In this video, the Slow Mo Guys play with liquid gallium, giving us a chance to see how molten metals behave (outside of, say, the Terminator movies). Near its melting point, gallium is about six times denser than water, with a viscosity three times higher, and a surface tension about ten times greater. So how do those properties affect its behavior?
You may be surprised that when watching the gallium vibrate on a speaker or get poured into a pan, it doesn’t look all that different from water. Yes, it’s highly reflective, but, on the whole it doesn’t look radically different from a distance. We can use the Reynolds number to quantify what’s going on here. It’s a dimensionless number that compares the fluid’s inertial force to the viscous force. Imagine we have two versions of an experiment, one where we pour gallium at a given speed and one pouring at the same volume and speed but with water. If we compared the Reynolds numbers of the water and the gallium, they only differ by a factor of two. Overall, that’s not very much. That’s why the two pours look similar.
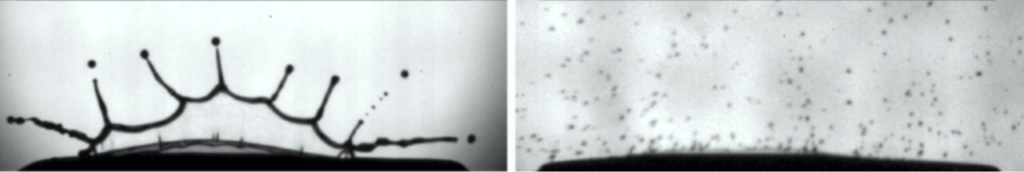
The story is different, though, if we look at individual drops of gallium and water, like when the first few drops of our pour hit the surface. Check out the gallium drops below. They’re conical on either end! This looks very different from what we expect with water droplets. You might think that’s because the metal is more viscous, but if we compare a water drop with a gallium drop of the same characteristic size and impact speed, we find a different story. For this, we’ll use the Ohnesorge number, which compares the viscous forces to a combination of inertia and surface tension. In this case, we find that the gallium drop’s Ohnesorge number is almost an order of magnitude smaller than the water droplet’s. That means that viscosity isn’t a major factor for our gallium drop. Both surface tension and inertia are more important.
But if the surface tension is so high, then why aren’t the droplets spherical? Mostly because they don’t have time to form spheres before they hit. Their shape suggests that they’ve only just broken into droplets, which makes sense if the pour is fast and the surface tension is strong. (Video and image credit: The Slow Mo Guys)