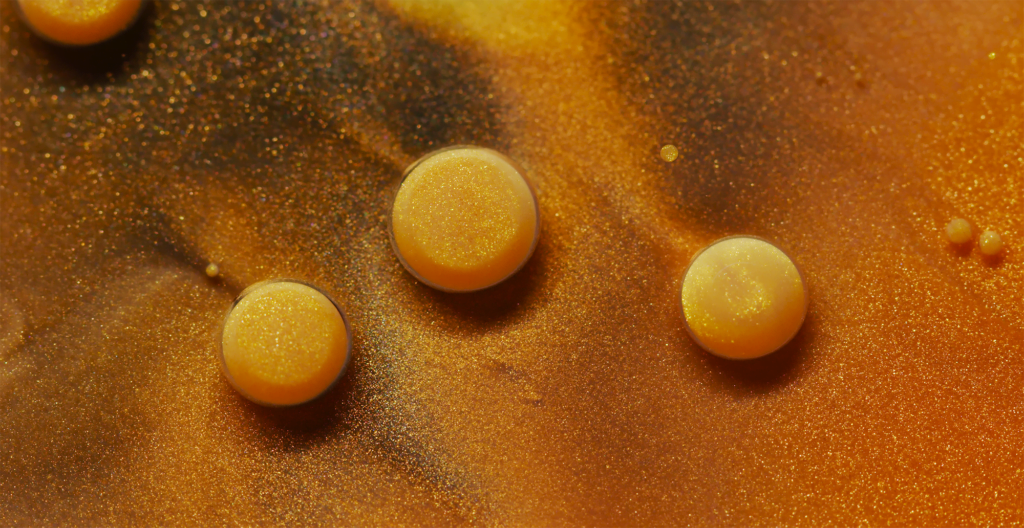
“Emerald and Stone” is filmmaker Thomas Blanchard’s tribute to the music of Brian Eno. The short film is made, as Blanchard puts it, with “inks and painting,” but I suspect there’s some oil in there, too, to coat the droplets we see. Much of the movement is likely driven by surface tension variations in the background fluid. I love the effect this has on the droplets. If you watch closely, some of them appear to rotate like a miniature planet; others have counter-rotating sections within the drop. The difference, I suspect, is one of scale: I think the smaller drops rotate altogether while larger ones develop more complex internal flows. (Video and image credit: T. Blanchard)
Search results for: “surface tension”

Gravity Changes Droplet Shapes
With small droplets, gravity usually has little effect compared to surface tension. An evaporating water droplet holds its spherical shape as it evaporates. But the story is different when you add proteins to the droplet, as seen in this recent study.

The protein-filled sessile drop starts out largely spherical, but as the drop evaporates, the concentration of proteins reaches a critical point and an elastic skin forms over the drop. From this point onward, the drop flattens. As a protein-doped droplet sitting on a surface evaporates, it starts out spherical, like its protein-free cousin. But, as the water evaporates, it leaves proteins behind, gradually increasing their concentration. Eventually, they form an elastic skin covering the drop. As water continues to evaporate, the droplet flattens.

For a hanging droplet, the shape again starts out spherical. But as the drop’s water evaporates and the proteins concentrate, it also forms an elastic skin. As the drop evaporates further, the skin wrinkles. In contrast, a hanging droplet with proteins takes on a wrinkled appearance once its elastic skin forms. The key difference, according to the model constructed by the authors, is the direction that gravity points. Despite these droplets’ small size, gravity makes a difference! (Image, video, and research credit: D. Riccobelli et al.; via APS Physics)

“Discovery”
Colors stream and mix in Rus Khasanov’s short film “Discovery.” Droplet-like liquid lenses float in the mixture until ethanol or other ingredients cause them to spontaneously rupture, sending their interior flowing outward until the lens reaches a new equilibrium. Gradients in surface tension guide Marangoni flows across the screen. There’s never-ending beauty in the world of macro fluids. (Video and image credit: R. Khasanov)

Getting Water Out of Your Ear
Swimming often results in water getting stuck in our ear canals. The narrow space, combined with the waxy surface, is excellent at trapping small amounts of water. If left in place, that excess fluid distorts hearing, can cause pain, and may eventually lead to an ear infection. So most people’s common response is to tilt their head sideways and shake it or jump to knock the water out. This recent study looks at just how much acceleration is needed to dislodge that water.

An acceleration of 7.8g isn’t enough to remove the water from this artificial ear canal. The team built an artificial ear based on the shape of a human’s ear canal and observed how much acceleration was needed to knock the water out. The answer? Quite a bit. As seen above, nearly 8g of acceleration was enough to distort the interface of the water in the ear canal, but it didn’t move the water out.
At higher accelerations — above 20 times the acceleration due to gravity – the air-water interface distorts enough to get the water to flow. But accelerations that large are enough to potentially damage brain tissues.

