Pushing a fluid into a porous granular material can fracture it into branching, lightning-like patterns. Here, air is injected into wet grains as a laboratory analog to hydrocarbon extraction or fracturing to treat contaminated soil. The injection of air compacts grains along the branch boundaries, keeping individual branches separated from one another. The patterns that form change with grain shape and ultimately result from the interactions of pressure, surface tension, friction and viscous forces. Studies like these help optimize fluid flow, decontaminate polluted soil faster, and determine risk in gas-driven fracturing of hydrocarbon reservoirs. (Image and video credit: J. Campbell et al.; submitted by B. Sandnes)
Search results for: “surface tension”

Nestling Droplets
Pay attention after a rainfall, and you may notice beads of water gathering in the corners of a spider’s web or along the leaves of a cypress tree (bottom right). Look closely and you’ll notice that the largest droplets don’t form along a straight fiber. Instead they nestle into the corners of a bent fiber (top image). Researchers recently characterized this corner mechanism and found that the angle at which the largest droplets form is about 36 degrees. This angle provides the optimal conditions for capillary action and surface tension to hold large drops in place. At smaller angles, a growing droplet’s weight pulls it down until the thin film holding the droplet near the top ruptures and the droplet falls. At larger angles, a heavy droplet will slowly detach from one side of its fiber and shift toward the other side until its weight is too great for the wetted length of fiber to hold. Then it detaches completely and falls. (Research and image credit: Z. Pan et al.; via T. Truscott)

Paint Balloons
The Slow Mo Guys have a history of personal sacrifice in the name of cool high-speed footage, and their Super Slow Show is no exception. In a recent segment, both Dan and Gav were knocked flat by giant swinging balloons of paint, and, as you might expect, the splashes are spectacular. The speed is just right for some of the paint to form nice sheets before momentum pulls them into long ligaments. Eventually, that momentum overcomes surface tension’s ability to keep the paint together, and the paint separates into droplets, which, as you see below, rain down on the hapless victims. (Video and image credit: The Slow Mo Guys)
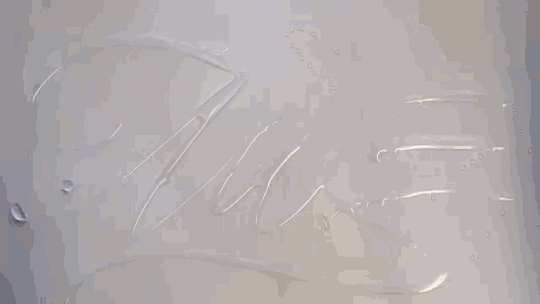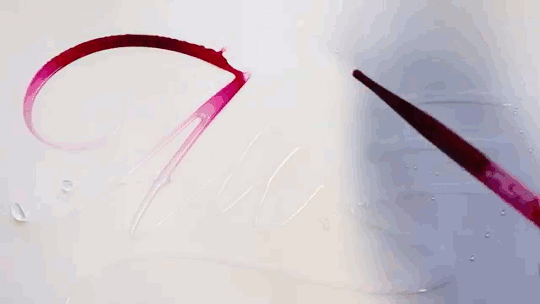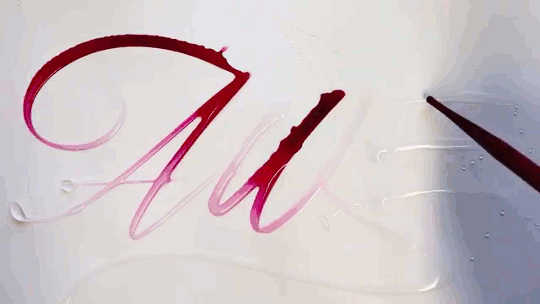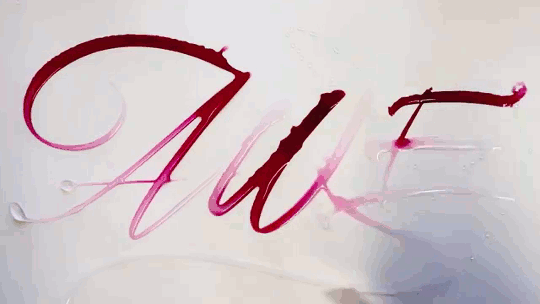
Water Calligraphy
Artist Seb Lester creates calligraphy using ink and water, but not in the way you might expect. After writing in water, the artist applies ink a drop at a time, allowing fluid forces to spread it. There are a few effects at play here. Molecular diffusion – the random motion of molecules – can help two fluids mix, but it’s an extremely slow process. The fast, dramatic spread of ink seen in the video is more likely a Marangoni effect. The water and ink have different surface tensions, creating a gradient in surface tension that depends on the relative concentration of the two fluids. Gradients in surface tension create flow, which is why the ink spreads most quickly when it’s applied in an area that’s pure water. For similar physics, check out maze-solving soaps and the title sequence for “Marco Polo”. (Video and image credit: S. Lester, source; via Gizmodo)

The Fishbone
The simple collision of two liquid jets can form striking and beautiful patterns. Here the two jets strike one another diagonally near the top of the animation. One is slanted into the screen; the other slants outward. At their point of contact, the liquid spreads into a sheet and forms what’s known as a fishbone pattern. The water forms a thicker rim at the edge of the sheet, and this rim destabilizes when surface tension can no longer balance the momentum of the fluid. Fingers of liquid form along the edge, stretching outward until they break apart into droplets. Ultimately, this instability tears the liquid sheet apart. Under the right conditions, all kinds of beautiful shapes form in a system like this. (Image credit: V. Sanjay et al., source)

Swimming Microdroplets
Simple systems can sometimes have surprisingly complex behaviors. In this video, the Lutetium Project outlines a scheme for swimming microdroplets. Most of the droplets shown are just water, but they’re released into a chamber filled with a mixture of oil and surfactants. All flow through the chamber is shut off, but the droplets swim around in complicated, disordered patterns anyway. To see why, we have to zoom way in. The surfactant molecules in the oil cluster around the droplets, orienting so that their hydrophobic parts are in the oil and their hydrophilic parts point toward the water. They actually draw some of the water out of the droplets. This creates a variation in surface tension that causes Marangoni flow, making the droplets swim. Over time, the droplets shrink and slow down as the surfactants pull away more and more of their water and the variations in surface tension get smaller. (Image and video credit: The Lutetium Project; research credit: Z. Izri et al.)


