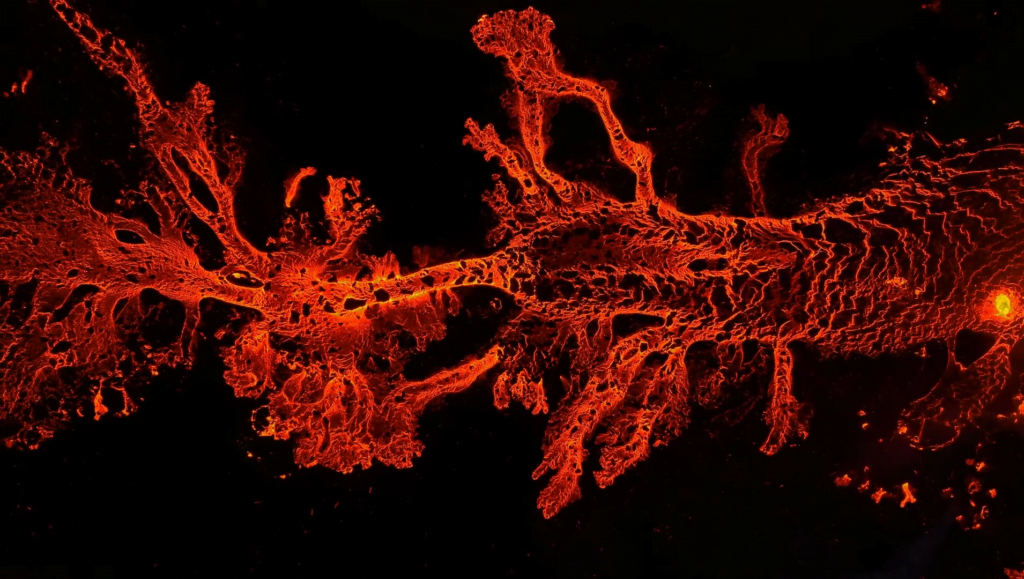
North of Iceland’s Fagradalsfjall, a new volcanic fissure opened in July 2023. This drone footage from Isak Finnbogason captures that fissure on its first night. Lava fountains jet from the earth, forming a complex, slow-moving river. The similarities between flowing lava and more common liquids like water never ceases to fascinate me. Even with the vast differences in temperature and viscosity, so much of their physics remains recognizably the same. (Image and video credit: I. Finnbogason; via Colossal)
Search results for: “liquid jet”

Polymers and Fluid Sheets
Even adding a small amount of polymers to a fluid can drastically change its behavior. Often polymer-doped fluids act more like soft solids, able to hold their shape like your toothpaste does when squeezed onto your toothpaste. Under a little stress, though, the fluids still flow; that’s why your toothpaste gets less viscous as you scrub.
To study the changes polymers make, this research team collides two jets of fluid to create a liquid sheet. Depending on the flow rate and the added polymers, the break-up pattern of the sheet changes. By observing changes in the sheet thickness and the holes that form, they can draw conclusions about what the polymers are doing. (Video credit: C. Galvin et al.)

Beneath the Cavity
When a drop falls into a pool of liquid, it creates a distinctive cavity, followed by a jet. From above the surface, this process is well-studied. But this poster offers us a glimpse of what goes on beneath the surface, using particle image velocimetry. This technique follows the paths of tiny particles in the fluid to reveal how the fluid moves.
As the cavity grows, fluid is pushed away. But the cavity’s reversal comes with a change in flow direction. The arrows now point toward the shrinking cavity — and they’re much larger, indicating a strong inward flow. It’s this convergence that creates the Worthington jet that rebounds from the surface. And, as the jet falls back, its momentum gets transferred into a vortex ring that drifts downward from the point of impact. (Image credit: R. Sharma et al.)

Founts of Enceladus
In its exploration of Saturn, Cassini discovered that the moon Enceladus is home to icy eruptions. Beneath its shell of ice, Enceladus has a global ocean of salty liquid water. The average thickness of the ice is 20 kilometers, putting the ocean seemingly out of reach — except at the moon’s southern pole, where icy plumes of ocean water jet out.
Here, where the ice is thinnest, the tidal forces Enceladus experiences from Saturn and its fellow moon Dione break through the ice. As the cracks open and close, liquid from the ocean sprays out, freezing into plumes that Cassini measured. Plans are underway for new missions that prioritize further sampling of Enceladus’ ocean. For now, we can only imagine what hides in its interior ocean. (Image credit: NASA/JPL-Caltech/SSI; for more, see M. Manga and M. Rudolph)

