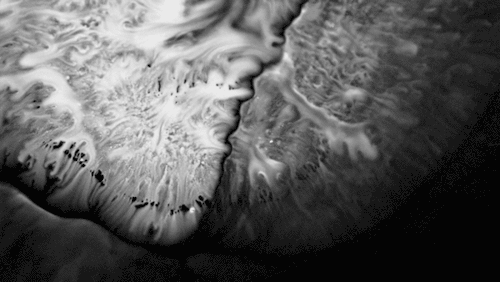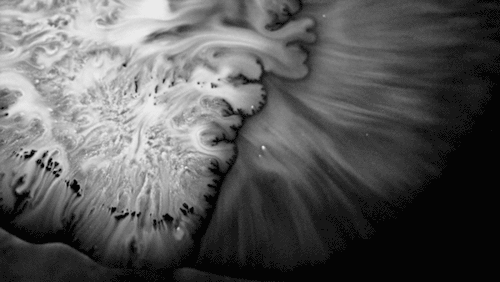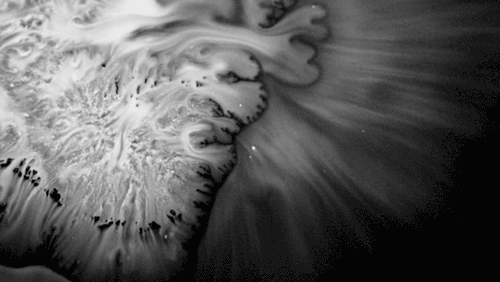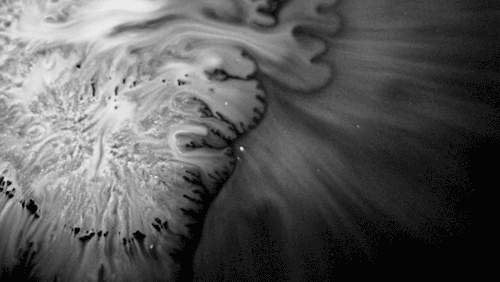
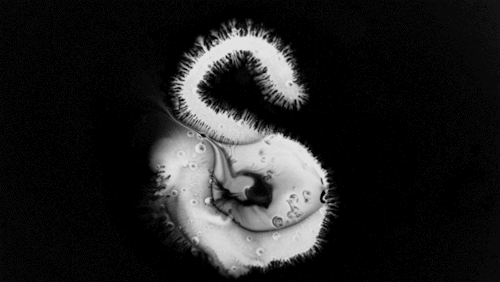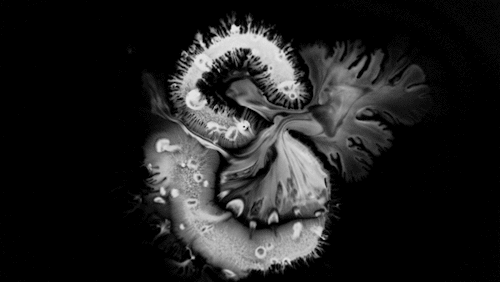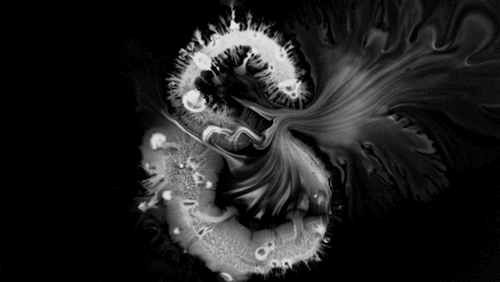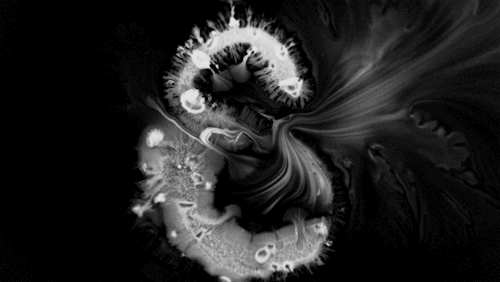
Highly viscous liquids – like cake batter, lava, or the spider silk above – fold as they fall. Several factors impact this instability including the fluid’s density, viscosity, surface tension, and how thin the falling sheet is. As with the coiling of falling honey, this behavior is actually a form of buckling. It’s also fascinating to watch how persistent the layers are. Even out near the edge of the puddle, you can still see individual folds. This is a sign of just how incredibly viscous the spider silk is. Imagine if this were cake batter instead: we’d see folding just like we do with the spider silk proteins, but the individual folds would quickly fade as the batter flowed to fill its container. The spider silk is more viscous, so it’s more resistant to flowing. (Image credit and submission: D. Breslauer, source)
Search results for: “surface tension”

