With 3D printing and other recent technologies, manufacturing options are always in flux. Here, researchers explore a method for printing a liquid inside of a liquid. Their materials are specially chosen such that the injected liquid forms an emulsion at its interface with the surrounding fluid. Once injection ends, the interface forms a wrinkly, viscoelastic skin that acts like a tube. As shown below, the tube is robust enough that it can be pumped full of yellow-dyed water without any loss of structure. (Image and research credit: P. Bazazi et al.)
Category: Research

Inhibiting Marine Lightning
Thunderstorms over the ocean have substantially less lightning than a similar storm over land. Scientists wondered whether this difference could be due to lower cloud bases over the ocean or differences in the cloud droplets’ nuclei. But a new study instead implicates coarse sea spray as the deciding factor. By tracking the full lifetime of storm systems through remote sensing, the team found that fine aerosols can increase lightning activity over both land and ocean. But adding coarse sea salt from sea spray reduced lightning by 90% regardless of fine aerosols. With sea salt in the mix, clouds seem to develop fewer but larger condensation droplets, providing less opportunity for the electrification necessary to generate lightning. (Image credit: Z. Tasi; research credit: Z. Pan et al.)

Rain-Driven Prey Capture
Pitcher plants often entice their insect victims with sweet nectar before trapping them in inescapable viscoelastic goo. But some species go even further. Nepenthes gracilis, a species native to Southeast Asia uses its leafy springboard to lure its prey. Once an ant crawls to the underside of the leaf, a falling rain drop will spell its doom. When drops hit the leaf, it deflects down and jerks up, thanks to its shape and stiffness. The motion catapults insects into the pitcher, where digestive fluids await. While we’ve seen some fast-moving plants before, this is a rare example of a plant with an externally-driven speed mechanism. With it, the pitcher plant doesn’t have to wait or expend any metabolic effort to reset for the next insect. (Image credit: GFC Collection/Alamy; research credit: A. Lenz and U. Bauer; via New Scientist)

Absorbing Sound with Moth Wings
Manmade soundproofing tends to be porous and bulky or very limited in the range of frequencies it can handle. In contrast, moths are natural absorbers of ultrasound, having evolved to avoid reflecting those frequencies back to the bats hunting them. Researchers took the structures from a moth wing and applied them to an aluminum disk to see how the coating performed. They found that the moth wing’s structures reduced sound reflection by as much as 87% at the lowest frequency tested (20kHz, still beyond human hearing.) As researchers explore how the individual structures of the wing perform, they hope to adapt the moth’s prowess to soundproof within the human range of hearing. (Image and research credit: T. Neil et al.; via Physics World)

Aligning by Bubble Array
Assembling structures from small components is often difficult. Techniques like optical tweezers are limited to very small objects, and magnetic techniques only work with certain materials. Here, researchers use acoustical forces on bubbles to move and align centimeter-sized objects.
When a single bubble oscillates in an ultrasonic field, its changing size creates pressure variations around it. When an acoustic wave scatters off one bubble and impacts another, it sets up a small attractive force between the bubbles, known as the secondary Bjerknes force. For individual bubble pairs, this force is extremely small and unable to affect much. But using arrays of bubbles — one array on a fixed object and another on a floating object — researchers amplified the attraction and showed that the resulting forces could manipulate and align their components. (Image credit: top – J. Thomas, others – R. Goyal et al.; research credit: R. Goyal et al.; via APS Physics)

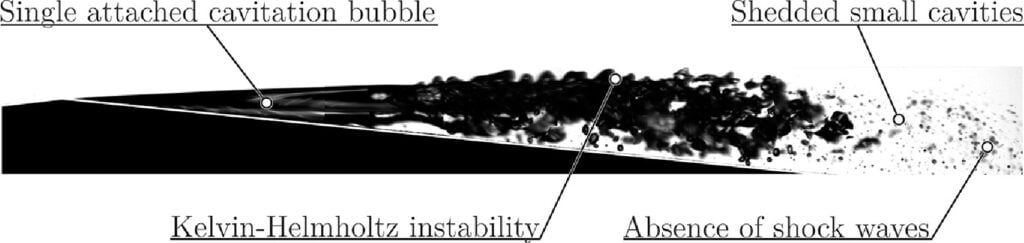
Microscale Kelvin-Helmholtz
When we think of cavitation in a flow, we often think of it occurring at a relatively large scale — on the propeller of a boat, for example. But cavitation takes place on microscales, too, including around fuel-injection nozzles. In this study, researchers investigated submillimeter-scale cavitation using a flow through a tiny Venturi tube. What they found was something we usually associate with larger scale flows: the Kelvin-Helmholtz instability.

