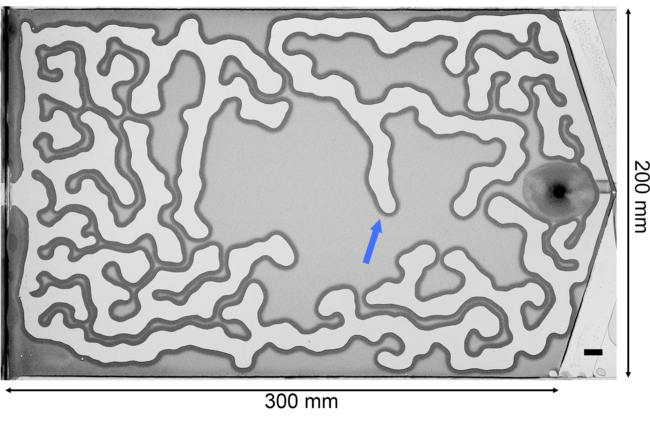
Air pushes into a thin gap filled with water and granular particles in the labyrinth-like image above. The encroaching air pushes grains like a bulldozer’s blade, building up a compacted wall. The invasion continues until the pressure of the air is countered by the combined capillary and frictional forces of the wet grains. Researchers built an analytical model that explains how these frictional fingers form and grow. Unlike Saffman-Taylor fingering patterns, which depend on long-range viscous forces, these patterns depend entirely on short-range forces from surface tension and friction. (Image and research credit: E. Flekkøy et al.)
Search results for: “viscous”

Controlling Finger Formation
When gas is injected into thin, liquid-filled gaps, the liquid-gas interface can destabilize, forming distinctive finger-like shapes. In laboratories, this mechanism is typically investigated in the gap between two transparent plates, a setup known as a Hele-Shaw cell. In the past, researchers looking to control the instability have explored how surface tension, viscosity, and the elasticity of the gap itself affect the flows. But a new set of studies look at the compressibility of the gas being injected.
The team found that viscous fingers formed later the higher the gas’s compressibility. That provides a potential control knob for people trying to exploit the mechanism, especially geologists. For geologists trying to extract oil, viscous fingering is detrimental, but, on the flip side, viscous fingers are desirable when injecting carbon dioxide for sequestration. With these results, users can tweak their injection characteristics to match their goals. (Image credit: C. Cuttle et al.; research credit: C. Cuttle et al. and L. Morrow et al.; via APS Physics)

Granular Gaps
Push air into a gap filled with a viscous fluid, and you’ll get the branching, dendritic pattern of a Saffman-Taylor instability. Here, researchers use a similar set-up: injection into a narrow gap between transparent planes to explore something quite different. In this experiment, the gap was initially filled with a mixture of air and tiny hydrophobic glass beads. When the team injected a viscous mixture of water and glycerol, new patterns emerged. At low injection rates, a single finger structure formed. But at high injection rates, a whole spoke-like pattern formed. (Image and research credit: D. Zhang et al.; via Physics Today)

Imitating a Cough
Coughing and sneezing create violent air flows in and around our bodies. As that fast air rushes over mucus layers in our lungs, throat, and sinuses, the resulting flow breaks up the mucus into droplets. To explore the details of that process, researchers built a “cough machine” that sends a rush of air over a thin film of water mixed with glycerol. The setup allows them to observe the physics in a way that’s nearly impossible in a human cough or sneeze.

Imitating a cough: high-speed video shows how a thin film made of water and glycerol breaks down in a strong airflow. Parts of the film inflate into hollow bags that form thinner weak spots. When the film breaks in those places, it forms rims and ligaments that create a spray of droplets. As seen above, air flowing past shears the viscous fluid, stretching it out. The leading edge of the film destabilizes and breaks into large drops, but it’s what comes next that really gets things going. Areas of the film inflate to form hollow bags. When sections of the bag thin to about 1 micron, the film ruptures and the bags burst. This triggers a cascade of instabilities in the film’s rim that ultimately rip the film into a spray of tiny aerosol droplets. The researchers found that, despite their tiny size, these droplets collectively carry a large volume of liquid, making them all the more important for understanding transmission of respiratory illnesses. (Image credit: top – A. Piacquadio, experiment – P. Kant et al.; research credit: P. Kant et al.)

