Soap bubbles are ephemeral creations. The slightest prick will send them tearing apart in the blink of an eye. It may come as a surprise, therefore, that dropping a water droplet through a bubble will not break it. Instead, the bubble will heal itself using the Marangoni effect. In a soap bubble, the soap molecules act as a surfactant, lowering the surface tension of the water and allowing the fragile structure to hold together. When the water drop impacts the bubble, the local surface tension increases because of the relative lack of soap molecules. This increase in surface tension pulls at the rest of the bubble, drawing more soap molecules toward the point of contact. The effect evens out surface tension across the surface and stabilizes the bubble. You can test the effect at home, too. If you wet your finger, you can poke a soap bubble without popping it. (Video credit: G. Mitchell; via io9)
Search results for: “water droplet”
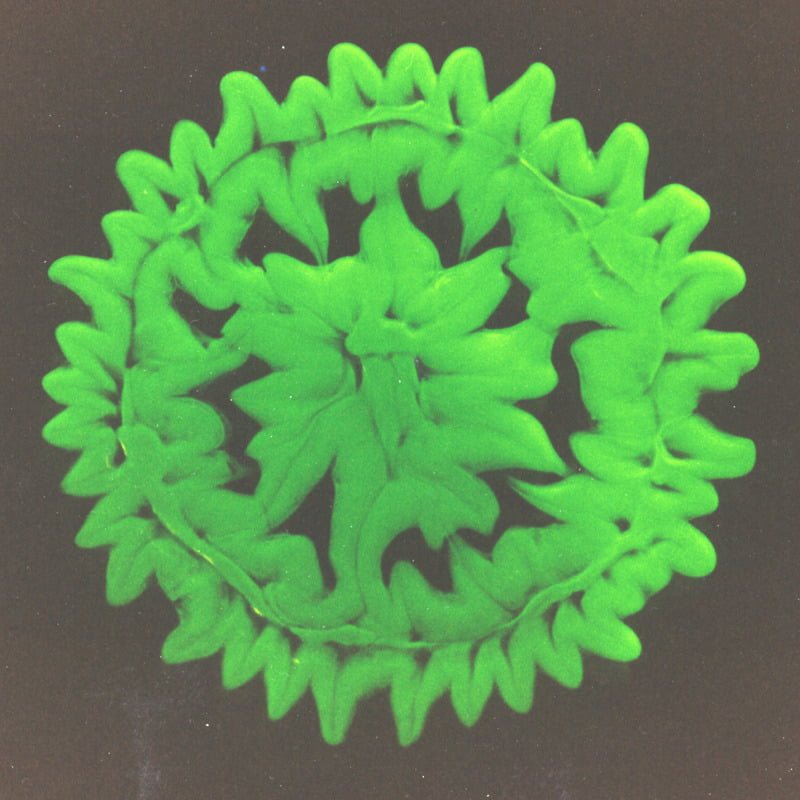
Distorted Rings
The Marangoni effect is generated by variations in surface tension at an interface. Such variations can be temperature-driven, concentration-driven, or simply due to the mixing between fluids of differing surface tensions as is the case here. The pattern in the image above formed after a dyed water droplet impacted a layer of glycerin. The initial impact of the drop formed an inner circle and outer ring. This image is from 30 seconds or so after impact, after the Marangoni instability has taken over. The higher surface tension of the water pulls the glycerin toward it, resulting in a flower-like pattern. (Photo credit: E. Tan and S. Thoroddsen)

Pointed Drops
When water droplets sit on a cold substrate, they freeze into a shape with a pointed tip. At first glance, this behavior seems very odd since surface tension usually acts to prevent such sharp protrusions. The shape is, however, a result of water’s expansion as it freezes. The droplet freezes from the substrate upward, with a concave shape to the solidification front. The angle of the point does not depend on the substrate temperature or the wetting angle between the water and surface. Instead, it turns out that this concave front shape and water’s expansion are the key factors that determine the pointed cusp’s angle, and that the final geometry of the cusp is essentially universal. (Video credit: M. Nauenberg; additional research credit: A. Marin et al.)
Vibrating on a Subwoofer
[original media no longer available]
Vibrating a liquid droplet produces some awesome behavior. The video above shows a water droplet vibrating on a subwoofer at real-time speeds. The behavior and shape of the droplet shifts with the frequency of vibration, which we hear as a change in pitch. To see more clearly the shapes a particular frequency induces, check out this high-speed video of vibrating water droplets. For a given driving frequency, the droplet’s shape, or mode, is distinct and consistent. For a droplet vibrating to a song, though, there is more than one frequency driving its motion. In this case, the droplet’s shape is a superposition of the individual modes, which is just a way of saying adding the shapes together. So frequency determines the droplet’s shape. The vibration amplitude, or audible volume, affects how energetic the drop’s motion is. And the fluid’s surface tension and viscosity act as dampers to the system, controlling how quickly the drop can change shape as well as how well it holds together. (Video credit: A. Read)

