On a surface much hotter than their boiling point, droplets can surf on a layer of their own vapor due to the Leidenfrost effect. Recent research has shown that textured surfaces like ratchets can create corrals, traps, and mazes for such droplets. Here, researchers manipulate the propulsion of Leidenfrost drops using non-parallel grooves instead. When placed between two non-parallel plates, the droplet is squeezed by side forces perpendicular to the walls, with the resultant force in the direction where the gap widens. In most states, friction forms an opposition to this squeeze, but for Leidenfrost droplets that frictional force is negligible. Instead, the squeezing from the plates launches droplets toward the wider end of the groove, allowing researchers to design repellers (top) and traps (bottom) for the fast-moving drops. (Image credits: C. Luo et al., source)
Search results for: “water droplet”

Creating Clouds
Despite their ubiquity and importance, we know surprisingly little about how clouds form. The broad strokes of the process are known, but the details remain somewhat fuzzy. One challenge is understanding how nucleation – the formation of droplets that become clouds or rain – works. A recent laboratory experiment in an analog cloud chamber suggests that falling rain drops may help spawn more rain drops.
The experiment takes place in a chamber filled with sulfur hexafluoride and helium. The former acts like water in our atmosphere, appearing in both liquid and vapor forms, while the latter takes the place of dry components of our atmosphere, like nitrogen. The bottom of the chamber is heated, forming a liquid layer of sulfur hexafluoride, seen at the bottom of the animation above. The top of the chamber is cooled, encouraging sulfur hexafluoride vapor to condense and form droplets that fall like rain. A top view of the same apparatus during a different experiment is shown in this previous post.
When droplets fall through the chamber, their wakes mix cold vapor from near the drop with warmer, ambient vapor. This changes the temperature and saturation conditions nearby and kicks off the formation of microdroplets. These are the cloud of tiny black dots seen above. Under the right conditions, these microdroplets grow swiftly as more vapor condenses onto them. In time, they grow heavy enough to fall as rain drops of their own. (Image credits: P. Prabhakaran et al.; via APS Physics; submitted by Kam-Yung Soh)

Fluid Black Holes
Fluid systems can sometimes serve as analogs for other physical phenomena. For example, bouncing droplets can recreate quantum effects and a hydraulic jump can act like a white hole. In this work, a bathtub vortex serves as an analog for a rotating black hole, a system that’s extremely difficult to study under normal circumstances. In theory, the property of superradiance makes it possible for gravitational waves to extract energy from a rotating black hole, but this has not yet been observed. A recent study has, however, observed superradiance for the first time in this fluid analog.
To do this, the researchers set up a vortex draining in the center of a tank. (Water was added back at the edges to keep the depth constant.) This served as their rotating black hole. Then they generated waves from one side of the tank and observed how those waves scattered off the vortex. The pattern you see on the water surface in the top image is part of a technique used to measure the 3D surface of the water in detail, which allowed the researchers to measure incoming and scattered waves around the vortex. For superradiance to occur, scattered waves had to be more energetic after interacting with the vortex than they were before, which is exactly what the researchers found. Now that they’ve observed superradiance in the laboratory, scientists hope to probe the process in greater detail, which will hopefully help them observe it in nature as well. For more on the experimental set-up, see Sixty Symbols, Tech Insider UK, and the original paper. (Image credit: Sixty Symbols, source; research credit: T. Torres et al., pdf; via Tech Insider UK)

Chinese Spouting Bowl Physics
In their newest video, the Slow Mo Guys recreated one of my favorite effects: vibration-driven droplet ejection. For this, they use a Chinese spouting bowl, which has handles that the player rubs after partially filling the bowl with water. By rubbing, a user excites a vibrational mode in the bowl. Watch the GIFs above and you can actually see the bowl deforming steadily back and forth. This is the fundamental mode, and it’s the same kind of vibration you’d get from, say, ringing a bell.
Without a high-speed camera, the bowl’s vibration is pretty hard to see, but it’s readily apparent from the water’s behavior in the bowl. In the video, Gav and Dan comment that the ripples (actually Faraday waves) on the water always start from the same four spots. That’s a direct result of the bowl’s movement; we see the waves starting from the points where the bowl is moving the most, the antinodes. In theory, at least, you could see different generation points if you manage to excite one of the bowl’s higher harmonics. The best part, of course, is that, once the vibration has reached a high enough amplitude, the droplets spontaneously start jumping from the water surface! (Video and image credits: The Slow Mo Guys; submitted by effyeah-artandfilm)

The Elastic Leidenfrost Effect
Drop some hydrogel beads in a hot frying pan and they’ll bounce, hiss, and screech. Normally, if you drop a ball, it bounces to ever smaller heights until it comes to rest. In contrast, on a hot surface the hydrogel can bounce to a steady height for minutes at a time, raising a question: where does it get the energy for its incessant bounce?
Upon close examination of the impact, researchers found the hydrogel beads are actually slapping the surface over and over on each bounce. The frequency of the slapping exactly matches that of the audible screech, so what you’re actually hearing is this bounce-slap. Now what causes the slapping?
Contact with the hot surface vaporizes some of the water inside the hydrogel. If it were a droplet, this vapor would form a thin, almost frictionless layer the droplet could glide on; that’s the classic Leidenfrost effect. Here the shell of the bead prevents that until the pressure really builds up. When the pressure gets high enough, the vapor finally escapes, opening up a gap. As the gap reaches its largest point, the bead rebounds elastically, bringing it back in contact with the surface and starting the process again. Each of these cycles acts like a tiny engine, harvesting energy that drives the larger bounce. This elastic Leidenfrost effect may be particularly helpful in soft robotics, providing robots with a new mechanism for movement. (Image and video credit: S. Waitukaitis et al.,arXiv)

