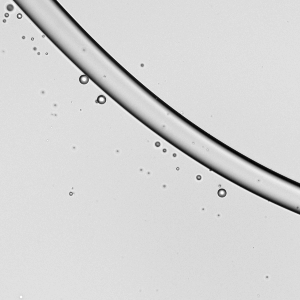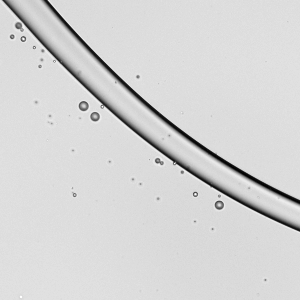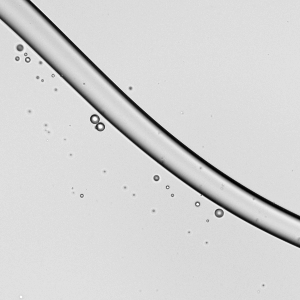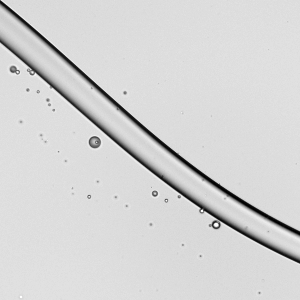
Spreading paint with a brush or with fingers is familiar activity for most people. It’s also similar to processes used in industry for spreading thin layers of paint and other complex fluids. In a recent study, researchers took a look at how a soft, elastic blade (similar to a paintbrush or one’s fingers) spreads shear-thinning fluids (like paint) and Newtonian fluids (like water). Surprisingly, they found that it actually takes 30% more mechanical work to spread a shear-thinning fluid than the same volume of an equivalent Newtonian one. That’s pretty much the opposite of what we’d expect since the action of spreading (and shearing) the complex fluid should reduce its viscosity. However, they did find that the shear-thinning fluid spreads to a thin layer more consistently than the Newtonian fluid does. (Image credit: A. Kolosyuk; research credit: M. Krapez et al.)
Search results for: “shear”

Stably Jammed
Granular materials like sand, gravel, and medications can become a rigid mass when squeezed or sheared. Even with a relatively loose packing, these materials can jam together to act like a solid if the contacts between grains no longer allow particles to shift or rotate. In this video, researchers explore how stable these jammed states are by repeatedly shearing the mixture and observing how it changes.
Most of the videos are set up as a triptych, where all three panels show the same material. On the left, you see a simple view showing the position of each particle. In the middle, the disks are viewed through polarized filters, so that the material looks brightest where it is stressed. This view lets us see the force chains that run through the material. On the right, UV-sensitive ink on each marker glows to show any rotation particles experience.
In the first sample, repeated shearing slowly unjams the mixture and allows it to shift and flow once more. We see this from the decreasing brightness in the middle panel. The slow fade to black means that the force chain network has disappeared entirely. In contrast, the second sample ultimately reaches an “ultra-stable” jammed state, in which further shear cycles cause no change to the network. Once again, this is easiest to observe in the middle image, where the bright force network stops changing after 2,000 cycles or so. (Image and video credit: Y. Zhao et al., research pre-print)

Asperitas Formation
In 2017, the World Meteorological Organization named a new cloud type: the wave-like asperitas cloud. How these rare and distinctive clouds form is still a matter of debate, but this new study suggests that they need conditions similar to those that produce mammatus clouds, plus some added shear.
Using direct numerical simulations, the authors studied a moisture-filled cloud layer sitting above drier ambient air. Without shear, large droplets in this cloud layer slowly settle downward. As the droplets evaporate, they cool the area just below the cloud, changing the density and creating a Rayleigh-Taylor-like instability. This is one proposed mechanism for mammatus clouds, which have bulbous shapes that sink down from the cloud.
When they added shear to the simulation, the authors found that instead of mammatus clouds, they observed asperitas ones. But the amount of shear had to be just right. Too little shear produced mammatus clouds; too much and the shear smeared out the sinking lobes before they could form asperitas waves. (Image credit: A. Beatson; research credit: S. Ravichandran and R. Govindarajan)

Dispelling Ice
In winter weather, delays pile up at airports when planes need de-icing. Our current process involves spraying thousands of gallons of chemicals on planes, but these chemicals are easily removed by shear stress and dissolution, meaning that by the time a plane takes off, there is little to no de-icing agent remaining on the plane. Instead, those chemicals become run-off.
Researchers looking to change that have developed a family of anti-icing coatings — including creams, sprays, and gels — that are easy to use and apply, non-toxic, and much longer lasting than conventional methods. Ice slides easily off their gel coatings, which remain optically transparent even under freezing conditions — and ice can take 25 times longer to form on the gels compared to current anti-icing tech.
The team envisions using their coatings on much more than airplanes. Imagine traffic lights that can’t be obscured by ice or snow, a windshield on your car that never freezes over, or even an anti-icing spray that could protect crops from a sudden freeze! (Image, video, research, and submission credit: R. Chatterjee et al.; see also)

