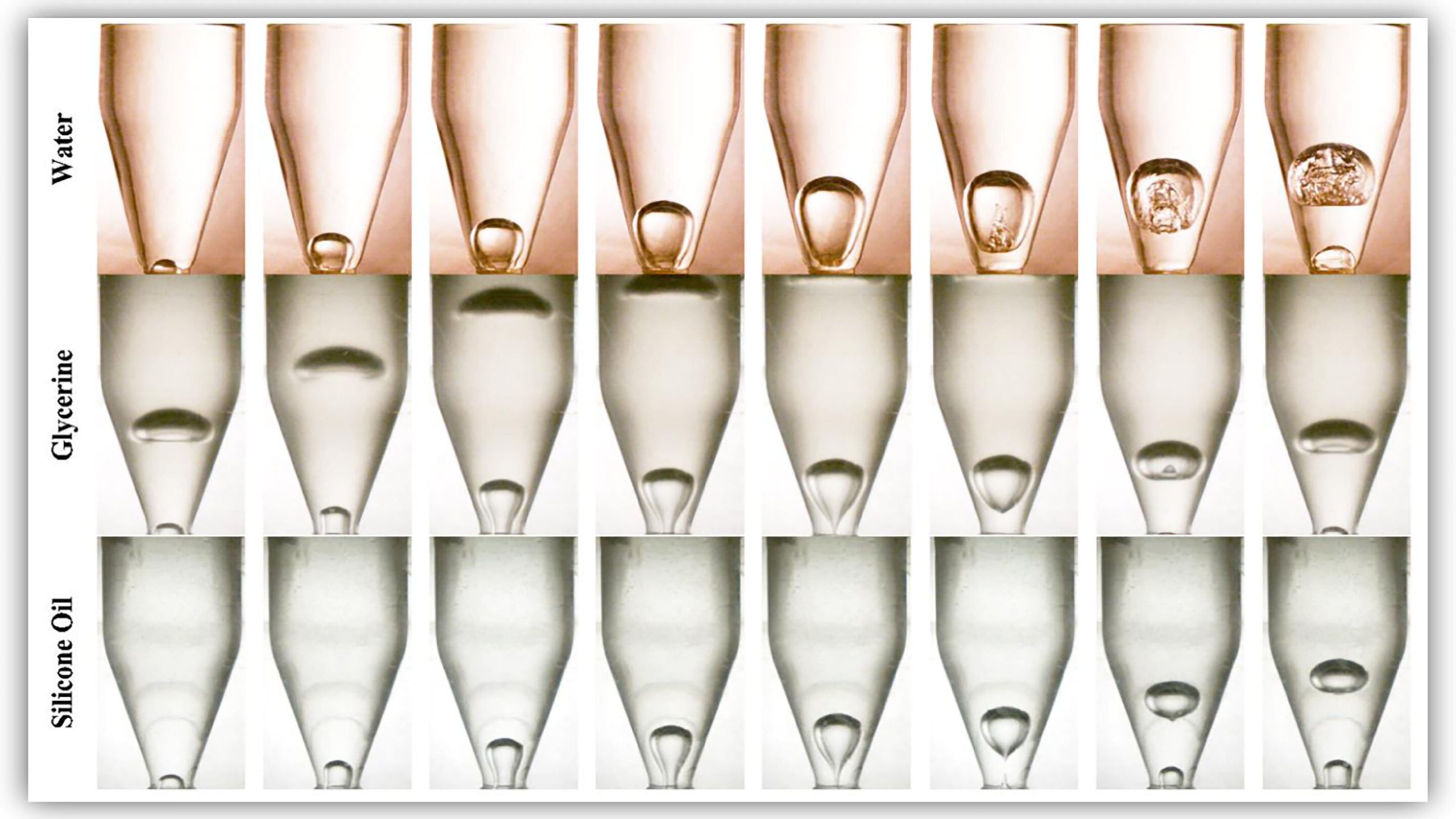
I’ve covered some odd studies in my time, but this might be the strangest: to understand how active polymers affect viscosity, researchers loaded drunk worms into a rheometer. Active polymers are long-chain molecules that, like worms, can move on their own using stored energy or by extracting energy from their surroundings. Their dynamics are tough to study, though, because individual polymers are almost impossible to observe while a suspension of them is being deformed.
Enter the humble sludge worm. Often sold as fish food, these worms — like the polymers they’re meant to imitate — are individually quite wiggly but, given their size, are far easier to observe. Researchers placed them in a custom rheometer in a solution of water and observed how the worm mass responded when sheared by a spinning top plate (Image 3). Like active polymers, the worms exhibited shear-thinning; the faster the plate spun, the lower the worms’ viscosity, likely because the additional force helps align the worms.
But how do active worms compare with passive ones? The obvious solution would be to repeat their tests with dead worms, but the researchers found a more humane method: by adding some alcohol to the water, they temporarily reduced the worms’ activity, allowing them to compare active and passive worms (Image 2). Once rinsed in water, the worms sobered up and returned to their normal activity levels.
The researchers found that both the active and passive worms exhibited shear-thinning as the force on them increased, but the shear-thinning in the active worms was not as pronounced, presumably because the movements of individual worms prevented them from aligning smoothly. (Image and research credit: A. Deblais et al.; via Gizmodo and APS Physics)