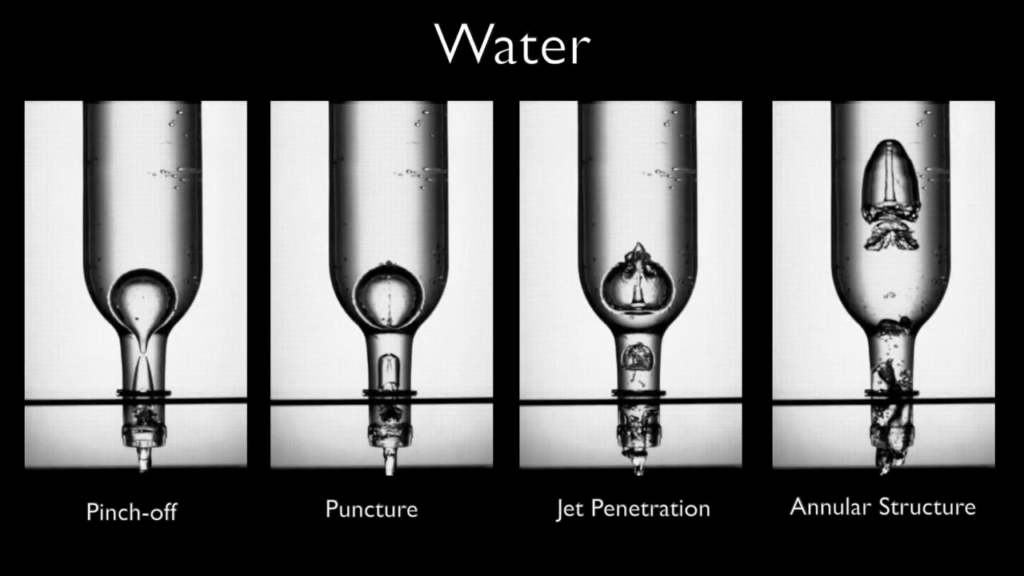
Draining a Bottle
Turn a bottle upside-down to empty it, and you’ll hear a loud glug-glug-glug as the liquid in the bottle empties and air rushes in. In this video, researchers aim a high-speed camera at the very first bubble that forms during the process. Once the bubble reaches the wider area of the bottle, it tends to pinch off in the bottle’s neck. That creates a narrow jet that pierces the bubble and flies all the way to the other side, leaving a column of liquid inside the rising bubble. Increasing the fluid’s viscosity has remarkably little effect, at least until the liquid is extremely viscous. (Image and video credit: H. Mayer et al.)

Never Break the Chain
Pour water out of a bottle, and you’ll see a jet with a shape that resembles chain links. Sometimes known as a “liquid chain,” this phenomenon occurs when water pours through a non-circular hole. It’s quite a complex behavior, as shown in this recent study of the nonlinear effect. Even so, the authors found that the amplitude and wavelength of the chain’s sections are tied directly to the shape of the opening. Current models of the effect don’t account for the viscosity of the liquid, though, so future experiments will have to explore how fluids other than water behave. (Image and research credit: D. Jordan et al.; via APS Physics; submitted by Kam-Yung Soh)