At over 24g, the acceleration is enough to dislodge the water from this artificial ear canal. But accelerations this high can cause brain damage. The problem is worse for children and babies, whose tiny ear canals necessitate even larger accelerations. For them, shaking hard enough to remove water could cause real damage. Instead, a couple drops of vinegar or alcohol in the ear will lower the surface tension and make the fluid easier to remove. (Image credit: top – J. Flavia, others – S. Kim et al.; research credit: S. Kim et al.; submitted by Sunny J.)

Liquid Lens Rupture
A blob of sunflower oil floating on soapy water forms a disk known as a liquid lens. But add some dyed ethanol and things take a turn. The lens rapidly expands and distorts as the ethanol and soapy water meet. These surface flows are driven by the imbalance of surface tension between the different liquids. The liquid lens deforms and abruptly ruptures, releasing dye and ethanol before rebounding into a stable lens again. Adding more ethanol to the lens will repeat the cycle. (Image credit: C. Kalelkar and P. Dey; research credit: D. Maity et al.)

Can Water Solve a Maze?
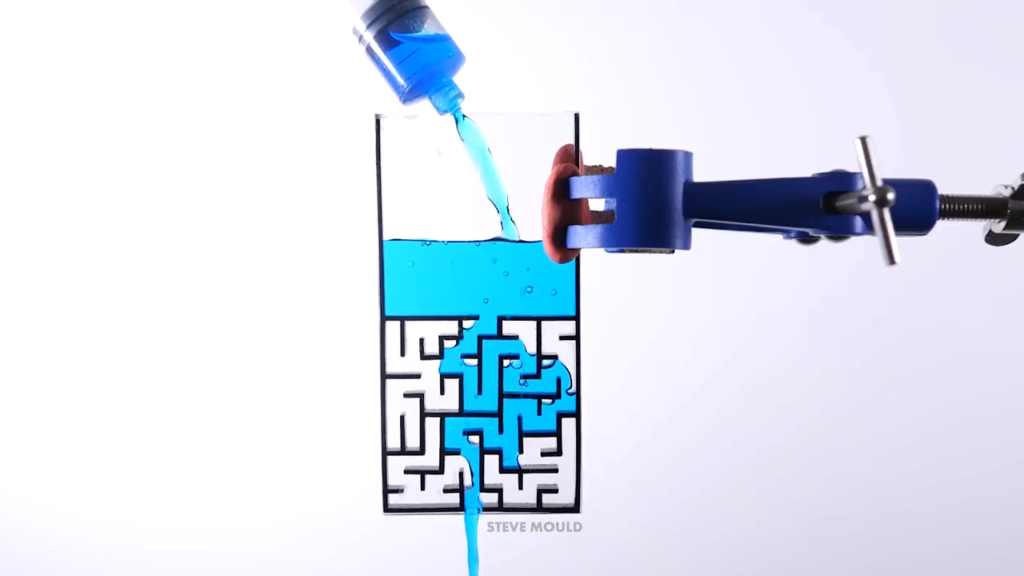
Inspired by a simulation, Steve Mould asks a great question in this video: can water solve a maze? Yes — with some caveats. Steve makes two different maze patterns — a simple and a complex path — in two different sizes. With the small, simple-path version, the water immediately follows the correct path without taking any wrong turns. What keeps it on the right path seems to be a combination of air pressure and surface tension. In the dead-end passages, the air has nowhere to go in order to allow the water in. So the pressure of the trapped air and the narrowness of the passages (which allows surface tension to help hold the water in place) keeps the water out of the false paths.
With the larger mazes, the water is able to take some false turns as it seeks the lowest possible path. But after awhile the incorrect region fills and the water takes the next lowest path available, which eventually leads it to the outlet.
Toward the end of the video, Steve notes that the large mazes sometimes stop flowing, even though water is still in the reservoir. I’ll quibble slightly here with his explanation, though; I don’t think surface tension is playing as much of a role in this stoppage as friction. The water is basically being driven through a long, narrow pipe, which means quite a lot of friction between it and the walls. Just as you need a certain driving pressure to keep water in a pipe flowing, the maze needs a high enough driving pressure to keep the water going. The point at which drainage stops is the point where the upstream pressure (caused by the depth of the reservoir above the maze) is equal to the pressure lost due to friction in the pipe. All in all, it’s a very cool experiment and a video well-worth watching! (Video and image credit: S. Mould)

