Antibubbles are peculiar and ephemeral creations. A bubble typically encloses a gas within a thin layer of fluid. As the name suggests, an antibubble does the opposite: it’s a thin film of gas enclosing a liquid droplet within a larger background liquid. That thin gas film makes antibubbles extremely delicate. Disturb it at all – as the thinning jet at the top of the animation above does – and that film will break apart, much like a soap bubble. To see more antibubble action, check out some of our previous entries, including antibubbles in a vortex and a simple way to create antibubbles. (Image credit: C. Kalelkar and S. Phansalkar, source)
Search results for: “droplet”

Using Paper to Avoid Splashback
Daily life and countless pool parties have taught us all that objects falling into water create a splash. Sometimes that splash is undesirable, and while there are many ways to tune a splash – by adding surfactants or changing the fluid’s viscosity – there’s a relatively common one that’s escaped scientific study until now. Researchers looked at how splashes change when you add a thin, penetrable fabric – commonly known as toilet paper – to the water surface.
Now, the common assumption is that adding a sheet of toilet paper can prevent splashback, but the story is not quite that simple. On the left, you see a splash generated without toilet paper. Because the ball is hydrophilic (water-loving), it does not pull any air into a cavity as it passes. There’s a nice axisymmetric Worthington jet formed, and it doesn’t splash very high, although some of the satellite droplets go quite a bit higher.
On the right, we see a splash with a single sheet of toilet paper. In this case, the impact of the sphere penetrates the paper, and the way the paper deforms causes air to get sucked down into a cavity behind the ball. That creates a wider, amorphous jet that rebounds higher than the jet in clean water, though it does not shed satellite drops.
The researchers found that single and even double sheets of toilet paper can actually increase the height of the splash jet if the object penetrates them. The hole the object makes actually helps focus the jet. Adding a couple more layers, though, can eliminate splashing completely. (Image and research credit: D. Watson et al.)

Coalescence
Simple acts like the coalescence of two droplets sitting on a surface can be beautiful and complex. As the droplets come together, they form a thin neck between them, and the curvature of that surface causes capillary forces that drive fluid into the neck. For two dissimilar droplets, like the ones above, there can be additional forces. Here, the upper drop is pure water, but the lower one has added surfactants, which reduce its surface tension. That difference in surface tension creates a Marangoni flow that tends to pull fluid away from the neck. The result is that full coalescence takes longer. Depending on other factors in this tug-of-war between capillary action and Marangoni flow, the process of coalescence can look very different. In this example, there’s a fingering instability that occurs as the neck spreads. Change the circumstances slightly and the drops may chase each other instead of merging or will merge with a perfectly smooth contact front. (Image and research credit: M. Bruning et al.)

A Star Drop
There are many ways to make a droplet oscillate in a star-shape – like vibrating its surface or using acoustic waves to excite it – but these methods involve externally forcing the droplet’s oscillation. Leidenfrost drops – liquids levitating on a film of their own vapor caused by the extremely hot surface below – turn themselves into stars. It all starts with the constant evaporation driven by the heat below. This creates a thin, fast-moving layer of vapor flowing beneath the drop. That vapor shears the drop, causing capillary waves – essentially ripples – that travel through the drop in a characteristic way. Those ripples in turn cause pressure oscillations in the vapor layer, alternately squeezing and releasing it. Feedback from the vapor layer then drives the droplet into star-shaped oscillations. Under the right conditions, water drops can form stars with as many as 13 points! (Image and research credit: X. Ma and J. Burton, source)

A Viscous Splash
The splash of a drop may be commonplace, but it is still a mesmerizing and fertile phenomenon. When it comes to splashing, scientists are still learning how to predict the outcome. Here a drop of silicon oil impacts a film of silicon oil with an even higher viscosity. The momentum of that impact creates a crater and a splash curtain that rises and expands from the initial point of impact. Because the film viscosity is higher than the drop’s, the evolution of the corona slows down. Eventually, surface tension and gravity start pulling the splash curtain back down as the crater collapses. Meanwhile at the top of the splash, capillary forces pull fluid into the rim, which becomes unstable and grows cusps that eventually eject a cloud of smaller droplets. (Image and research credit: H. Kittel et al., source)

Breaking With a Wave
For rocket combustion and other applications, like watering your lawn with a hose, a stream of fluid may need to be broken up into droplets. While simply spraying a liquid jet will make it break up, waving that jet back and forth will break it up faster. A recent study simulated this problem numerically to determine the exact mechanisms driving that break-up. The researchers found two major culprits.
The first is a Kelvin-Helmholtz, or shear-based, instability. When a jet leaves the nozzle, there’s friction between it and the comparatively still air surrounding it. This creates tiny ripples in the surface that eventually grow into the distortions we can see, and it’s found in all jets, regardless of their side-to-side motion.
The second culprit, which is only found in the oscillating jet, is a Rayleigh-Taylor instability. By moving the jet side-to-side, you’re driving the dense liquid into less dense air, which creates a different set of disturbances that also help break up the jet. The final result: swinging the jet side-to-side breaks it into smaller droplets faster. (Image and research credit: S. Schmidt et al.)