Mixing the Immiscible
Immiscible liquids — like oil and water — do not combine easily. Typically, with enough effort, you can create an emulsion — a mixture formed from droplets of one liquid suspended in the other — like the one above. But a team of researchers have taken mixing immiscible liquids to a new level using their Vortex Fluid Device (VFD).
Longtime readers may remember the group from their Ig-Nobel-winning demonstration of unboiling an egg, but this time the team is used the VFD to mix and de-mix immiscible liquids. As shown in the video below, the VFD is essentially a fast-spinning tube tilted at a 45-degree angle. As it spins, the liquids inside are forced into thin films with very high shear rates — high enough that immiscible liquids like water and toluene are forced together without forming an emulsion. Essentially, the mechanical forces mixing the liquids are strong enough to overcome the chemistry that typically keeps them apart.
Impressively, the device manages this without using harsh surfactants or catalysts that other methods rely on. As a result, the technique offers a greener method for mixing chemicals for pharmaceuticals, cosmetics, food processing, and more. (Image credit: pisauikan; research credit: M. Jellicoe et al.; video credit: Flinders University; submitted by Marc A.)

Beijing 2022: Why Are Ice and Snow Slippery?
Although every Olympic winter sport relies on the slippery nature of snow and ice, exactly why those substances are so slippery has been an enduring mystery. Michael Faraday hypothesized in the nineteenth century that ice may have a thin, liquid-like layer at its surface, something that modern studies have repeatedly found.
One recent study used an entirely new instrument to probe the characteristics of this lubrication layer and found that it is only a few hundred nanometers thick. But the fluid in this layer is nothing like the water we’re used to. Instead it has a viscosity more akin to oil and its response to deformation is shear-thinning and viscoelastic, more like the complex fluids in our kitchens and bodies than pure, simple water. They found that using a hydrophobic probe modified the interfacial viscosity even further, which finally provides a hint at the mechanism behind waxing skis and skates.
Fortunately for us, we’ve found plenty of ways to employ and enjoy water’s slipperiness, even as the mystery of it slowly gives way to understanding. (Image credit: M. Fournier; research credit: L. Canale et al.; via Physics World; submitted by Kam-Yung Soh)
Changing with the Flow
Chemically-reacting flows are some of the toughest problems to unravel. In this new study, researchers found that the very act of flowing through narrow channels can change the speed of chemical reactions. In particular, they found that protein molecules carried through a capillary tube (comparable in size to human capillaries) changed their local shape as a result of the shear forces they experienced. Those changes actually sped up the proteins’ chemical reactions compared to the reaction speed for the chemicals in bulk.
That finding suggests two important takeaways: 1) chemicals may be absorbed in the human bloodstream differently in capillaries than in other parts of the cardiovascular system, and 2) mimicking these tiny capillaries in microfluidic devices could be useful in speeding up certain biochemical reactions. (Image credit: top – KazuN, visual abstract – T. Hakala et al.; research credit: T. Hakala et al.; via Science; submitted by Kam-Yung Soh)