A Hot Tub, Turned Fluidized Bed
Fluidized beds continue to be all the rage among science YouTubers, but Mark Rober supersizes his by turning a broken hot tub into a massive bath of bubbling sand. His video includes a nice explanation of how a granular material like sand gets fluidized as well as how to make your own miniature bed. One of my favorite moments is shown in the animation below. When Mark drops a bowling ball into the fluidized bed, it creates a remarkably liquid-like splash. The ball sprays a splash curtain of sand up on impact and sinks into its own cavity. When the cavity seals behind the ball, it shoots up a tall jet of sand, just like a Worthington jet in water. Even with air fluidizing it, the sand doesn’t have surface tension, though, so the jet breaks up quite differently than water! (Video and image credit: M. Rober; submitted by clogwog)

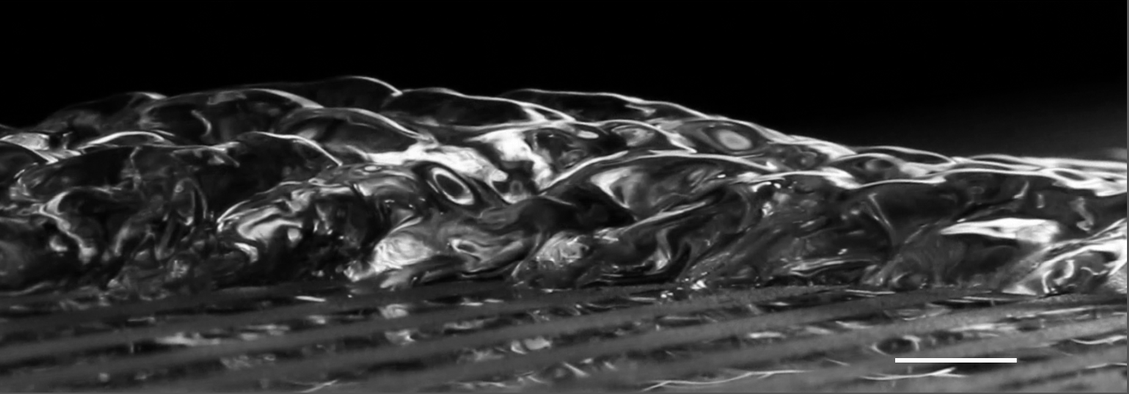
Corrugating Water
The characteristics of a surface can have a major impact on the form a flow takes. The photo above shows a corrugated, almost pinecone-like water surface. It’s the result of a sheet of water flowing over a surface with alternating bands of hydrophobic (water-repelling) and hydrophilic (water-loving) properties. The water sheet narrows over hydrophobic sections and expands over hydrophilic ones. Gravity, inertia, and surface tension compete to create the overall braided appearance. You can see a top-down view of the flow in the original poster. (Image credit: M. Grivel et al., source)

Solving Mazes
Earlier this fall, I attempted my first corn maze. It didn’t work out very well. Early on I unknowingly cut through an area meant to be impassable and thus ended up missing the majority of the maze. Soap, as it turns out, is a much better maze-solver, taking nary a false turn as it heads inexorably to the exit. The secret to soap’s maze-solving prowess is the Marangoni effect.
Soap has a lower surface tension than the milk that makes up the maze, which causes an imbalance in the forces at the surface of the liquid. That imbalance causes a flow in the direction of higher surface tension; in other words, it tends to pull the soap molecules in the direction of the highest milk concentration. But that explains why the soap moves, not how it knows the right path to take. It turns out that there’s another factor at work. Balancing gravitational forces and surface tension forces shows that the soap tends to spread toward the path with the largest surface area ahead. That’s the maze exit, so Marangoni forces pull the soap right to the way out! (Video credit: F. Temprano-Coleto et al.; research credit: F. Peaudecerf et al.)
ETA: Based on the latest research results, gravity may play less of a role than originally thought. Instead, it seems as though the soap chooses its path in part through pre-existing background levels of surfactant. As the dye advances, it compresses the background surfactant, decreasing the local surface tension until it is in equilibrium with dyed area. Because longer paths take longer to reach that equilibrium, the dye spreads preferentially toward the largest surface area.


Blowing Bubbles in Space
Blowing bubbles in your fruit juice is a bad idea when you’re in space, as astronaut Jack Fischer demonstrates. On Earth, gravity dominates water’s behavior, except when things are very small. But in microgravity, a liquid’s other characteristics become more obvious. Adhesion between the straw and juice guides it up and onto Fischer’s face. Surface tension is strong enough to hold the expanding juice bubble together. Capillary action, the ability of fluids to climb up narrow spaces, is far more apparent in microgravity as well, although it’s not important for this demo. We sometimes forget how powerful these forces can be, but microgravity is a good reminder that fluids are more complicated than we think. (Image credit: J. Fischer, source)





