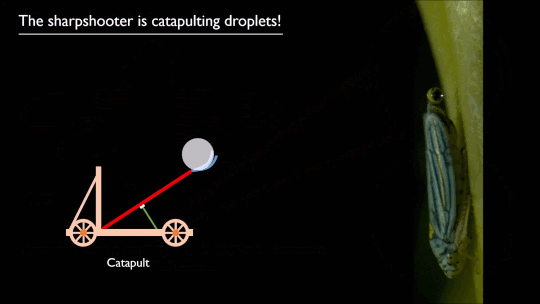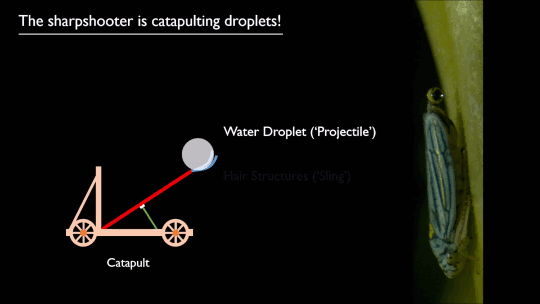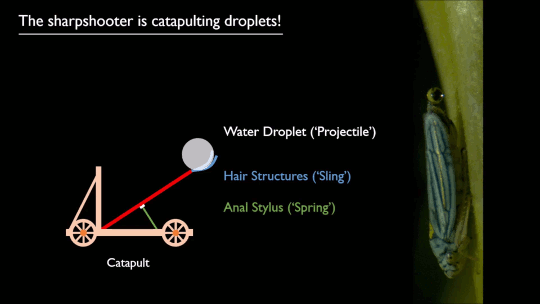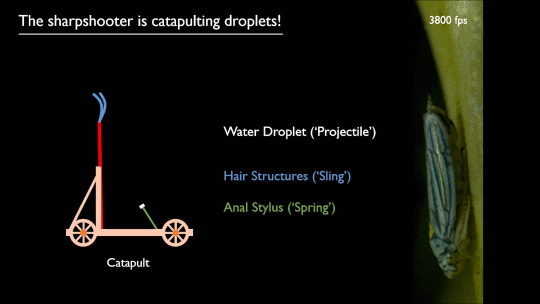
Researchers put a spin on splashing droplets with selective wetting. When a drop impacts on a water-repellent, superhydrophobic surface, it will spread circularly, then pull back together and rebound off the surface. That’s because the surface coating resists actually touching – or being wetted by – the water. But just as there are surface coatings that resist water, there are those that attract it.
Above, researchers have coated a surface so that it’s mostly superhydrophobic, but it also has narrow pinwheel-like arms that are hydrophilic. As the drop impacts, it spreads across the surface and then retracts. But where the hydrophilic arms are, the drop lingers. This creates the four lobes we see on the droplet, and the asymmetric retraction gives the drop angular momentum. As it leaves the surface, the spin continues. In some configurations, the researchers could make the drop spin at more than 7300 rpm. (Image and research credit: H. Li et al; via Science; submitted by Kam-Yung Soh)