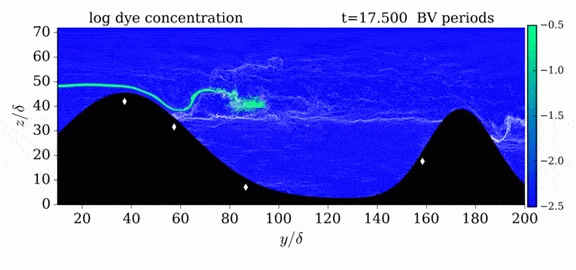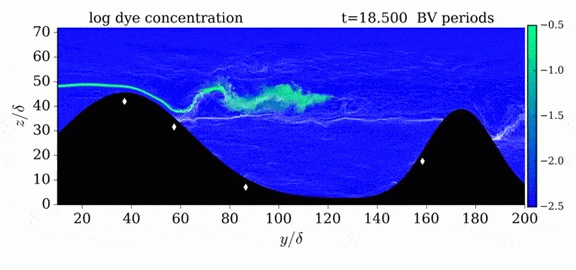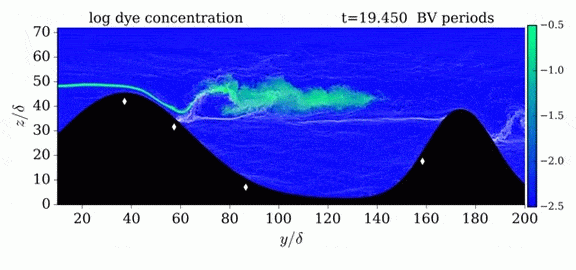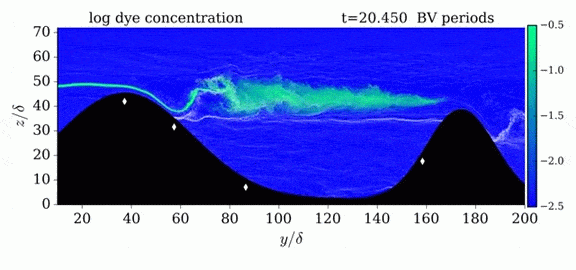
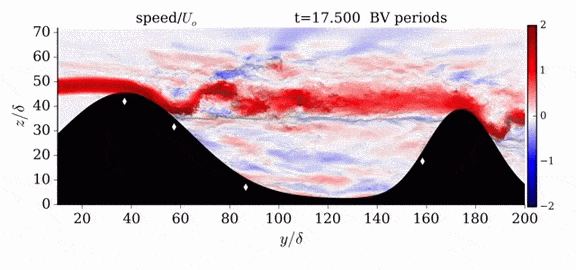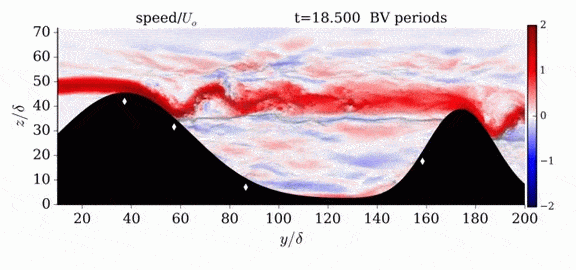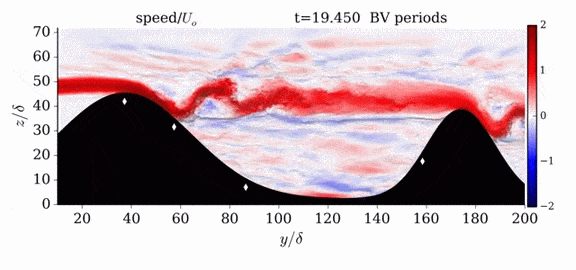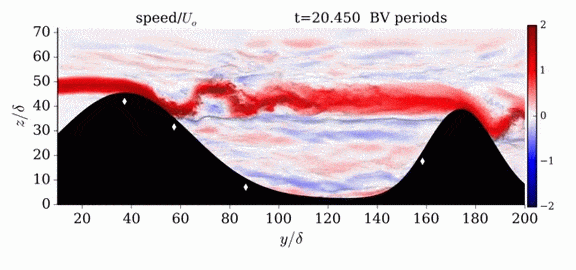
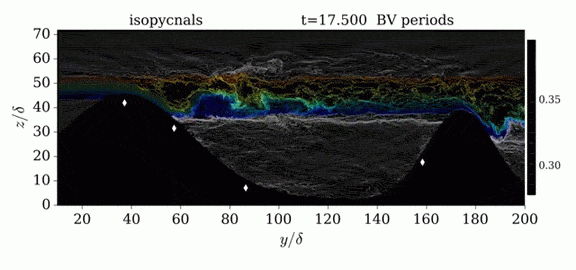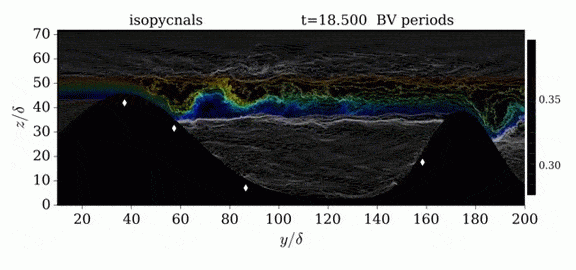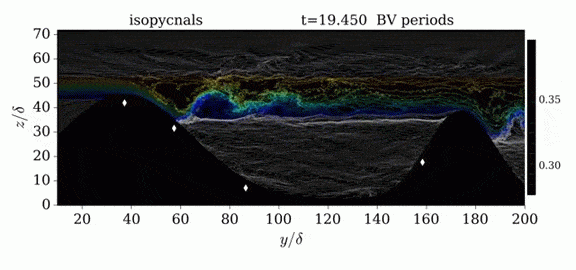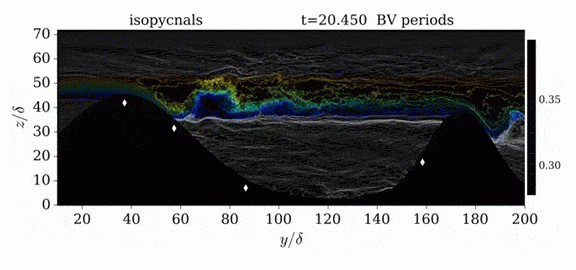
When we first learn about states of matter, we’re taught about three: solid, liquid, and gas. In a solid, atoms are held close to one another–typically, but not always, in an orderly lattice structure. In liquids and gases, atoms are free to slip, slide, bounce, and move. So what really separates a liquid from a gas?

That’s the question at the heart of this video by Steve Mould, in which he explores a weird fourth phase of matter: supercritical fluids. This phase has the diffusive properties of a gas and the solvent properties of a liquid–without really being either one.(Video and image credit: S. Mould)