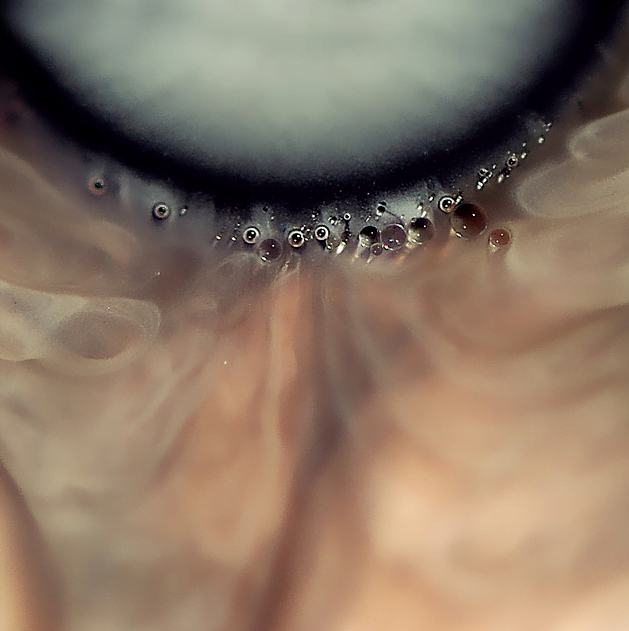
Photographers Cassandra Warner and Jeremy Floto produced the “Clourant” series of high-speed photographs of colorful liquid splashes. The artists took special care to disguise the origin of splashes, making them appear like frozen sculptures. The photos are beautiful examples of making fluid effects and instabilities. Many of them feature thin liquid sheets with thicker rims just developing ligaments. In other spots, surface tension has been wholly overcome by momentum’s effects and what was once ligaments has exploded into a spray of droplets. (Photo credit: C. Warner and J. Floto; submitted by jshoer; via Colossal)
Search results for: “surface tension”
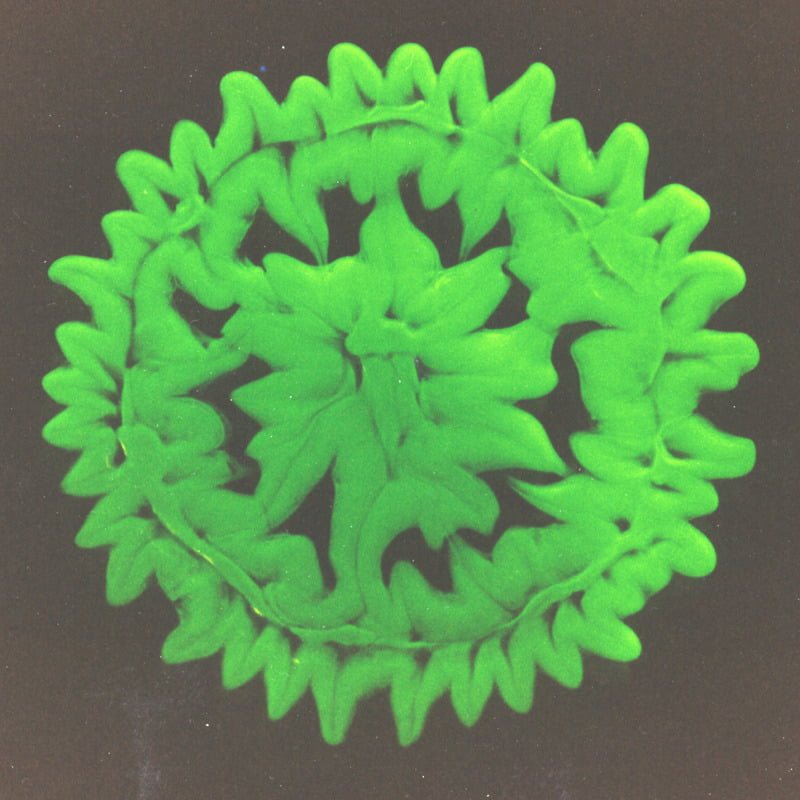
Distorted Rings
The Marangoni effect is generated by variations in surface tension at an interface. Such variations can be temperature-driven, concentration-driven, or simply due to the mixing between fluids of differing surface tensions as is the case here. The pattern in the image above formed after a dyed water droplet impacted a layer of glycerin. The initial impact of the drop formed an inner circle and outer ring. This image is from 30 seconds or so after impact, after the Marangoni instability has taken over. The higher surface tension of the water pulls the glycerin toward it, resulting in a flower-like pattern. (Photo credit: E. Tan and S. Thoroddsen)

