Marsh grass shrimp, despite their small size, are zippy swimmers. They move using a series of closely-spaced legs that stroke asynchronously. Researchers found that the flexibility and stiffness of the legs are critical for the shrimp’s efficiency. During the power stroke, the shrimp’s leg is held stiff, maximizing the force it’s able to transfer to the water. But during the forward-moving recovery stroke, the shrimp bends its legs almost horizontal and presses both legs in the pair together tightly. This action minimizes the area of the leg pair and reduces the drag they cause as they move into position for the next stroke. (Image, video, and research credit: N. Tack et al.; via Ars Technica; submitted by Kam-Yung Soh)
Category: Research

Toilet Plumes
Toilet flushes are gross. We’ve seen it before, though not in the same detail as this study. Here, researchers illuminate the spray from the flush of a typical commercial toilet, like those found in many public restrooms. They found that flushing generates a plume of droplets that reaches 1.5 meters in under 8 seconds, producing many thousands of droplets across a range of sizes.
The experiments were conducted in a ventilated lab space, and the flushes involved only clean water — no fecal matter or toilet paper — so they don’t perfectly mimic the confines of a public toilet stall. But the implications are still pretty gross. Without a lid to contain the flush’s spray, these energetic toilets are spraying droplets capable of carrying COVID, influenza, and other nastiness all over our bathrooms. (Image and research credit: J. Crimaldi et al.; via Gizmodo)

Bending in Bubbles
Inside a cavity with a square cross-section, bubbles form an array. The shapes of their edges are determined by surface tension and capillarity (lower half of center image). Adding an elastic ribbon into the bubbles (upper half of center image) means that the bubbles’ shapes are determined by a competition between the elasticity of the ribbon and the capillarity of the fluid. Researchers found that they could tune the rigidity of the ribbon to dictate the shape of the bubble array, or, conversely, they could use the bubbles to set the shape of a UV-curable ribbon. (Image and research credit: M. Jouanlanne et al., see also)

Icicles and Impurities
In nature, icicles often form horizontal ripples along their outer surface. Researchers found that these shapes only form when impurities are present in the water forming icicles; icicles made from pure water are smooth. Now researchers are uncovering more details of the ripple formation process, though the underlying mechanism remains unknown.

Icicle using sodium fluorescein as an impurity. a) A vertical cross-section through the icicle shows chevron-like inclusions where impurities are concentrated. b) A similar icicle using salt as the impurity shows a similar pattern. c) A horizontal cross-section through the icicle reveals tree-like rings of concentrated impurities. Researchers first grew wavy icicles, then melted through them to reveal cross-sections of the icicle. They found chevron-like patterns within the ice, corresponding to areas with higher concentrations of impurities. The team think these chevrons record the process by which flowing water accumulates on the surface of the icicle prior to freezing. (Image credit: top – M. Shturma, cross-sections – J. Ladan and S. Morris; research credit: J. Ladan and S. Morris; via APS Physics)

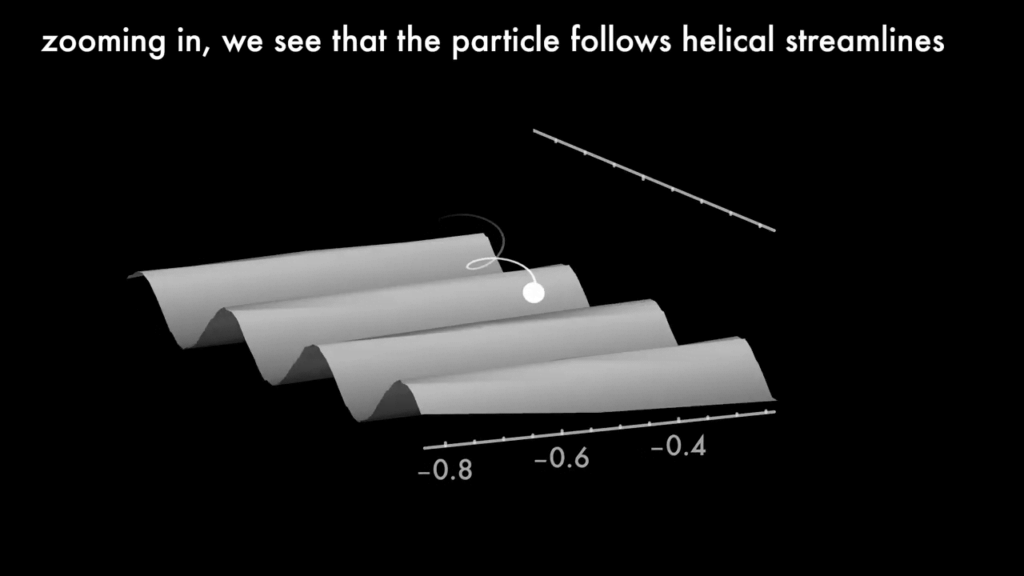
Dancing Over Ridges
When flowing over a ridged surface, particles follow a drifting, helical trajectory. In this video, researchers delve into the physics behind this phenomenon. Differences in the pressure gradient along different parts of the corrugation push particles along the groove. With their analysis, the team is able to predict particle trajectories above surface roughness of any shape. With these tools, they can design roughened microchannels that disperse particles at a desired speed, something that could be especially helpful in medical diagnostics. (Image and video credit: D. Chase et al.; research credit: D. Chase et al.)