Relax With Hummingbirds
Quick, agile, and fierce, the hummingbird is an amazing creature. Small for a bird but much larger than an insect, it’s able to hover in place and eat nectar directly from flowers. Many species use a forked tongue with curled edges that help it capture the sweet, viscous fluid. Even their distinctive sounds are fluid-influenced, coming from their wingstrokes and the fluttering of tail and wing feathers. (Image and video credit: BBC Earth)

Studying Earth’s Interior
The Earth’s interior is almost entirely inaccessible to humanity, so how do we know what it consists of? As explained in this video, our knowledge of the planet’s interior is based on measuring waves sent out by earthquakes and nuclear blasts. Both produce two kinds of waves — pressure waves (P-waves) and shear waves (S-waves) that travel through the earth and get picked up by seismometers. Scientists noticed that pressure waves travel through the center of the planet while shear waves — which get dissipated in liquids — do not. This led them to conclude that part of Earth’s interior is a liquid. The idea of a solid inner core came from observations of pressure waves scattering in a way that only made sense if they’d hit something solid. (Video and image credit: Science)
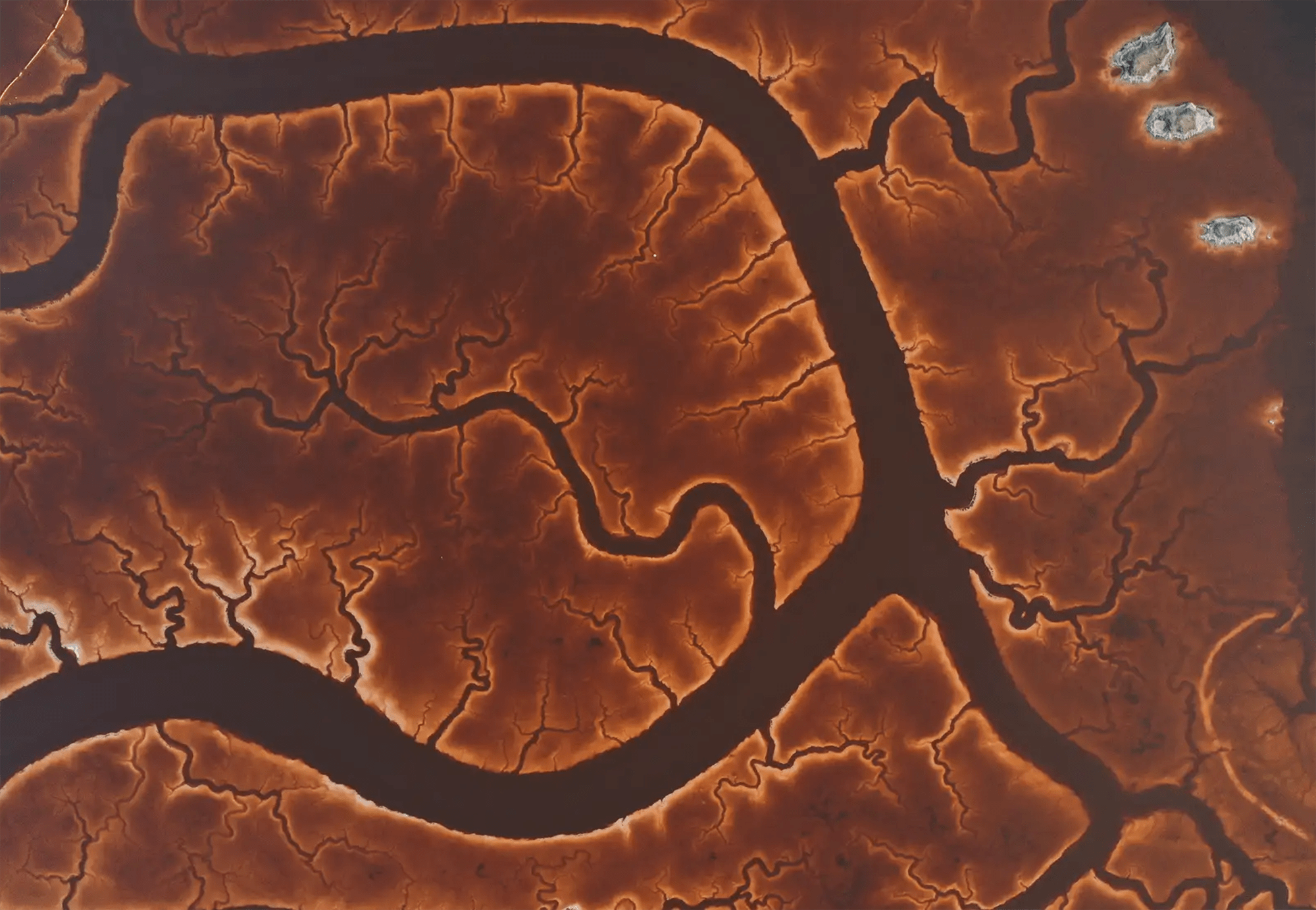
Marshy Veins
From above, the salt marshes of Alviso Marina County Park look like veins and capillaries in this photo from Tayfun Coskun. The waterways curve and branch, forming fractal patterns only apparent from the air. Although the mechanisms that form these dendritic patterns vary, they are very common in fluids, appearing over and over at many scales. (Image credit: T. Coskun; via Gizmodo)

