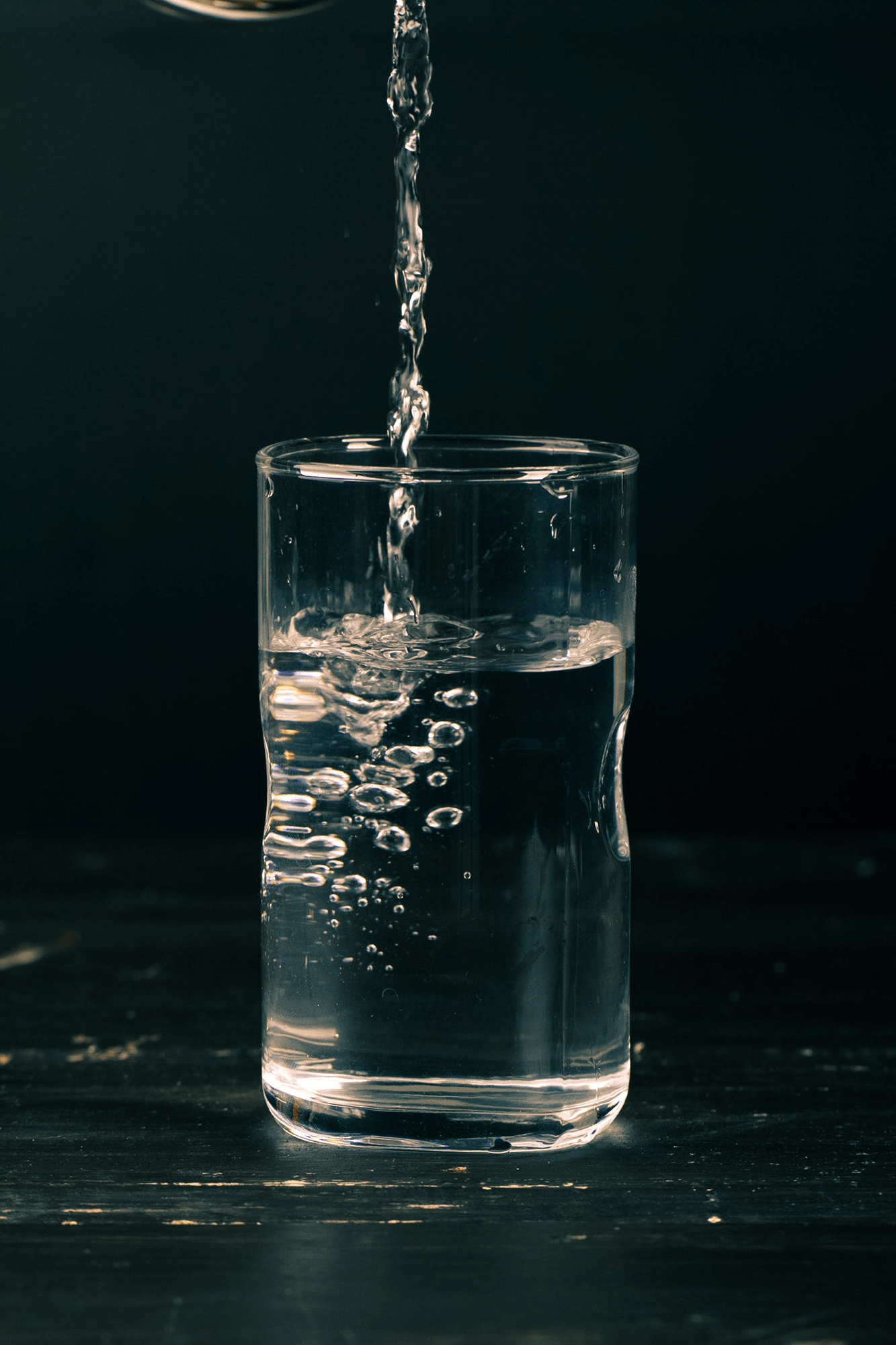If you’ve ever made ice in a freezer, you’ve probably noticed the streaks of frozen bubbles inside the ice. In its liquid state, water is good at dissolving various gases — like the carbon dioxide in sparkling water. During freezing, though, those gases cannot remain in solution; the water simply doesn’t have space between its crystalline ice lattice for non-water molecules. So the gases are forced out of solution, where they form bubbles. The final shape of the frozen bubble depends on the interplay between the speed of a bubble’s growth and how quickly the ice freezes. Here, the researchers used polarized light to outline the bubbles in color, highlighting the wide array of possible shapes. (Image credit: J. Meijer and D. Lohse; via GoSM)
Tag: bubbles