Slab Avalanche Physics
Slab avalanches like the one shown here begin after weak, porous layers of snow get buried by fresher, more cohesive snow layers. On a steep slope, the weight of the new snow can be too great for friction to hold the slab in place, causing the upper layer to crack and slide at speeds up to 150 meters per second. Scientists had two competing theories for how slab avalanches began. One theory presumed that the weak layer of snow failed under shear; the other argued that the collapse of the lower, porous layer was at fault.
In a new study combining large-scale numerical simulation with real-life observations, scientists came to a new conclusion: cracks began to form in the porous layer as the weight of heavier snow crushed down, but once the cracks formed, the shear mechanism took over. Cracks formed by shear could propagate along the existing cracks in the porous layer, allowing faster crack propagation than through undamaged snow. In the end, it’s the combination of the two mechanisms that triggers the avalanche. (Image credit: R. Flück; research credit: B. Trottet et al.; via Physics World)

Little Surfer
Here’s another look at SurferBot, a low-cost, vibration-based robot capable of traversing both water and land. SurferBot’s vibration creates asymmetric ripples on the water surface. Because the waves are bigger at the rear of the robot, it gets propelled forward. But there doesn’t have to be water for SurferBot to get around! It’s actually amphibious, moving on both land and water. It can even transition from land to water on its own. (Image and video credit: E. Rhee et al.; research credit: E. Rhee et al.)

To Fizz or Not to Fizz
Place a drop of carbonated water on a superhydrophobic surface and it will slide almost frictionlessly, much the way Leidenfrost drops do. The drop behaves this way thanks to the self-produced layer of carbon dioxide vapor that it levitates on. As the gas escapes, the drop eventually settles back into contact with its surface. But until then, its levitation makes for some fun.

On the treated half of the glass (left), bubbles form a continuous film against the glass. On the untreated side (right), bubbles nucleate, grow, and rise as expected for a fizzy drink. Single droplets aren’t the only source of fun, however. In the images above, researchers coated the left half of a wine glass with a superhydrophobic treatment, while leaving the right half of the glass untouched. Once (dyed) carbonated water is poured into the glass, we see a bizarre dichotomy. In the right, untreated half of the glass, carbon dioxide bubbles nucleate, grow, and rise through the glass. But on the left side, the liquid appears still and bubble-less. In fact, the carbon dioxide gas on the left side is forming a continuous bubble film by the surface of the glass! (Image, video, and research credit: P. Bourrianne et al., see also)

Squeeze or Splatter?
Many a white shirt has met the disaster of a nearly-empty condiment bottle. One moment, you’re carefully squeezing out ketchup, and the next — sppplltlttt — you’re covered in red splatters. This messy phenomenon of gas displacing a liquid is widespread, showing up in condiments, some volcanic eruptions, and even the reinflation of a collapsed lung. Researchers have now constructed a mathematical model to fully capture and explain the process.
When you squeeze a container with both air and a liquid — like ketchup — in it, the air is easily compressed but the liquid is not. The extra pressure of the air creates a driving force that pushes the liquid out, despite its viscous resistance. Most of the time, these two forces are balanced, and the ketchup flows smoothly out of the container. But when the volume of ketchup is small compared to the air, squeezing can overpressurize the air, driving the ketchup out in an uncontrolled burst.
Luckily, the mathematics also suggest a solution to this problem: squeeze more slowly and double the size of the nozzle. You can also, they note, simply remove the top to avoid splatter. (Image credit: Rodnae Productions; research credit: C. Cuttle and C. MacMinn; via Ars Technica; submitted by Kam-Yung Soh)

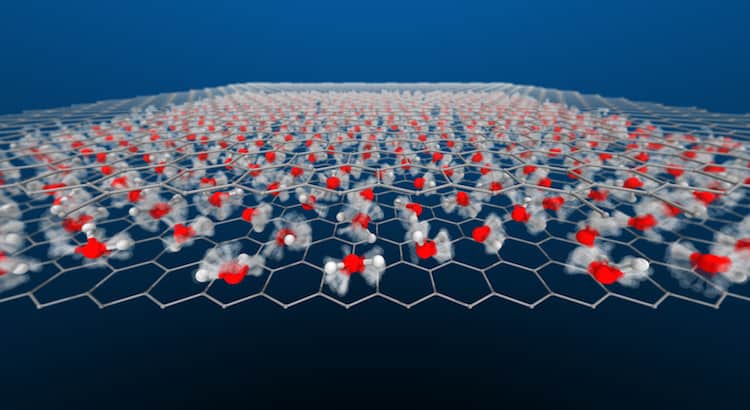
Nanoconfined Water
Water is a decidedly weird substance. It’s densest above its freezing point; it has a slippery liquid-like layer on its solid form; and, in the right form, it can bend like a wire. So it’s not surprising that water demonstrates some odd behaviors when it’s confined inside a space so narrow it’s only one molecule thick.
A new, simulation-based study finds that this nanoscale-confined water flows with a wide variety of behaviors, depending on the temperature and pressure. In some conditions, the water ceases to act molecularly, with hydrogen atoms flowing through a lattice of oxygen atoms. These superionic forms were thought only to exist in the extreme conditions of a gas giant’s interior, but these simulations suggest we can find them under far milder circumstances. (Image and research credit: V. Kapil et al.; via Physics World; submitted by Kam-Yung Soh)