Polymers and Fluid Sheets
Even adding a small amount of polymers to a fluid can drastically change its behavior. Often polymer-doped fluids act more like soft solids, able to hold their shape like your toothpaste does when squeezed onto your toothpaste. Under a little stress, though, the fluids still flow; that’s why your toothpaste gets less viscous as you scrub.
To study the changes polymers make, this research team collides two jets of fluid to create a liquid sheet. Depending on the flow rate and the added polymers, the break-up pattern of the sheet changes. By observing changes in the sheet thickness and the holes that form, they can draw conclusions about what the polymers are doing. (Video credit: C. Galvin et al.)

A Toad’s Sticky Saliva
Frogs and toads shoot out their tongues to capture and envelop their prey in a fraction of a second. They owe their success in this area to two features: the squishiness of their tongues and the stickiness of their saliva. The super squishy toad tongue deforms to touch as much of the insect as possible. That shape-changing helps deliver the saliva, which is an impressively fast-acting, shear-thinning fluid. Under normal circumstances, the saliva is sticky and about as viscous as honey. But the shear from the tongue’s impact makes the saliva flow like water, spreading across the insect’s body. Then it morphs back into its viscous, sticky self, providing enough adhesive power that the insect can’t escape the toad pulling its tongue back in. (Video credit: Deep Look/KQED; research credit: A. Noel et al.)

Honeybee Feeding
Busy bees feed on millions of flowers for each kilogram of honey they produce. To gather nectar, bees use their hairy tongues, which project out of a sheath-like cover. Protraction (i.e., sticking their tongue out) is relatively fast because all the hairs on the tongue initially lie flat. In the nectar, those hairs flare out, creating a miniature forest that traps viscous nectar and drags it back into the bee during retraction.

Bees feed by projecting their tongues into nectar. Tongue extension is faster because the tongue’s hairs lie flat. During the slower retraction phase, the hairs flare out, trapping nectar and pulling it back into the bee. Through modeling and experiments, researchers found that the time it takes a bee to retract its tongue depends on the bee’s overall mass. Smaller bees are slower to the retract their tongues, likely to allow enough time for their shorter tongues to capture enough nectar. With bee populations on the decline, the team’s predictions may help communities select flowers with nectar concentrations that best fit their local bees’ needs. (Image credits: top – J. Szabó, bee eating – B. Wang et al.; research credit: B. Wang et al.; via APS Physics)