Rebounding
A water droplet can rebound completely without spreading from a superhydrophobic surface. The photo above is a long exposure image showing the trajectory of such a droplet as it bounces. In the initial bounces, the droplet leaves the surface fully, following a parabolic path with each rebound. The droplet’s kinetic energy is sapped with each rebound by surface deformation and vibration, making each bounce smaller than the last. Viscosity damps the drop’s vibrations, and the droplet eventually comes to rest after twenty or so rebounds. (Image credit: D. Richard and D. Quere)

Holiday Fluids: Snowflakes
Just about everyone wishes for a White Christmas, but even when that happens, it’s rare to get a good look at the beauty of individual snowflakes. Alexey Kljatov’s macro photography of snowflakes is simply stunning and highlights the incredible variety of forms snowflakes take. A snowflake forms when a water droplet freezes onto dust or other particles and grows as more water vapor freezes onto the initial crystal. The symmetry of the snowflakes, as with any crystal, comes from the internal order of its water molecules. The shape and features that form vary due to the local temperature and humidity level while vapor is freezing onto the crystal. Check out this handy graph showing which shapes form for various situations. Since snowflakes can encounter wildly different conditions on their path to the ground, it’s rare or next-to-impossible to find any two alike. Join us all this week at FYFD as we look at holiday-themed fluid dynamics. (Photo credit: A. Kljatov)

Explosive Boiling
A superheated liquid can reach temperatures higher than its boiling point without actually boiling – similar to how liquids can be supercooled below their freezing point without solidifying. The photo sequence above shows how explosive the boiling of a superheated water droplet submersed in sunflower oil can be. Image (a) in the lower left shows the superheated droplet resting on the bottom of its container. Then droplet vaporizes explosively in (b), expanding dramatically. The bubble overexpands and and begins to oscillate around its equilibrium radius. This triggers a Rayleigh-Taylor instability in the bubble’s interface, creating the large lobes in © and enlarged in the upper image. Finally, the bubble fragments in (d). See the original paper for more on superheated droplet boiling. (Image credit: M. A. J. van Limbeek et al.; via @AIP_Publishing)

Fluids Round-up – 5 October 2013
This is the last week that my IndieGoGo project is open for donations. All money above and beyond what is needed for the conference will go toward FYFD-produced videos. Also, donors can get some awesome FYFD stickers.
As a reminder, those looking for more fluids–in video, textbook, or other form–can always check out my resources page. And if you know about great links that aren’t on there, let me know so that I can add them. On to the round-up!
- Popular Science has look at what it was like to fly on the Concorde, the only supersonic commercial airliner ever flown.
- For the cyclists and CFD folks out there, Zipp has put out a new video discussing their Firecrest wheels’ aerodynamics.
- io9 explains how superhydrophobic surfaces impart a charge to water droplets and how this can be used to increase efficiency at power plants.
- BuzzFeed UK has 32 fun science GIFs, several of which are fluids-related, and several of which will look familiar to long-time readers. (via Flow Visualization on FB)
- Wired has an intriguing short on Acoustic Archives, a group that focuses on capturing the acoustic qualities of historic locations using custom-designed 3D microphones.
- Congratulations to Richard over at Flow Viz for hitting his 100th post! Here’s to many more.
- Finally, our lead image comes from Martin Klimas. Smithsonian’s blog has a feature on his work in which he transforms songs from artists like Pink Floyd, Daft Punk, and Bach into sonic sculptures using paint on speakers. (via Flow Visualization on FB)
I had a lot of fun earlier this week giving a talk for the Texas A&M Applied Mathematics Undergraduate Seminar series. I didn’t get a chance to record it, but the slides are up here if anyone is interested.(Photo credit: M. Klimas)
Rebounding Off Dry Ice
Droplet rebound is frequently associated with superhydrophobic surfaces but can also be generated by very large temperature differences. For very hot substrates, a thin layer of the drop vaporizes on contact via the Leidenfrost effect and helps a drop rebound by preventing it from wetting the surface. This video shows almost the opposite: a water droplet hitting solid carbon dioxide (-79 degrees C). Upon contact, the solid carbon dioxide sublimates, creating a thin layer of gas that separates the droplet from the surface. You can also see the vortex ring that accompanies the drop’s impact. Water vapor near the carbon dioxide surface has condensed into tiny airborne droplets that act as tracer particles that reveal the vortex’s formation and the rebounding droplet’s wake. (Video credit: C. Antonini et al.; Research paper)

Hydrophobia
Hydrophobic literally means water-fearing, and, once a surface is treated with a hydrophobic coating, the effect on water droplets is stark. The tendency of the non-polar hydrophobic molecules to repel the polar water molecules leads to high contact angles – which make the droplets almost spherical as they glide along the surface. The droplets dance across the surface, colliding and bouncing and coalescing. (Video and submission credit: M. Bell)




