. By studying the final moments before bubbles coalesce in both pure and salt water, researchers found that dissolved salts slow down the drainage of the thin film of liquid between two bubbles. Once the film reaches a 30-50 nanometer thickness, its electrolyte concentration causes a difference in surface tension that slows the outward flow of liquid in the film. That keeps the film in place longer and makes bubbles form foams instead of merging or popping. (Image credit: P. Kuzovkova; research credit: B. Liu et al.; via APS Physics)