Surfactants and Waves
In the ocean, waves often curl over and trap air, becoming plunging breakers. How do surfactants like soap or oil affect this process? That’s the question behind this video, where researchers visualize breaking waves with differing amounts of added surfactant. In the case of pure water, the wave forms a smooth jet that curls over and traps air when the wave breaks. As more and more surfactant gets added, the shape of that jet and cavity change. In one case, they become irregular. In another, they disappear entirely, and with the most surfactant added, the wave suddenly looks just like the water-only case.
The key to these behaviors, it turns out, is not how much surfactant there is, but how much the concentration of surfactant varies along the length of the wave. When there are significant changes in the surfactant concentration along the wave, local Marangoni flows try to even out the surface tension, causing the wave to break up in an irregular fashion. (Image and video credit: M. Erinin et al.)

Paint Ejection
Shaking paint on a speaker cone and filming it in high speed is an oldie but a goodie. Here, artist Linden Gledhill films paint ejection at 10,000 frames per second, giving us a glorious view of the process. As the paint flies upward, accelerated by the speaker, it stretches into long ligaments. As the ligaments thin, surface tension concentrates the paint into droplets, connected together by thinning strands. When those strands break, they snap back toward the remaining paint, imprinting swirling threads of different colors, thanks to their momentum. Eventually, surface tension wins the tug-of-war and transforms all the paint into droplets. (Video and image credit: L. Gledhill)

Bending in Bubbles
Inside a cavity with a square cross-section, bubbles form an array. The shapes of their edges are determined by surface tension and capillarity (lower half of center image). Adding an elastic ribbon into the bubbles (upper half of center image) means that the bubbles’ shapes are determined by a competition between the elasticity of the ribbon and the capillarity of the fluid. Researchers found that they could tune the rigidity of the ribbon to dictate the shape of the bubble array, or, conversely, they could use the bubbles to set the shape of a UV-curable ribbon. (Image and research credit: M. Jouanlanne et al., see also)

Self-Propelled Droplets
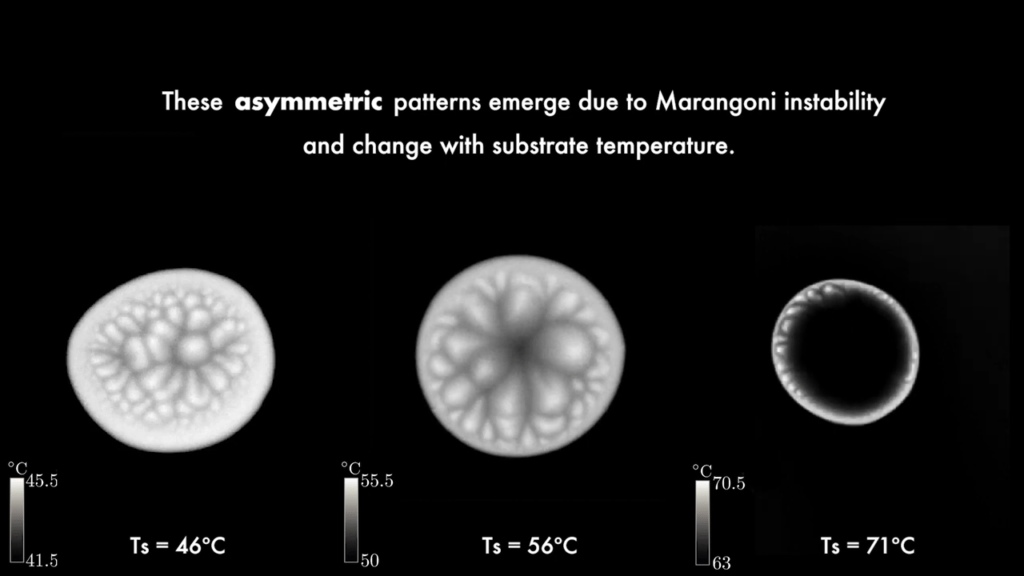
Drops of ethanol on a heated surface contract and self-propel as they evaporate. My first thought upon seeing this was of Leidenfrost drops, but the surface is not nearly hot enough for that effect. Instead, it’s significantly below ethanol’s boiling point. Looking at the drops in infrared reveals beautiful, shifting patterns of convection cells on the drop. The patterns are driven by the temperature difference along the drop; at the bottom, the drop is warmest, and at its apex, it is coldest. Those differences in temperature create differences in surface tension, which drives a surface flow that breaks the drop’s symmetry. The asymmetry, the authors suggest, is responsible for the drop’s propulsion. (Image and video credit: N. Kim et al.)