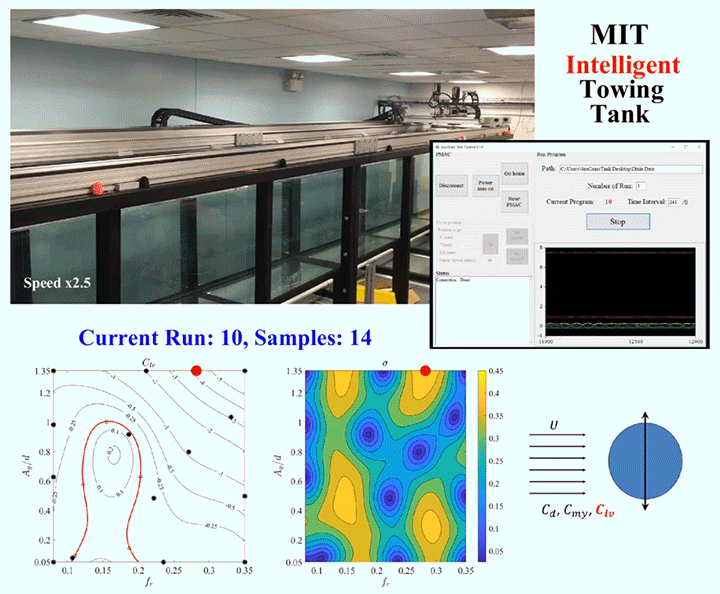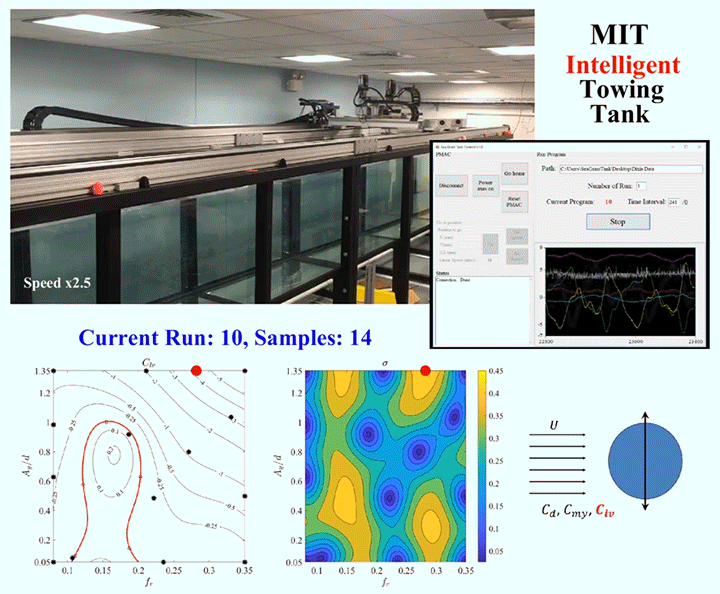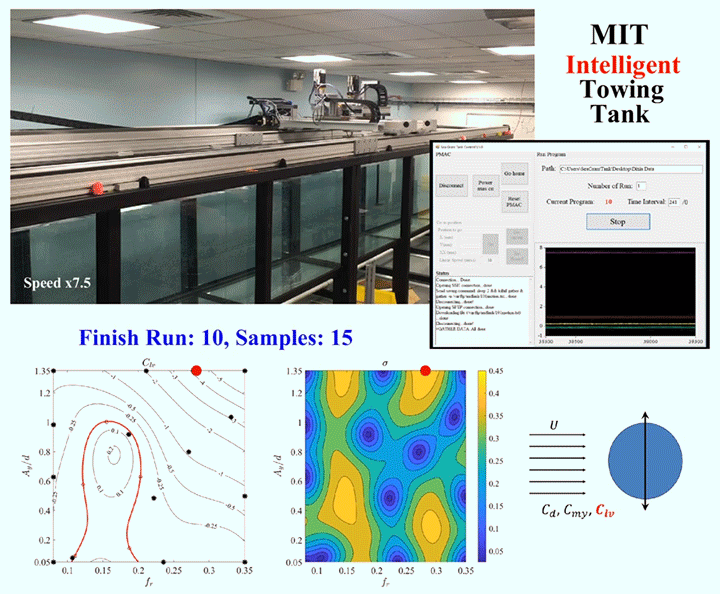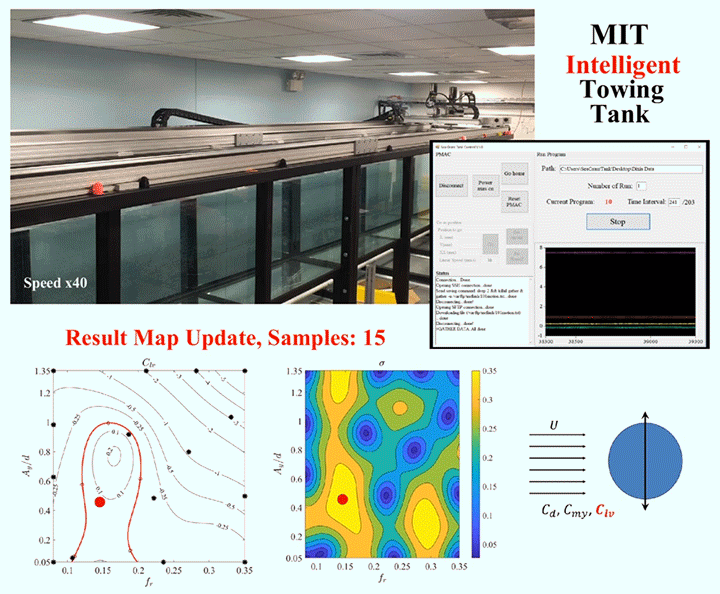
One of the major challenges in fluid dynamics is the size of the parameter spaces we have to explore. Because many problems in fluid dynamics are non-linear, making small changes in the initial set-up can result in large differences in the results. Consider, for example, a simple cylinder towed through a water tank. As the cylinder moves, vortices will form around it and shed off the back, causing the cylinder to vibrate. The details of what will happen will depend on variables like the cylinder’s size and flexibility, the speed it’s being towed at, and which directions it’s allowed to vibrate in. Mapping out the parameter space, even sparsely, could take a graduate student hundreds of experiments.
To speed up this process, engineers are now building robotic facilities like the Intelligent Towing Tank (ITT) shown above. Like graduate students, the ITT can work into the wee hours of the night, but, unlike graduate students, it never needs to eat, sleep, or stop experimenting. Now, one could use a facility like this to brute-force the answers by testing every possible combination of parameters, but even working 24 hours a day, that would take a long time. Instead, researchers use machine learning to guide the robotic facility into choosing test parameters in a way that optimizes the factors the researchers define as important.
Essentially, the system starts with experiments chosen at random within the parameter space, and then uses those results to select areas of interest until it’s gathered enough data to satisfy the limits specified by human researchers. In theory, a well-designed algorithm can dramatically reduce the number of experiments needed to explore a parameter space. (Image and research credit: D. Fan et al.; submitted by Kam-Yung Soh)