A Drip’s Vortex
Drip food coloring into water and you can often see a torus-shaped vortex ring after the drop’s impact. That vortex rings form during droplet impact has been well known for over a century, but only recently have we begun to understand the process that leads to that vortex ring. Part of the challenge is that the vortex formation is very small and very fast, but recent work with x-ray imaging has allowed experimentalists to finally capture this event.
When a drop impacts a pool, surface tension draws some of the pool liquid up the sides of the drop. At the same time, the impact causes ripple-like capillary waves down the sides of the drop. This causes pool liquid to penetrate sharply into the drop, triggering the spirals that mark the forming vortex ring. When drops impact with even higher momentum, multiple vortex spirals can form, as seen on the lower right image. The authors observed as many as four rings during an impact. For more, check out the (open access) article. (Image and research credit: J. Lee et al., source)
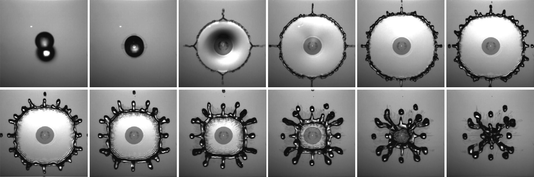
Superhydrophobic Splashes
Superhydrophobic surfaces have a complicated microscale structure that changes how water interacts with them, like the hairs on a lotus leaf or the scales of a butterfly’s wing. The photo above shows snapshots at each millisecond as a water drop hits a superhydrophobic surface covered in rows of 18 micron-tall posts. The drop hits with enough speed to drive some water into the space between posts, as shown by the dark area near the center of the splash. As the rest of the droplet spreads, four microjets form along the directions of the micropost array. Those jets remain apparent until the drop reaches its maximum radius and starts to recoil. The rectangular shape of the post array affects how the water pulls away from the surface, or depins, causing the round droplet to instead take on a square-like shape as it pulls back. (Image credit: M. Reyssat et al.)

Blue Man Group in Slow Mo
In their latest video, the Slow Mo Guys team up with the Blue Man Group for some high-speed hijinks, some of which make for great fluidsy visuals. Their first experiment involves dropping a bowling ball on gelatin. The gelatin goes through some massive deformation but comes out remarkably unscathed. Gelatin is what is known as a colloid and essentially consists of water trapped in a matrix of protein molecules. This gives it both solid and liquid-like properties, which means that the energy the bowling ball’s impact imparts can be dissipated through liquid-like waves ricocheting through the gelatin before the elasticity of the protein matrix allows it to reform in its original shape.
The video ends with buckets of paint flung at Dan. The paints form beautiful splash sheets that expand and thin until surface tension can no longer hold them together. Holes form in the sheet and eat outward until the paint forms thin ligaments and catenaries. As those continue to stretch, surface tension drives the paint to break into droplets, though that break-up may be countered to some extent by any viscoelastic properties of the paint. (Image and video credit: The Slow Mo Guys + Blue Man Group, source)

Spreading Bubbles Help Nature’s Scuba Divers
How liquid droplets spread on solid surfaces is pretty well understood, but researchers have looked less at the related problem of how a gas spreads. In a recent paper, scientists have examined the spreading dynamics of bubbles impacting an immersed solid. As the bubble contacts the surface, it quickly squeezes out water trapped between the bubble and the gas layer trapped at the solid surface. The bubble squishes as surface tension tries to flatten the liquid-gas interface. Buoyancy also helps flatten the bubble. The spreading is remarkably fast, taking only about 10 milliseconds. That’s good news for the many insects who use trapped air bubbles like these to breathe underwater. Check out the video below to learn about some of these natural scuba divers. (Image credit: H. de Maleprade et al., source; video credit: Deep Look)

How Rainfall Can Spread Pathogens

Rainfall may provide a mechanism for soil bacteria to spread. A new study examines how raindrops hitting infected soil can eject bacteria into the air. When drops fall at the rate of a light rainfall, they form tiny bubbles after impact (upper left). Those microbubbles rise to the top of the water and burst, sending extremely tiny droplets – or aerosols – spraying up into the air (upper right). Soil bacteria can hitch a ride on these aerosols, staying alive for up to an hour while the wind transports them to fresh, new soil. The researchers found that the most aerosols were produced when soil temperature was about 86 degrees Fahrenheit (30 degrees Celsius) – the temperature of tropical soils. Depending on the conditions, a single raindrop could aerosolize anything from zero to several thousands of soil bacteria. (Image and research credit: Y. Joung et al.; video credit: MIT News)
