Lasing Bubbles
The thin shells of bubbles interact with light in fascinating ways; that is, of course, the source of their brilliant colors. In this recent study, researchers discovered that bubbles can serve as tunable lasers. A laser has three major components: an energy source, an optical resonator, and a gain medium that amplifies light in the resonator. For bubble lasers, an external pump laser provides energy and the bubble’s thin shell acts as a resonator. Fluorescent dye in the bubble serves as the gain medium.
Once formed, the bubble lasers are incredibly sensitive to electric fields and pressure changes, making them excellent sensors. For added stability, the team is using smectic liquid crystal bubbles, which, unlike soap bubbles, don’t evaporate easily. (Video, image, and research credit: Z. Korenjak and M. Humar; via APS Physics)

The Sound of Bubbles
Every day I stand in front of my refrigerator and listen to the water dispenser pouring water into my glass. The skinny, fast-moving jet of water plunges into the pool, creating a flurry of bubbles. Those bubbles come from air the water jet pulls in with it, and the sound the water makes (minus the fridge’s noises) comes from those bubbles. A short, laminar jet will make fewer bubbles and, therefore, be quieter than a a jet that falls farther before hitting the water.
The reason? That tall jet falls for long enough that its walls start to wobble or even break up completely into separate droplets. Compared to a smooth jet, these wobbly or broken-up jets pull in more air and create more bubbles. That makes them louder. Researchers even suggest that listening to these bubbles can give a noninvasive method for finding how much fresh oxygen is in the water. (Image credit: R. Piedra; research credit: M. Boudina et al.; via APS Physics)

Viscoelasticity and Bubbles
Bursting bubbles enhance our drinks, seed our clouds, and affect our health. Because these bubbles are so small, they’re easily affected by changes at the interface, like surfactants, Marangoni effects, or, as a recent study shows, viscoelasticity.

A bubble released in pure water pops at the surface, creating a rebounding jet and a daughter droplet. In clean water, a bubble’s burst generates a rebounding jet that shoots off one or more daughter droplets, as seen in the animation above. But when researchers added proteins that modify only the water’s surface, they found something very different. As seen below, the bursting bubble no longer generated a jet, and, instead of forming droplets, it made a single, tiny daughter bubble. The difference, they found, comes from the added viscoelasticity of the surface. The long protein molecules resist getting stretched, which damps out the tiny waves that surface tension usually produces on the collapsing bubble cavity. (Image and research credit: B. Ji et al.; submission by Jie F.)

When the surface of water is viscoelastic, a bursting bubble creates no jet and a daughter bubble instead of a drop. 
Dancing Peanuts
Bartenders in Argentina sometimes entertain patrons by tossing a few peanuts into their beer. Initially, the peanuts sink, but after a few seconds they rise, wreathed in bubbles. Once on the surface, they roll, causing the bubbles to pop, and the peanut sinks once again. The cycle repeats, sometimes for as long as a couple hours.
There are a couple physical processes governing this dance. The first is bubble nucleation. Most beers are carbonated; they contain dissolved carbon dioxide gas that remains in solution while the beer is under pressure. Once poured, that storage pressure is gone and bubbles start to form in the liquid. The shape of the peanut means that bubbles form more easily on it than on the glass walls or in the liquid. And once the peanut is covered in bubbles, buoyancy comes into play. The bubbles attached to the peanut reduce its density relative to the surrounding fluid, enabling the peanut to rise up and float.
This same process is seen with other objects in carbonated fluids, too, such as blueberries in beer and lemon seeds in carbonated water. But it’s also reflected elsewhere in nature. For example, magnetite crystals are thought to float in magma due to a similar nucleation of dissolved gases on their surface. (Image and research credit: L. Pereira et al.; via APS Physics)

Bubble Trails – Straight or Wonky?
Watch the bubbles rising in a glass of champagne and you’ll see them form tiny straight lines, with each bubble following its predecessor. But in a carbonated soda, the bubbles rise all over the place, each following its own zig-zaggy line. Why the difference? A recent study points out the culprits: bubble size and surfactants.