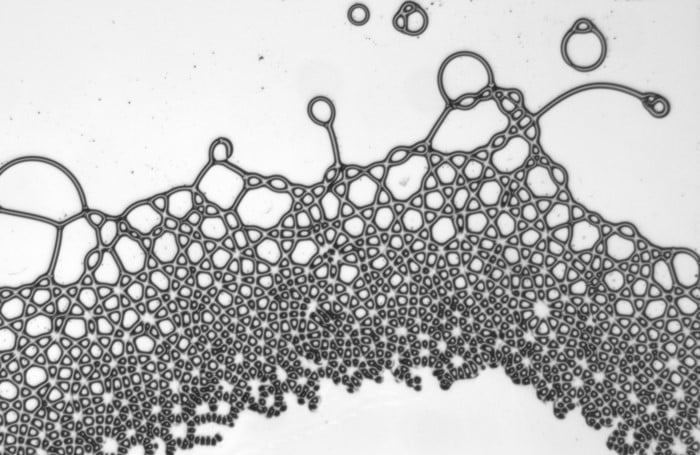
Driven From Equilibrium
With the right application of force, liquids can take on shapes that defy our intuition. Here researchers sandwiched two immiscible oils between glass slides and applied an electric field. Because the two oils have different electrical responses, charges build along the interface between them. These charges lead to non-trivial electrohydrodynamic flows and a multitude of bizarre shapes. They observed polygonal droplets, streaming droplet lattices, and spinning filaments among others. As long as the electric field remains on, the wild behaviors continue; once the field is turned off, the oils relax back to typical, rounded drops. (Image, video, and research credit: G. Raju et al.; via Physics World)
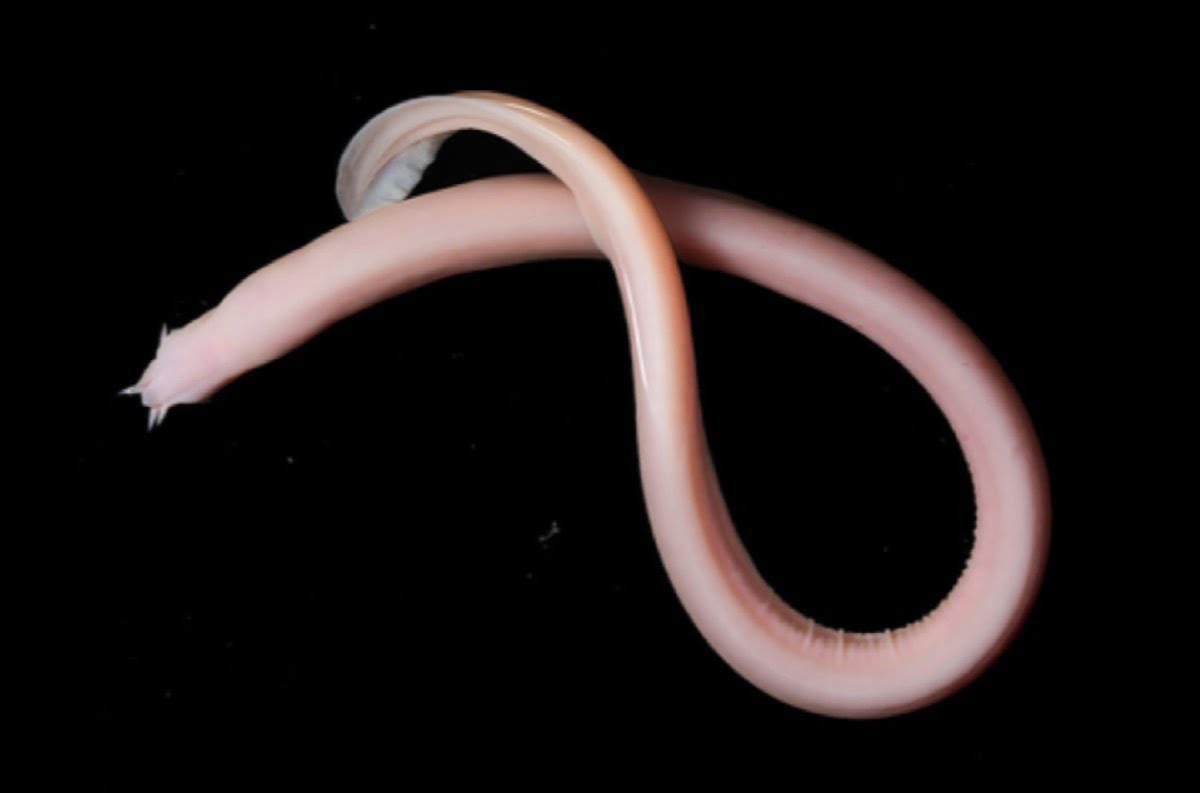
Hagfish Slime
The eel-like hagfish is a superpowered escape artist, thanks to its slime. When threatened, the hagfish releases long protein-rich threads that, when combined with turbulent sea water, unravel to form large volumes of viscoelastic slime that clog the gills of its predators. A new study shows that larger hagfish produce longer and thicker threads in their slime, enabling them to escape larger predators than their smaller brethren can.
The properties of hagfish slime are tuned for defense. When stretched, the long protein threads resist, making the slime more viscous. Since most fish use suction methods to catch prey, that means a predator attacking a hagfish will quickly exacerbate its slimy problems. But the hagfish itself can easily escape its slime by tying itself in a knot. The threads inside the slime collapse when sheared, so the knot-tying of the hagfish slips the slime right off. (Image credit: T. Winegard; research credit: Y. Zeng et al.; via Ars Technica; submitted by Kam-Yung Soh)

Breaking Up Is(n’t) Hard to Do
Engineers often need to break a liquid jet up into droplets. To do so quickly, they surround the jet with a ring of fast-moving air in a set-up known as a coaxial jet. Shear between the gas and liquid creates instabilities that quickly distort the jet’s initial cylinder into sheets and ligaments. Those formations then undergo their own instabilities to break up into drops. The method is, as you can see in the high-speed images above, quite effective, though the breakup mechanism itself is tough to quantify. (Image credit: G. Ricard et al.)