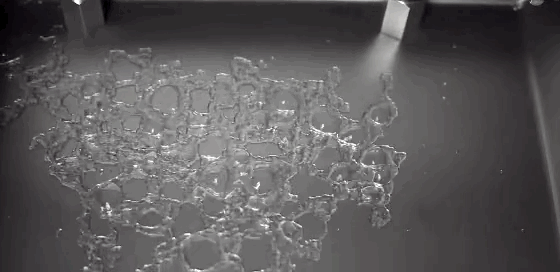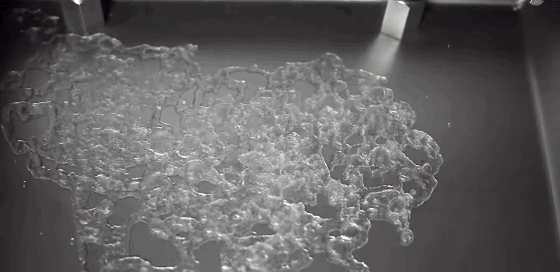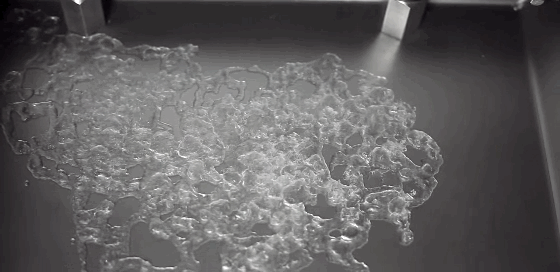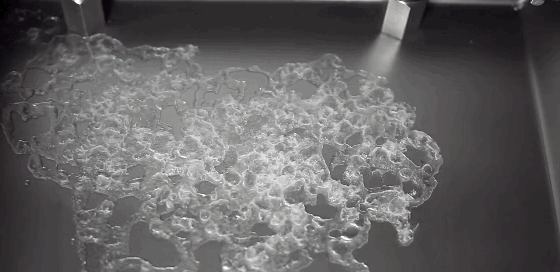Today’s Mars is cold and dry, with a thin and insubstantial atmosphere. One of the challenges facing planetary scientists is unraveling the processes behind the complex terrain we can observe on the surface. Without flowing water, how do we explain these features? A new experiment suggests that the answer lies in boiling.
Surface conditions on Mars include atmospheric pressures low enough to be below the triple point of water* – the critical temperature and pressure where water vapor, liquid water, and ice can all exist simultaneously. This means that liquid water is unstable under Martian conditions; any water that seeped up to the surface would immediately begin to boil. That explosive boiling ejects sand particles, as seen in the animation above. The authors suggest that this hybrid process of wet percolation combined with vaporous ejection of sediment may better explain the Martian surface features we observe. (Image credit: M. Masse et al., source: Supplementary Movie 3; via Gizmodo; submitted by Paul vdB)
* The evidence we’ve seen so far on Mars points to briny water flowing near the surface. Although brines have lower freezing temperatures than pure water, the authors’ argument holds for them, as well. The boiling is simply not as vigorous.