As bubble size increases from left to right, the bubble trail straightens. Looking at a variety of beverage scenarios, researchers found that both a bubble’s size and its surfactant concentration affected what sort of path it followed. For clean (surfactant-free) bubbles, small bubbles take a winding path, but bigger ones move in a straight line. Simulations show that bubbles can only form a straight path if they produce enough vorticity on their surface. Small bubbles just can’t deform enough to do that.

For bubbles of the same size, increasing the surfactants on the bubbles straightens their path. When surfactants get added, though, the story changes. For bubbles of a set size, adding surfactants made their paths straighter. This was due, the team found, to a bump in vorticity provided by the stabilizing effect of the surfactants. Champagne, they concluded, has straight bubble paths despite its tiny bubbles because of the drink’s high number of flavorful surfactants. (Image credit: top – D. Cook, experiments – O. Atasi et al.; research credit: O. Atasi et al.; via APS Physics)

Bubble Cleaning
Removing dirt and bacteria from fruits and vegetables is a delicate job; too much force can bruise the produce and hasten spoiling. That’s why fluid mechanicians want to give the job to bubbles. Placing objects in a stream of air bubbles inside a bath is a surprisingly effective method for gently cleaning surfaces. A recent study finds that 22.5 degrees is the optimal angle for sliding bubbles to scrape a surface clean.
As the bubbles slide past the surface, they exert a shear force that scrapes away debris, just as you might use a loofah in the shower. The angle the bubble makes with the surface determines how long it’s in contact and how much force the bubble exerts. Increasing the angle makes the bubble slide faster, increasing its shear force. But above 22.5 degrees, the bubble’s buoyancy means that it spends less time pressed against the surface, which decreases its cleaning ability.
The team hopes to use their results to build a “fruit Jacuzzi” device that will direct bubble streams to gently and effectively clean fruits and vegetables in a matter of minutes. (Image and research credit: A. Hooshanginejad et al.; via APS Physics)

Long-Lived Bubbles
Without surfactants to stabilize them, bubbles don’t last long at room temperature. But adding a little heat changes the picture. When heated, the bubbles get stabilized by a thermal gradient that lifts fluid toward the bubble’s peak, where it cools and gathers. Eventually, the cold fluid grows heavy enough to sink down the side of the bubble (in either a constant stream or occasional drips); with warm fluid getting pulled up to replace it (via the Marangoni effect), the process repeats and the bubble lives on. (Video credit: S. Nath et al.; see also)

Oil-Covered Bubbles Popping
When bubbles burst, they release smaller droplets from the jet that rebounds upward. Depending on their size, these droplets can fall back down or get lofted upward on air currents that spread them far and wide. Thus, knowing what kind of bubbles produce small, fast droplets is important for understanding air pollution, climate, and even disease transmission.

The jet from a bubble of clean water is broad and slow, releasing fewer and larger drops. In a recent study, researchers compared droplets made by clean, water-only bubbles, and the ones generated from water bubbles with a thin layer of oil coating them. The clean bubbles created jets that were broad and relatively slow moving; this motion produced a few large drops that quickly fell back down.

The jet from an oil-covered bubble is skinny and fast-moving. It produces many small droplets. In contrast, the oil-slicked bubbles made a narrow, fast-moving jet that broke into many small droplets. These droplets could stay aloft for longer periods, indicating that contaminated water can produce more aerosols than clean. (Image credit: top – J. Graj, bursting – Z. Yang et al.; research credit: Z. Yang et al.; submitted by Jie F.)

“Evanescent”
Giant iridescent inflatables dot public spaces in the “Evanescent” exhibit. The “bubble-tecture” is the work of Sydney-based artistic collaboration Atelier Sisu. Conceived during the pandemic, the duo “endeavoured to communicate this feeling of transient beauty and the need to live in the moment through the idea of the bubble.” The exhibit has appeared in more than 22 cities in 12 different countries. (Image credit: Atelier Sisu; via Colossal)