Collecting Fog
In some parts of the world, fog is a major source of freshwater, but collecting it is a challenge. Most systems use a wire mesh to capture and collect droplets, but the process is highly inefficient, pulling only 1-3% of droplets from the fog. Researchers found that this is due largely to aerodynamic effects. The presence of the wire deflects droplets around it (bottom left). To solve this, engineers introduced an electric charge into the fog. The subsequent electric field actually pulls droplets to the wires (bottom right). When applied to a mesh (top), the efficiency of fog capture improves dramatically.
The technique can also be used to capture water vapor that would otherwise escape from the cooling towers of power plants. The MIT researchers who developed the technique will conduct a full-scale test at the university’s power plant this fall. They hope the technique will recapture millions of gallons of water that would otherwise drift away from the plant. (Image credits: MIT News, source; image and research credits: M. Damak and K. Varanasi, source)

Using Embolisms to Fight Cancer
Blocking blood vessels by creating embolisms is, under most circumstances, very bad. But researchers are exploring ways to fight cancer by intentionally and strategically creating these blockages. In gas embolotherapy, researchers inject fluid droplets, which can carry chemotherapy drugs, into the bloodstream. Once they circulate into a cancerous tumor, they use ultrasound to vaporize the droplet and create a gas bubble. Those bubbles lodge inside the capillaries of the tumor, starving it of fresh blood and trapping the chemotherapy drugs inside. It’s a one-two punch to the cancer. Without blood flow, the cancer cells die, and, since the cancer-killing drugs get mostly trapped inside the tumor, patients may require lower dosages and endure fewer side effects. The technique is currently in animal testing, but hopefully it will be a valuable therapy for human patients in the future. (Image credit: Chemical & Engineering News; research credit: Y. Feng et al.; via AIP)
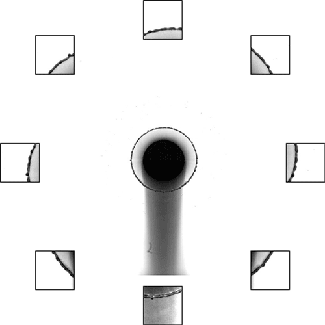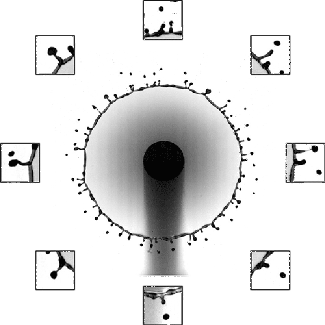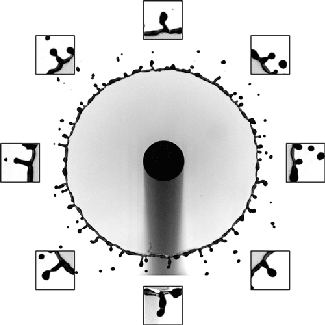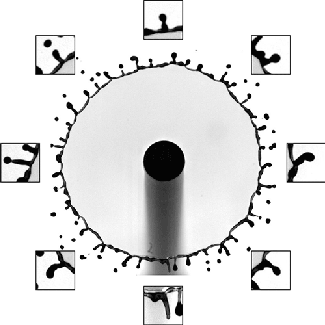
Rim Break-Up
Splashing drops often expand into a liquid sheet and spray droplets from an unstable rim. Although this behavior is key to many natural and industrial processes, including disease transmission and printing, the physics of the rim formation and breakup has been difficult to unravel. But a new paper offers some exciting insight into this unsteady process.
The researchers found that if they carefully tracked the instantaneous, local acceleration and thickness of the rim, it always maintained a perfect balance between acceleration-induced forces and surface tension. That means that even though different points on the rim appear very different, there’s a universality to how they behave. They found that this rule held over a remarkably large range of situations, including across fluids of different viscosities and splashes on various surfaces. (Image and research credit: Y. Wang et al.; via MIT News; submitted by Kam-Yung Soh)

Bouncing Off a Moving Wall
There are many ways to repel droplets from a surface: water droplets will bounce off superhydrophobic surfaces due to their nanoscale structures; a vibrating liquid pool can keep droplets bouncing thanks to its deformation and a thin air layer trapped under the drop; and heated surfaces can repel droplets with the Leidenfrost effect by vaporizing a layer of liquid beneath the droplet. But all of these methods will only work for certain liquids under specific circumstances.
More recently, researchers have begun looking at a different way to repel droplets: moving the surface. The motion of the plate drags a layer of air with it; how thick that layer of air is depends on the plate’s speed. (Faster plates make thinner air layers.) Above a critical plate speed, a falling droplet will impact without touching the plate directly and will rebound completely. This works for many kinds of liquids – the researchers used silicone oil, water, and ethanol – across many droplet sizes and speeds. The key is that the air dragged by the plate deforms the droplet and creates a lift force. If that lift force is greater than the inertia of the droplet, it bounces. (Image and research credit: A. Gauthier et al., source)