The wavy shape of a Kelvin-Helmholtz instability forms when two layers of fluid move past one another at different speeds and the interface where they meet becomes unstable. Here, that happens along a cavitation bubble, where the bubble and the flow meet. Interestingly, at these scales, the Kelvin-Helmholtz instability seems to be the primary method of break-up, instead of shock wave interactions.
For those keeping track, we’ve now seen the Kelvin-Helmholtz instability from the quantum scale up to 160 thousand light-years. It’s hard to achieve a much wider range than that! (Image and research credit: D. Podbevšek et al.; submitted by M. Dular)

Encapsulating Drops
Sometimes a droplet needs a little protection while it’s traveling to its destination. When that’s the case, we often try to encapsulate it in a layer of material that won’t be affected by whatever environment the drop is traveling through. In this study, researchers aimed to give their drops not one but two layers of protection — in as simple a way as possible.
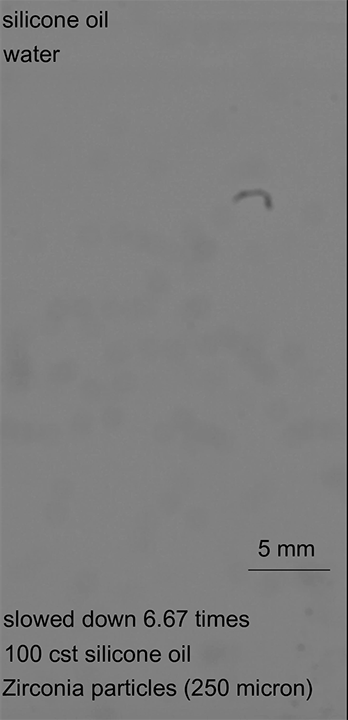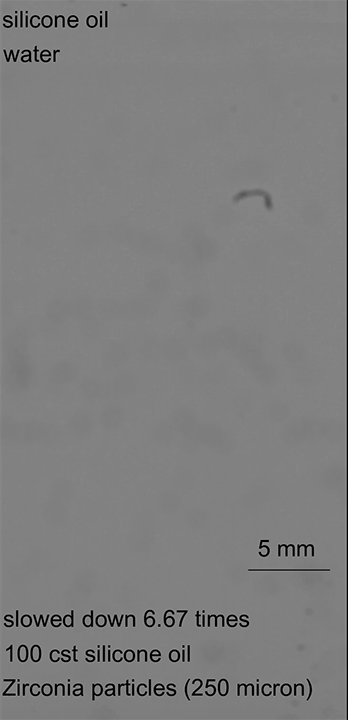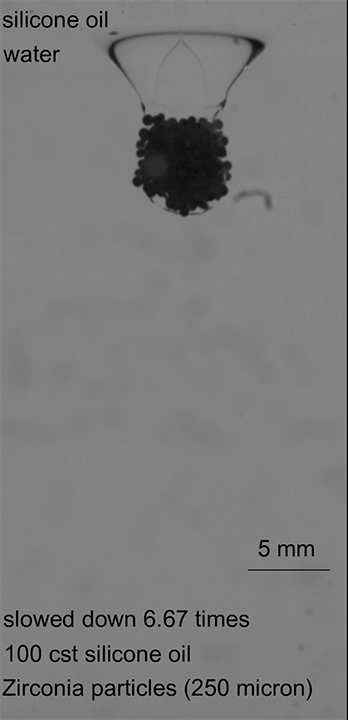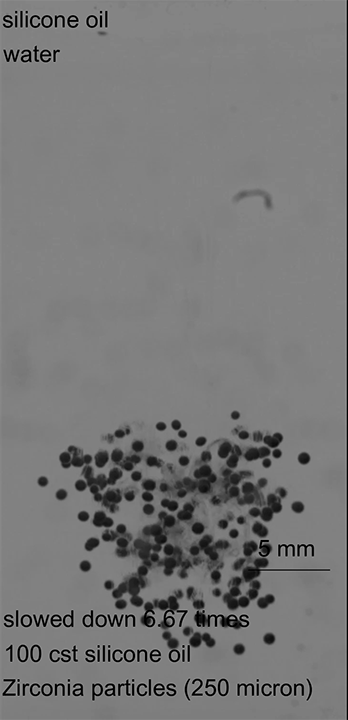
The team began with three layers of liquid. The lowest layer was water, the middle layer was an oil, and the top layer was a mixture of water and isopropyl alcohol. Next, they added glass particles that were denser than the alcohol, but less dense than the oil. This caused the particles to form a clump — a granular raft — along the interface between the alcohol and the oil (not shown). When the layer of particles became heavy enough, it began to sink into the oil, carrying some of the alcohol with them. This conglomeration formed the initial droplet of alcohol mixture encased in an armor of glass beads.
As this armored droplet sank, it approached the second interface: the oil-water interface. At this juncture, the team observed three different outcomes. When the glass particles were small or light, the armored drop would come to a rest at the oil-water interface. As the drop deformed, water would pierce the armor, causing the whole drop to rupture (Image 1).
In the second case, heavier particles caused the armored drop to sink through the oil-water interface, but a low oil viscosity meant that the oil film drained from the bottom of the drop before the drop was fully encapsulated. Once again, this let the water through and ruptured the droplet (Image 2).
In the final case, armored drops with just the right bead density and oil viscosity would sink through the oil-water interface until the oil pinched off behind the drop. This pinch-off allowed the oil to redistribute around the drop, encapsulating it in layers of both oil and particles, thereby protecting it as it continued its journey (Image 3). (Image credits: top – Girl with red hat, experiment – A. Hooshanginejad et al.; research credit: A. Hooshanginejad et al.)

