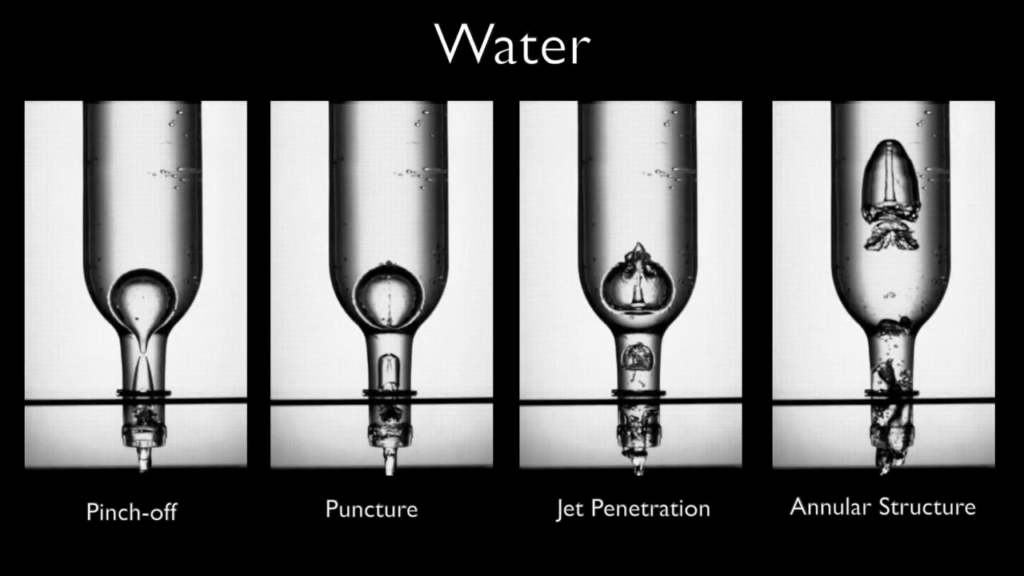
When bubbles burst at the ocean’s surface, they eject droplets that can carry high concentrations of contaminants like pollutants, viruses, and microplastics. Previous theories posited that only particles smaller than the microlayer surrounding the bubble could make their way into these drops, but new work shows otherwise.
As bubbles rise to the surface, they carry particles on their surface, collecting them to a concentration that’s even higher than the surrounding seawater. But which particles make it into the air depend on the details of what happens when the bubble pops. Previously, researchers assumed that the thin microlayer of fluid surrounding the bubble was uniform, but that turns out not to be the case. As the bubble pops, some regions of the microlayer stretch and thin, while others grow thicker. The thicker the microlayer, the larger the particles it can pull along. In their single-bubble experiments, the researchers found that 15- and 30-micrometer plastic beads — representing oceanic microplastics — appeared in high concentrations in ejected droplets.

Environmental scientists are keen to understand these mechanisms because they link our oceans and atmosphere, potentially affecting rainfall, pollution spread, and epidemiology. (Image, video, and research credit: L. Dubitsky et al.; via APS Physics)