Going Supercritical
Supercritical fluids exist at temperatures and pressures above the critical point, in a region of the phase diagram where there is no clear boundary between the liquid and gaseous state. Supercritical fluids have some of the properties of each state: they can move as freely as a gas, but they are still capable of dissolving materials like a liquid does. They also have no surface tension because there is no interface between liquid and solid. These properties make supercritical fluids very useful in industrial applications, including decaffeination and chemical deposition. Interestingly, the temperatures and pressures on Venus are so high that scientists think the atmosphere at the surface is a supercritical fluid. (Video credit: SCFED Project)
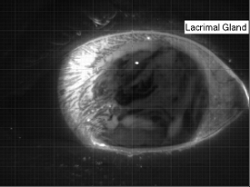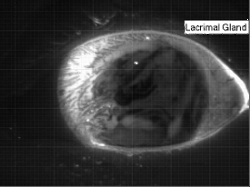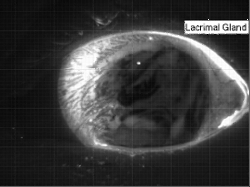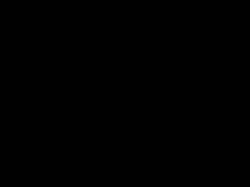
Tear Films
The human eye has a thin tear film over its surface to maintain moisture and provide a smooth optical surface. The film consists of multiple layers: a lipid layer at the air interface to decrease surface tension and delay evaporation; an aqueous middle layer; and an inner layer of hydrophilic mucins that keep the film attached to the eye. The entire film is a few microns thick, with the lipid layer estimated to be only 50-100 nm thick and the mucin layer just a few tenths of a micron. The aqueous portion of the tear film is supplied from the lacrimal gland in the corner of the eye. In the animation above, the fresh aqueous fluid is fluorescent. It gathers in the corner of the eye several seconds after a blink due to reflex tearing. The tear fluid then flows around the outer edges of the eye until the subject blinks and the fresh tear gets distributed throughout the film. (Research credit: L. Li et al.; original video)

Colliding in Microgravity
On Earth, it’s easy for the effects of surface tension and capillary action to get masked by gravity’s effects. This makes microgravity experiments, like those performed with drop towers or onboard the ISS, excellent proving grounds for exploring fluid dynamics unhindered by gravity. The video above looks at how colliding jets of liquid water behave in microgravity. At low flow rates, opposed jets form droplets that bounce off one another. Increasing the flow rate first causes the droplets to coalesce and then makes the jets themselves coalesce. Similar effects are seen in obliquely positioned jets. Perhaps the most interesting clip, though, is at the end. It shows two jets separated by a very small angle. Under Earth gravity, the jets bounce off one another before breaking up. (The jets are likely separated by a thin film of air that gets entrained along the water surface.) In microgravity, though, the jets display much greater waviness and break down much quicker. This seems to indicate a significant gravitational effect to the Plateau-Rayleigh instability that governs the jet’s breakup into droplets. (Video credit: F. Sunol and R. Gonzalez-Cinca)

Oily Foams
It is common in many industries to use oil as a defoamer to break up existing foams or prevent foams from forming. But with the right surfactants–additives that change the foam’s surface tension–it’s possible to make aqueous foams that are actually stabilized by the presence of oil. This video explores some of the ways that oil can interact with these kinds of foam, beginning with capillary action, which draws the oil up into the junctions between foam films. For more, see Piroird and Lorenceau. (Video credit and submission: K. Piroird)

