Cavitation refers to the formation of destructive bubbles of vapor within a liquid. Traditionally, we think of it as occurring when the velocity in a flow becomes high enough for the pressure to drop below the local vapor pressure, causing bubbles to form. This is what we see around turbine blades and ship propellers.
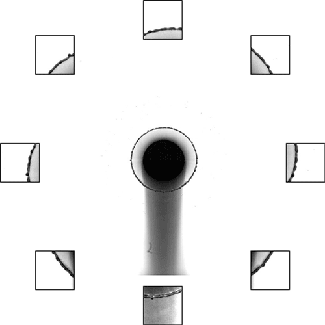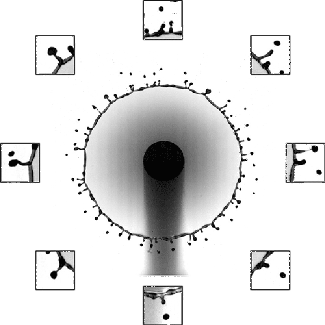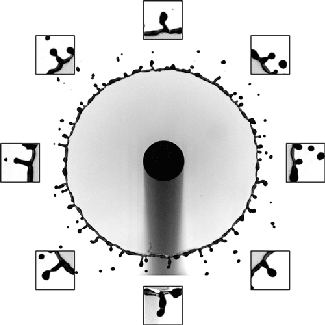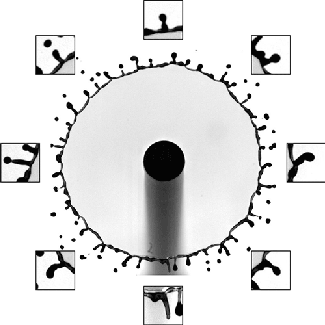
But cavitation also occurs in situations where the overall velocity is relatively low, provided there’s a sudden acceleration. That’s the situation we see above. The impact — either of a mallet off-screen or of the tube striking the floor — causes the liquid inside suddenly accelerate upward. Notice in the second image how the liquid interface moves upward as the first bubbles form.
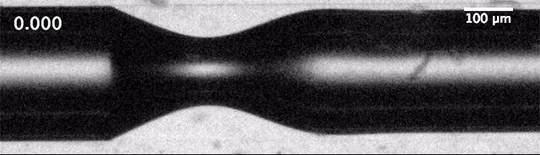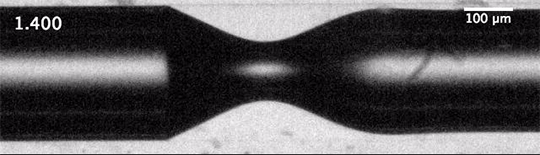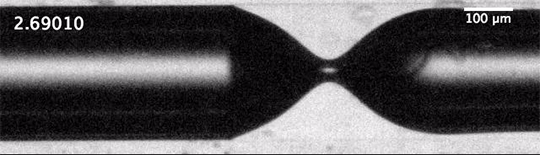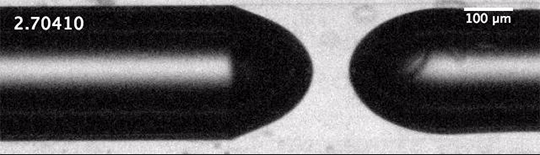
Each of these cavitation bubbles has such a low pressure that they’re basically a vacuum, and their collapse can cause shock waves that reverberate through the container, causing it to break. Check out that test tube in the last image. Notice that there’s no sign of cracking when the test tube hits the floor; in fact, the researchers demonstrate in their paper that an empty test tube dropped from the same height doesn’t break. Fractures only form after the cavitation bubbles do. (Image and research credit: Z. Pan et al.; submitted by A.J.F.)