Slow to Relax
Oobleck is a decidedly weird substance. Made from a dense suspension of cornstarch in water, oobleck is known for its mix of liquid-like and solid-like properties, depending on the force that’s applied. In a recent study, researchers took a look at what happens when you really push oobleck to the extreme. When the force applied to oobleck is small or slowly added, the water between cornstarch particles helps keep the particles apart and free of contact. It’s when the force is large that those particles start jamming up against each other and having friction between them, and then the oobleck suddenly acts like a solid. But what happens once that force is removed?
When the force is gone, we expect the particles to repel and for water to squeeze back into the spaces between them, breaking up the friction and allowing the oobleck to relax back to a liquid-like form. But the team found that sometimes the oobleck doesn’t relax as easily as expected; instead, it seems to retain some memory of its solid-like state, due to persisting friction between particles. (Image credit: T. Cox; research credit: J. Cho et al.)

Bubbles in Turbulence
In nature and industry, swarms of bubbles* often encounter turbulence in their surrounding fluid. To study this situation, researchers used numerical simulation to observe bubbles across a range of density, viscosity, and surface tension values relative to their surroundings. They found that density differences between the two fluids made negligible changes to the way bubbles broke or coalesced.
In contrast, viscosity played a much larger role. More viscous bubbles were less likely to deform and break, thanks to their increased rigidity. When looking at small deformations along the bubble interface, both density and viscosity had noticeable effects. With increasing bubble density, they observed more dimples on the interface; increasing the viscosity had the opposite effect, making the bubbles smoother. (Image credit: Z. Borojevic; research credit: F. Mangani et al.)
*We usually think of bubbles as air or another gas contained within a liquid. But this study’s authors use the term “bubble” more broadly to mean any coherent bits of fluid in a different surrounding fluid. Colloquially, this means their results apply to both bubbles and drops.

Rotating Waves of Grains
Rotating drums are a popular way to explore granular dynamics. Here, researchers fill a cylinder (seen below) with heavy grains and a low-viscosity fluid, then rotate the mixture about a horizontal axis. This sets up a contest between centrifugal forces and gravitational forces on the grains. At the right rotation rates, the grains form annular rings around the outside of the cylinder, where they rotate at a different speed than the fluid. This difference in speed between the two layers can trigger a Kelvin-Helmholtz instability and cause waves along the interface between the grains and the fluid, as seen in the examples above. (Image and research credit: V. Dyakova and D. Polezhaev; top image adapted by N. Sharp)