Spinning Droplet Galaxies
Water flung from a spinning tennis ball takes on a shape reminiscent of a spiral galaxy. As it detaches, water leaves the surface with both the tangential velocity of the spinning ball and a radial velocity due to the centrifugal force flinging it. The continued spin of the ball makes the thin ligaments of water still attached to it spiral and stretch. Eventually, surface tension can no longer hold the water together against the centrifugal forces, and the ligaments split into a spray of droplets. (Image credit: W. Derryberry and K. Tierney)

Bubble Art
Everyone loves soap bubbles, and bubble artist Melody Yang reveals how to make some pretty awesome ones in this video for Wired. The surface tension of bubbles makes them naturally seek a shape that minimizes their surface area relative to the volume they contain. For a single bubble, that’s a sphere. But once you start joining multiple bubbles, as Yang demonstrates, that minimal surface area can change, even to something unexpected like a cube.
Bubbles also have an impressive ability to self-heal. As long as whatever passes through them is wet – whether it’s a hand, a straw, or even a ball bearing – the soap film will probably heal itself rather than break. This is a key feature for many of Yang’s tricks, including the impressive planetary bubble. (Video credit: Wired; image credits: Wired/Colossal; via Colossal)

“Liquid Calligraphy”
In “Liquid Calligraphy,” artist Rus Khasanov’s letters dissolve once he draws them. At first, the white ink spreads in narrow fingers, probably driven by a combination of surface tension gradients, capillary action, and simple diffusion. But then, in flashes, the letters morph faster and flow outward. My best guess is that each jump is a spray from a bottle full of a low surface tension liquid like alcohol. The spray triggers faster outflows than before, like those seen when a strong difference in surface tension activates the Marangoni effect. It’s a beautiful and different artistic take on these important fluid forces. Check out more of his videos here or enjoy high-resolution stills and wallpapers in this style from his Behance page. (Image and video credit: R. Khasanov; submitted by TBBQoC)
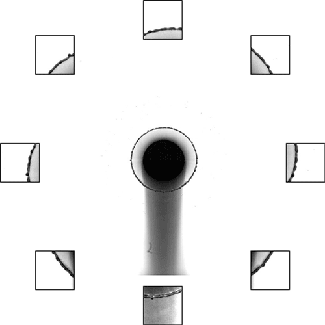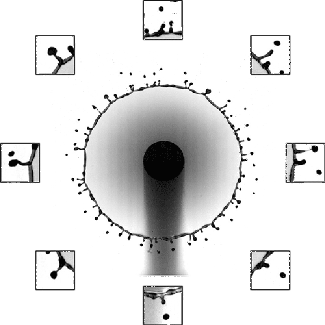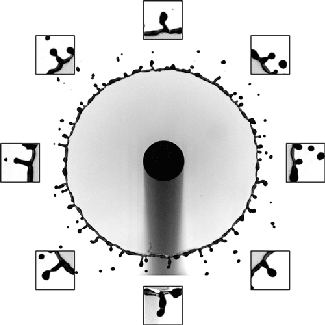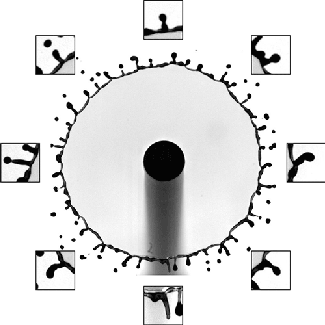
Rim Break-Up
Splashing drops often expand into a liquid sheet and spray droplets from an unstable rim. Although this behavior is key to many natural and industrial processes, including disease transmission and printing, the physics of the rim formation and breakup has been difficult to unravel. But a new paper offers some exciting insight into this unsteady process.
The researchers found that if they carefully tracked the instantaneous, local acceleration and thickness of the rim, it always maintained a perfect balance between acceleration-induced forces and surface tension. That means that even though different points on the rim appear very different, there’s a universality to how they behave. They found that this rule held over a remarkably large range of situations, including across fluids of different viscosities and splashes on various surfaces. (Image and research credit: Y. Wang et al.; via MIT News; submitted by Kam-Yung Soh)

Fractal Fingers
Dyed isopropyl alcohol atop a thin layer of acrylic medium spreads in a fractal fingering pattern. Although the shapes are reminiscent of the viscous fingers seen in in the Saffman-Taylor instability, these patterns are most likely a result of surface tension. The lower surface tension of the alcohol causes Marangoni forces to pull it outward. The branching shapes indicate an instability, likely driven by surface tension, but the details of the mechanism behind it are unclear. (Image credits: J. Nahabetian)

