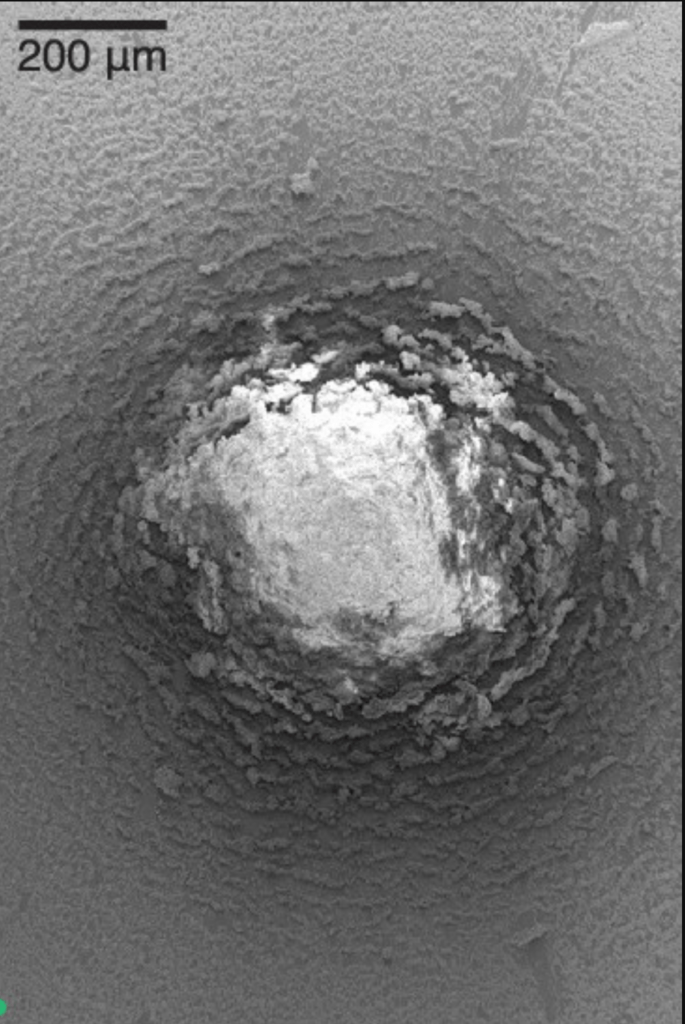
Granular mixtures show surprising similarities to fluids, even though their underlying physics differ. The latest example of this is a Rayleigh-Taylor-like instability that occurs when heavy particles sit atop lighter ones. By combining vertical vibration and an upward gas flow, researchers found that the lighter particles form fingers and bubbles that seep up between the heavier grains (upper left). Visually, it looks remarkably similar to a lava lamp or other Rayleigh-Taylor-driven instability (upper right).
But the physics behind the two are distinctly different. In the fluid, buoyancy drives the instability while surface tension acts as a stabilizing force. There’s no surface tension in a granular material, though. Instead, the drag force from gas flowing upward provides the vertical impetus while friction between the grains – essentially an effective viscosity – replaces surface tension as a stabilizing influence.
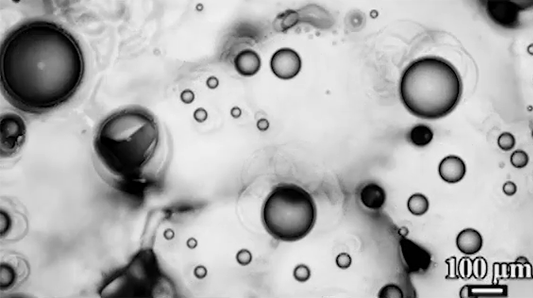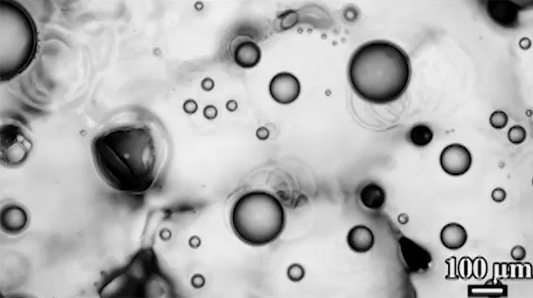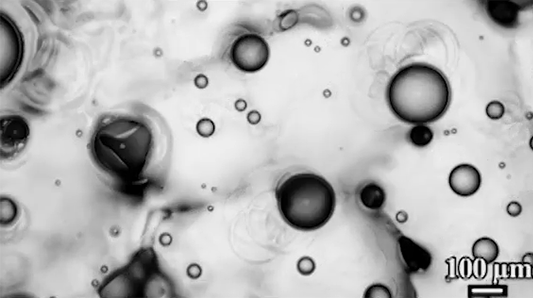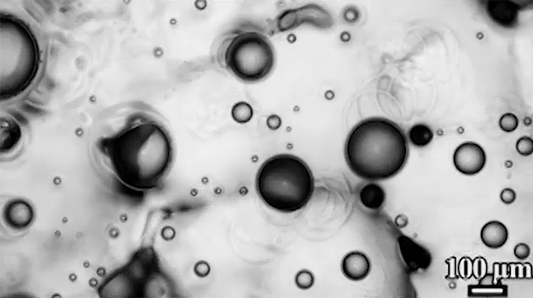
The similarities don’t stop there, though. When the researchers tested a “bubble” of heavy grains suspended in lighter ones (lower left), they found that, instead of sinking, the granular bubble split in two and drifted downward on a diagonal. Eventually, those daughter bubbles also split. Again, visually, this looks a lot like what happens to a drop of ink or food coloring falling through water (lower right), but the physics aren’t the same at all.
In the fluid, the breakup happens when a falling vortex ring splits. In the granular example, gas moving upward tends to channel around the heavy grains because they’re harder to move through. Eventually, this builds up a solidified region under the bubble. When the heavy grains can’t move directly down, they split and sink through the surrounding suspended particles until they build up another jammed area and have to split again. (Image credits: granular RTI – C. McLaren et al.; RTI simulation – M. Stock; bag instability – D. Zillis; research credit: C. McLaren et al.; submitted by Kam-Yung Soh)