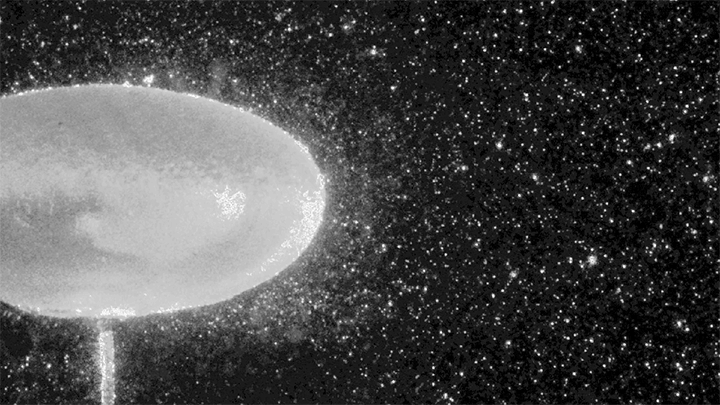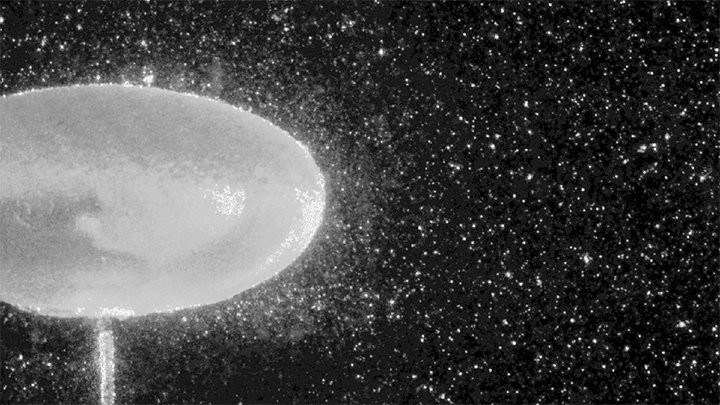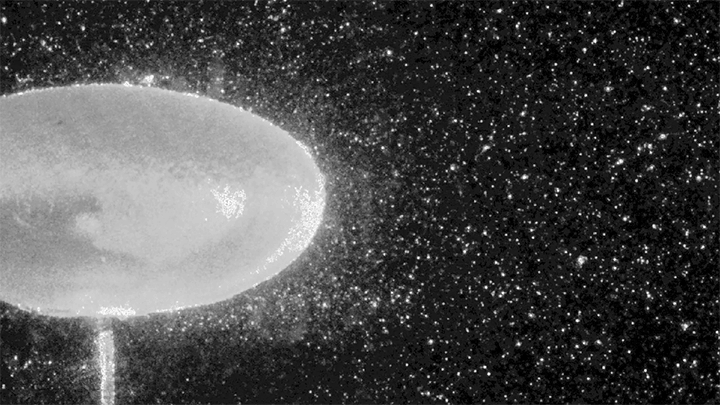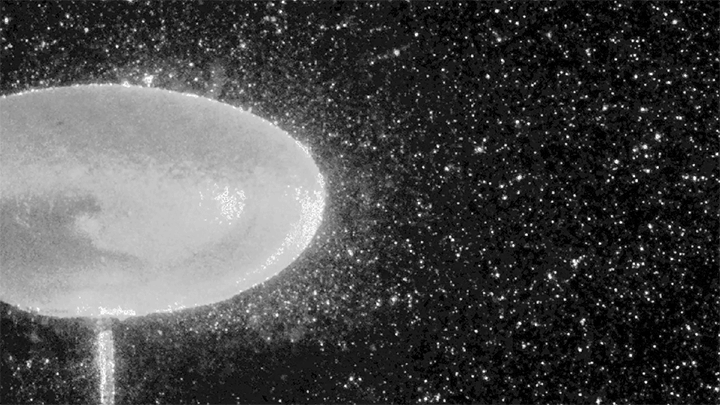
Where waves crash and meet, turbulence is inevitable. But exactly how large waves interact — whether in the ocean, in plasma, or the atmosphere — is far from understood. A new experiment is teasing out a better physical understanding by tweaking a variable that’s been hard to change: gravity.
To do so, the researchers conduct their experiments in a large-diameter centrifuge (shown above) where they can create effective gravitational forces as high as 20 times Earth’s gravity. This increases the range of frequencies where gravity-dominated waves occur by an order of magnitude.
By studying this extended frequency range, the authors found something unexpected: the timescales of wave interactions did not depend on wave frequency, as predicted by theory. Instead, those interactions were dictated by the longest available wavelength in the system, a parameter set by the size of the container. It will be interesting to see if future work can confirm that result with even larger containers. (Image credit: ocean waves – M. Power, others – A. Cazaubiel et al.; research credit: A. Cazaubiel et al.; via APS Physics; submitted by Kam-Yung Soh)