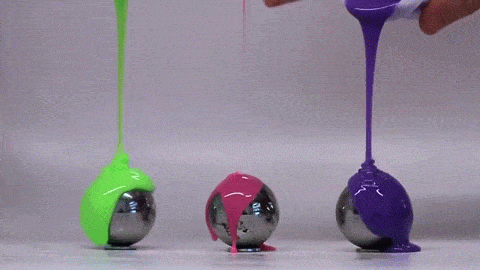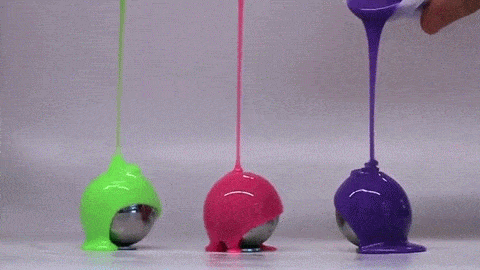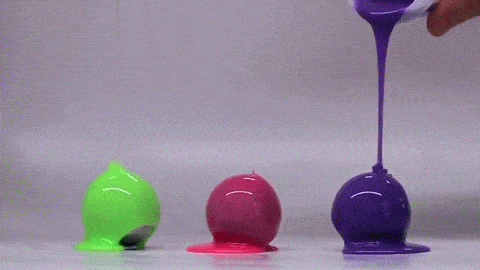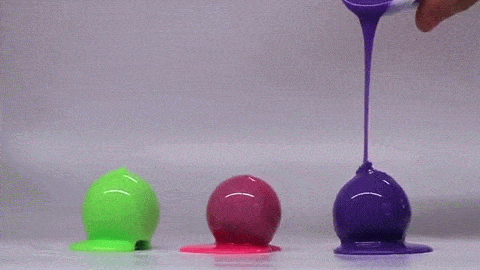If you’ve ever bitten into a chocolate-covered bonbon, you may have noticed that the candy’s chocolate coating is remarkably uniform. Inspired by this observation, a group of engineers have investigated how viscous fluids poured over a curved surface flow and solidify; their findings were published this week.
Rather than heated chocolate, the group used polymer-filled fluids that cure and harden over time. Interestingly, they found that the final shell is quite uniform and that its thickness does not depend on the pouring technique. Instead, they can predict the final shell thickness based on the radius of the mold and the rheological properties of the fluid–specifically its density, viscosity, and curing time. The reason for this is that the time it takes for the fluid to drain and coat the mold is much shorter than the time it takes for the polymer to cure. As a result, the amount of fluid that sticks to the mold depends on geometry and fluid properties – not how the fluid was poured.
Amateur confectioners rejoice: pouring uniform chocolate coatings may be easier than you thought! (Image credit: MIT News, video; research credit: A. Lee et al.)