This numerical simulation from NASA Goddard shows the motion of particulates in Earth’s atmosphere between August 2006 and April 2007. These aerosols come from various sources including smoke, soot, dust, and sea salt. As these fine particles move through atmosphere, they can have significant effects on weather as well as climate. For example, the particles serve as nucleation sites for the condensation and formation of rain drops. (Video credit: NASA Goddard SFC)
Category: Research

Stirring Faces
This video features simulation of the laminar flow around a plate plunging sinusoidally in a quiescent flow. As the plate moves up and down, it mixes the fluid around it. This is visualized in several ways, beginning with the vorticity. Clockwise and anti-clockwise vortices are shed by the edges of the plate as it moves. The flow is also visualized using particle trajectories, which are classified by their tendency to accumulate (attract) or lose (repel) particles. These trajectories are particularly intriguing to watch develop as they appear to show ornate faces and designs. (Video credit: S. L. Brunton and C. W. Rowley)

Fluorescing Shock Waves
Wind tunnel testing plays a major role in the planning of many space missions. Here a model of the Mars Sample Return Orbiter is tested at Mach 10 to determine the heat shield’s response to aerobraking off Mars’ atmosphere. The colors are the result of electron beam fluorescence, in which an electron gun is used to ionize molecules in the flow, which causes them to emit photons (light). The technique can be used for flow visualization–as in the case of the shock waves shown here–or to measure flow characteristics like density, temperature, and velocity. (Photo credit: Thierry Pot/DAFE/ONERA)

Swirling Jets
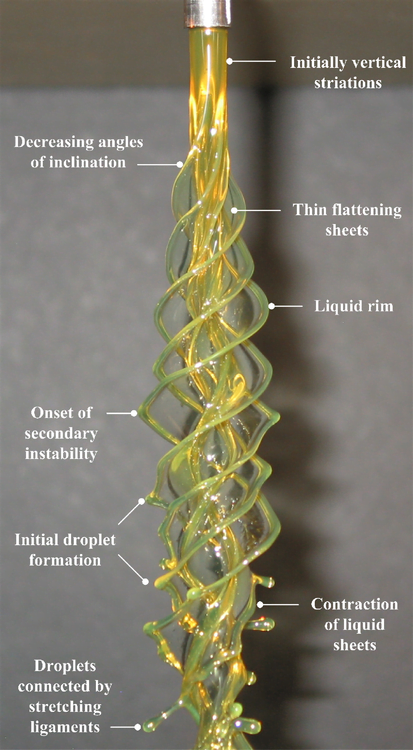
In fluid dynamics, we like to classify flows as laminar–smooth and orderly–or turbulent–chaotic and seemingly random–but rarely is any given flow one or the other. Many flows start out laminar and then transition to turbulence. Often this is due to the introduction of a tiny perturbation which grows due to the flow’s instability and ultimately provokes transition. An instability can typically take more than one form in a given flow, based on the characteristic lengths, velocities, etc. of the flow, and we classify these as instability modes. In the case of the vertical rotating viscous liquid jet shown above, the rotation rate separates one mode (n) from another. As the mode and rotation rate increase, the shape assumed by the rotating liquid becomes more complicated. Within each of these columns, though, we can also observe the transition process. Key features are labeled in the still photograph of the n=4 mode shown below. Initially, the column is smooth and uniform, then small vertical striations appear, developing into sheets that wrap around the jet. But this shape is also unstable and a secondary instability forms on the liquid rim, which causes the formation of droplets that stretch outward on ligaments. Ultimately, these droplets will overcome the surface tension holding them to the jet and the flow will atomize. (Video and photo credits: J. P. Kubitschek and P. D. Weidman)

Saturn’s Polar Vortex
Nothing quite compares to the beauty of fluid dynamics on astronomical scales. What you see here are raw photographs of recent storms at Saturn’s north pole. The recent change in Saturnian seasons has afforded Cassini a sunlit view of the northern pole, which had previously lain in darkness. A roiling vortex filled with clouds being twisted and sheared was revealed near the center of its famed polar hexagon. (Photo credit: NASA/JPL-Caltech/Space Science Institute; submitted by J. Shoer)

Supersonic Bubble Shock Waves
Supercomputing has been an enormous boon to fluid dynamics over the past few decades. Many problems, like the interaction between a supersonic shock wave and a bubble, are too complicated for analytical solutions and difficult to measure experimentally. Numerical simulation of the problem, combined with visualization of key variables, adds invaluable understanding. Here a shock wave strikes a helium bubble at Mach 3, and the subsequent interactions in terms of density and vorticity are shown. This situation is relevant to a number of applications, such as supersonic combustion and shockwave lithotripsy–a medical technique in which kidney stones are broken up inside the body using shock waves. After impact, an air jet forms and penetrates the center of the structure while the outer regions mix and form a persistent vortex ring. (Video credit: B. Hejazialhosseini et al.; via Physics Buzz)

Sandy Jets
When a fluid is vibrated, instabilities can form along its surface. With a sufficient amplitude, voids form inside the fluid and their collapse leads to a jet that shoots out from the fluid. A very different process leads to air cavities forming in a vibrated granular medium, but the jets produced are remarkably similar, as seen in this video. (Video credit: M. Sandtke et al.)

Frozen Powder Drops
Droplet impacts on granular surfaces and water interactions with superhydrophobic surfaces are not unfamiliar topics for FYFD. But this behavior of water droplets falling on a superhydrophobic powder is unusual, to say the least. When the droplets impact in powder, they rebound with a partial coating of powder. In the case of the superhydrophobic powder, the shape of the droplet is “frozen” by the powder. A satellite droplet is ejected from the region not coated in powder and the resultant main drop falls back to the surface and comes to rest with little to no deformation. The researchers report a critical velocity at which the behavior is observed. (Video credit: J. Marston et al.; via Physics Buzz)

Superfluid Vortices
Cooling helium to a few degrees Kelvin above absolute zero produces superfluid helium, a substance with some very bizarre behaviors caused by a lack of viscosity. Superfluids exhibit quantum mechanical properties on a macroscopic scale; for example, when rotated, a superfluid’s vorticity is quantized into distinct vortex lines, known as quantum vortices. These vortices can be visualized in a superfluid by introducing solid tracer particles, which congregate inside the vortex line, making it appear as a dotted line, as shown in the video above. When these vortex lines approach one another, they can break and reconnect into new vortices. These reconnections provoke helical Kelvin waves, a phenomenon that had not been directly observed until the present work by E. Fonda and colleagues. They are even able to show that the waves they observe match several proposed models for the behavior. (Video credit: E. Fonda et al.)

Sharkskin’s Secrets

Sharks are known as extremely fast and agile swimmers, due in part to the surface of their skin. Sharks are covered in very tiny tooth-shaped scales called denticles which are streamlined in the direction of flow over the shark. If you were to run a hand over a shark’s skin from head to tail, it would feel silky smooth, but rub against the grain and it’s like running your hand on sandpaper. Water encounters a similar resistance, which, according to new research, provides the shark with a passive flow control mechanism, requiring no effort on the part of the shark. When water near the shark’s denticles tries to reverse direction, an early stage in flow separation, the denticles naturally bristle, slowing and trapping the reversed flow. This prevents local flow separation which would otherwise increase the shark’s drag and hinder its agility. (Photo credit: James R. D. Scott; Research by A. Lang et al.)