A comparison of an oscillating jet’s shape and metal chains. Each view is rotated 45 degrees from the one before. 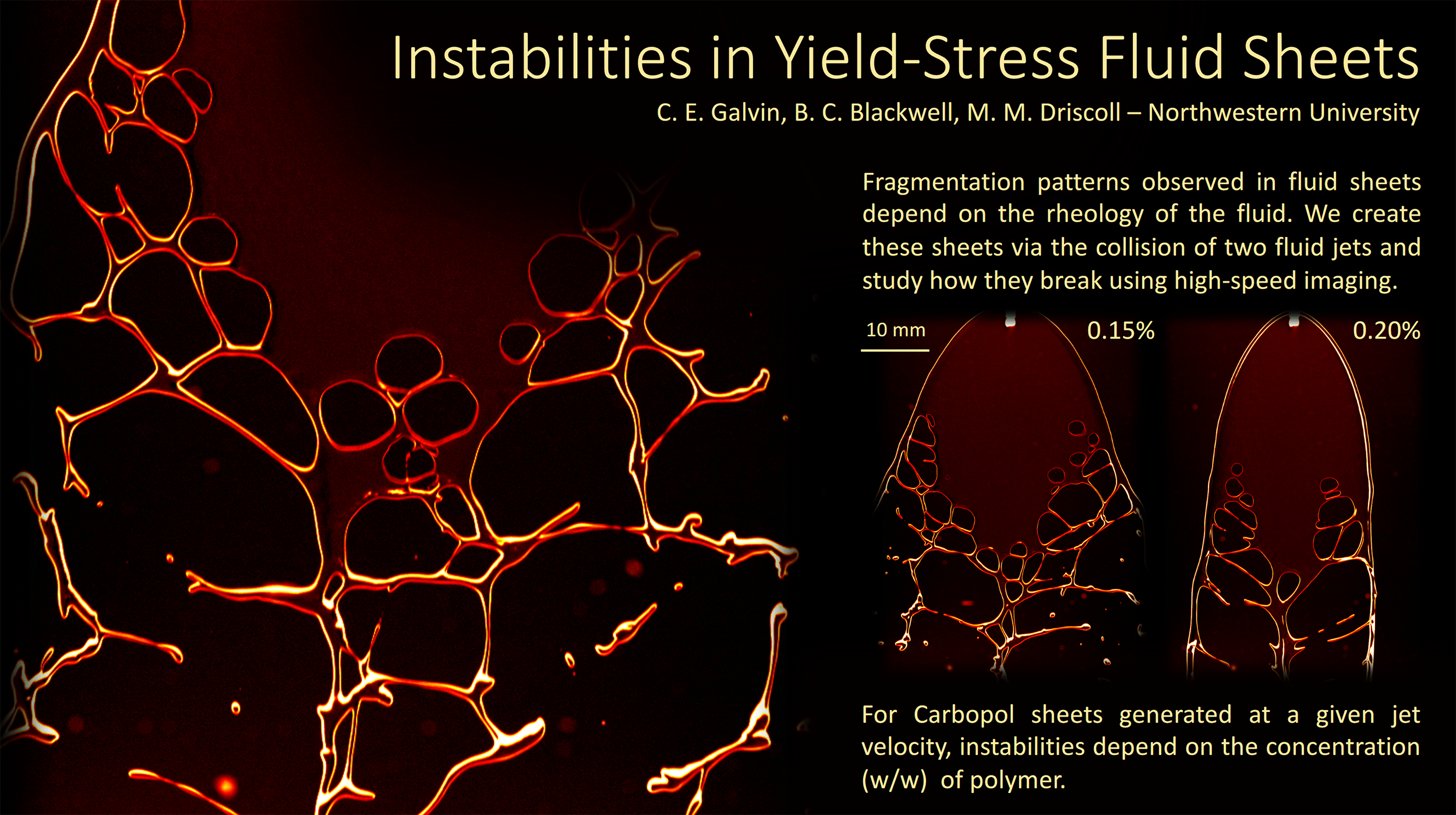
“Keeping Our Sheet Together”
When two liquid jets collide, they form a falling liquid sheet. Here researchers explore how that sheet breaks up when the liquids involved contain polymers. The intact areas of the sheet show as dark red or almost black. The edges of the sheet appear in brighter red and yellow, outlining the holes that form and grow during breakup. The type of breakup observed depends on the concentration of polymer in the liquid. (Image credit: C. Galvin et al.)

Self-Stopping Leaks
A leak can actually stop itself, as shown in this video. To demonstrate, the team used a tube pierced with a small hole. When filled, water initially shoots out the hole in a jet. The pressure driving the jet comes from the weight of the fluid sitting above the hole. As the water level drops, the pressure drops, causing the jet to sag and eventually form a rivulet that wets the side of the tube. As the water level and driving pressure continue to fall, the rivulet breaks up into discrete droplets, whose exact behavior depends on how hydrophobic the tube is. Eventually, a final droplet forms a cap over the hole and the leak stops. At this point, the flow’s driving pressure is smaller than the pressure formed by the curvature of the capping droplet. (Image and video credit: C. Tally et al.)

Fluid Chains
In this video, Steve Mould tackles a question many of us have likely wondered: just why does falling water make this chain-like shape? When pouring from a slit-like orifice, water jets take on this undulating pattern. While I have no issue with Steve’s explanation of surface tension oscillations driving the shape, I’ll quibble a little bit with the idea that this hasn’t been studied. Personally, I’d connect it to the fishbone instability, which classically occurs when two jets collide. At low flow rates, though, the colliding jets form a pattern very much like this one. And if you look just past the initial conditions at the container opening, all of these flows have thicker jet-like rims colliding. I think the flows in these videos are just a slightly messier version of the low-flow-rate fishbone. What do you think? (Video and image credit: S. Mould)

Oil in Water
In the decade since the Deepwater Horizons oil spill, scientists have been working hard to understand the intricacies of how liquid and gaseous hydrocarbons behave underwater. The high pressures, low temperatures, and varying density of the surrounding ocean water all complicate the situation.
Released hydrocarbons form a plume made up of oil drops and gas bubbles of many sizes. Large drops and bubbles rise relatively quickly due to their buoyancy, so they remain confined to a relatively small area around the leak. Smaller drops are slower to rise and can instead get picked up by ocean currents, allowing them to spread. The smallest micro-droplets of oil hardly rise at all; instead they remained trapped in the water column, where currents can move them tens to hundreds of kilometers from their point of release. (Image and research credit: M. Boufadel et al.; via AGU Eos; submitted by Kam-Yung Soh)