The Disintegrating Splash
A drop of blue-dyed glycerine impacts a thin film of isopropanol, creating a spectacular splash and breakup. The drop’s impact flings a layer of the isopropanol into the air, where air currents make the thin sheet buckle inward and break into a spray of droplets. Meanwhile, the liquid from the drop forms a thick, blue crown that rises and expands outward. When tiny droplets of the isopropanol hit the splash crown, their lower surface tension causes the blue glycerine to pull away, due to the Marangoni effect. This opens up holes in the crown, which grow quickly, until the entire sheet breaks apart. (Image and research credit: A. Aljedaani et al., source)

Snowmelt
Much of the rain that falls on Earth began as snow high in the atmosphere. As it falls through warmer layers of air, the snowflakes melt and form water droplets. The details of this melting process have been difficult to capture experimentally, but a new computational model may provide insight. The basic process has a couple stages. As snow begins to melt, surface tension draws the water into concave areas nearby. When those regions fill up, the water flows out and merges with neighboring liquid, forming water droplets around a melting ice core.
Although this same sequence was observed for many types of snow, scientists also observed some important differences between rimed and unrimed snowflakes. Rime forms when supercooled water droplets freeze onto the surface of a snowflake. Lightly rimed snow still looks light and fluffy, like the animation above, but heavily rimed snow forms denser and more spherical chunks. Because there are lots of porous gaps in heavily rimed snow, water tends to gather there during initial melting. Rimed snow was also more likely to form one large water droplet rather than breaking into multiple droplets like snow with less rime. For more, check out NASA’s video and the Bad Astronomy write-up. (Image credit: NASA, source; research credit: J. Leinonen and A. von Lerber; via Bad Astronomy; submitted by Kam Yung-Soh)

Happy 2000 Posts!
Happy Friday and happy 2000th FYFD post! To celebrate, I played with surface tension and the Marangoni effect to make some art. For a run-down on the physics, check out this previous post on water calligraphy. Two thousand posts feels like a major milestone. Not everyone realizes this, but FYFD is a one-woman operation, so 2000 posts is a whole lot of research, image editing, and writing. For fun, I’m including here eight completely random FYFD entries, representing less than one-half of one percent of my total archives:
1. Why did Chinese junks put holes in their rudders?
2. Making droplets in an ultrasonic humidifier
3. Floating on a granular raft
4. Air-trapping fur keeps otters warm
5. The physics of the knuckleball
6. What makes badminton so fast?
7. Playing with fluorescein
8. How frost formsWant to keep up the random walk? Use https://fyfluiddynamics.com/random to find random entries, or if you prefer your browsing to be more directed, check out the visual archive or the themed series page.
As always, a special thanks to those who help support FYFD through Patreon subscriptions – I couldn’t keep writing and making videos without your help! And thank you to all of you who read and share FYFD. Whether you’ve been following along for a week or for the last eight years, your enthusiasm keeps me motivated! Thank you!
(Image credits: 2k animation – N. Sharp; Chinese junk ship – Premier Ship Models; ultrasonic humidifier – S. J. Kim et al.; granular raft – E. Jambon-Puillet and S. Protiere; 3D-printed “fur” – F. Frankel; knuckleball – L. Kang; shuttlecock – Science Friday; fluorescein – Shanks FX; freezing droplets – J. Boreyko et al.)

Spinning Paint
Several years ago Fabian Oefner started spinning paint, and it’s been a perennial favorite online ever since. Here the Slow Mo Guys revisit their own paint-spinning antics by super-sizing their set-up. In some respects, it’s a little dissatisfying; as with their first time around, they don’t moderate the drill speed at all, so after the initial spin-up, the centrifugal acceleration is so strong that it just shreds the paint instead of showing off the interplay between the acceleration and surface tension’s efforts to keep the paint together.
In their largest experiment, though, the Slow Mo Guys get some interesting physics. Here there’s only a single slot for paint to exit, so the set-up doesn’t lose all its paint at once. The centrifugal acceleration flings the paint out in sheets that stretch into ligaments and then tear into droplets as they move further out. But there’s some more complicated phenomena, too. Notice the bubble-like shapes forming in the yellow paint on the lower right. These are known as bags, and they form because of the relative speed of the paint and the air it’s moving through. This is actually the same thing that happens to falling drops of rain! (Video and image credit: The Slow Mo Guys)