Space Balls (of Water!)
The microgravity environment of space is an excellent place to investigate fluid properties. In particular, surface tension and capillary action appear more dramatic in space because gravitational effects are not around to overwhelm them. In this animation, astronaut Don Petit injects a jet of air into a large sphere of water. Some of the water’s reaction is similar to what occurs on Earth when a drop falls into a pool; the jet of air creates a cavity in the water, which quickly inverts into an outward-moving jet of water. In this case, the jet is energetic enough to eject a large droplet. Meanwhile, the momentum, or inertia, from the air jet and subsequent ejection causes a series of waves to jostle the water sphere back and forth. Surface tension is strong enough to keep the water sphere intact, and eventually surface tension and viscosity inside the sphere will damp out the oscillations. You can see the video in full here. (Image credit: Don Petit/Science off the Sphere)

Pointed Drops
When water droplets sit on a cold substrate, they freeze into a shape with a pointed tip. At first glance, this behavior seems very odd since surface tension usually acts to prevent such sharp protrusions. The shape is, however, a result of water’s expansion as it freezes. The droplet freezes from the substrate upward, with a concave shape to the solidification front. The angle of the point does not depend on the substrate temperature or the wetting angle between the water and surface. Instead, it turns out that this concave front shape and water’s expansion are the key factors that determine the pointed cusp’s angle, and that the final geometry of the cusp is essentially universal. (Video credit: M. Nauenberg; additional research credit: A. Marin et al.)
Vibrating on a Subwoofer
[original media no longer available]
Vibrating a liquid droplet produces some awesome behavior. The video above shows a water droplet vibrating on a subwoofer at real-time speeds. The behavior and shape of the droplet shifts with the frequency of vibration, which we hear as a change in pitch. To see more clearly the shapes a particular frequency induces, check out this high-speed video of vibrating water droplets. For a given driving frequency, the droplet’s shape, or mode, is distinct and consistent. For a droplet vibrating to a song, though, there is more than one frequency driving its motion. In this case, the droplet’s shape is a superposition of the individual modes, which is just a way of saying adding the shapes together. So frequency determines the droplet’s shape. The vibration amplitude, or audible volume, affects how energetic the drop’s motion is. And the fluid’s surface tension and viscosity act as dampers to the system, controlling how quickly the drop can change shape as well as how well it holds together. (Video credit: A. Read)
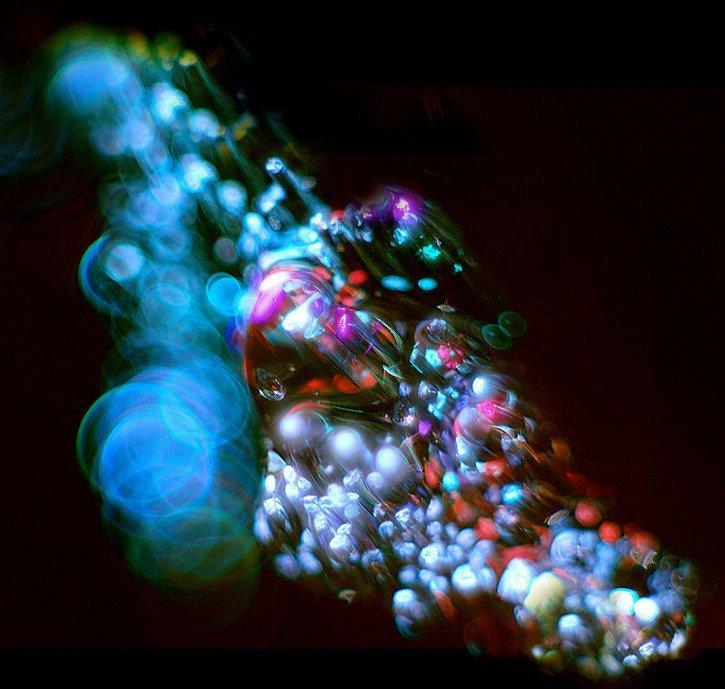
Abstract Fluids
Janet Waters’ abstract photography is full of effects created with fluid dynamics. Diffusion merges different fluids, and gradients in surface tension drive interfacial flows. Changes in density and viscosity produce fingers and streaks and all manner of forms. Be sure to check out her photostream for many more examples of fluids as art. (Photo credits: J. Waters)