One of the joys of science is the sense of discovery that can come even from looking at something seemingly simple. Take, for example, a water droplet sitting on a plate. If you slowly tilt the plate, the droplet’s shape will shift until a critical angle where it starts sliding down the plate. But what happens to two initially different droplets? As this video shows, tilting two droplets of initially different shapes and returning them to horizontal causes the droplets to assume the same shape. There’s a universal behavior at work here–like nature has a kind of reset button that makes gravity and surface tension work together such that a droplet will assume a preferred shape. For an experimentalist, it’s certainly a handy way to create repeatable experiments! (Video credit: M. Musterd et al.)
Tag: surface tension

Glow-Stick Ferrofluids
Ferrofluids create all kinds of fascinating shapes when exposed to magnetic fields. In this video, Dianna from Physics Girl shows off what happens when you combine a ferrofluid with glowsticks and explains how ferrofluids get some of their unique properties. Ferrofluids consist of tiny nanoparticles of magnetic material that are surrounded by surfactants and suspended in a carrier fluid. This creates a fluid whose shape depends on gravity, surface tension, and the local magnetic field. By manipulating the relative strength of these forces, you can create everything from spikes to maze-like patterns to whatever this is. (Video credit and submission: Physics Girl)

Breaking Jets Into Drops
A falling stream of water will break into droplets due to the Plateau-Rayleigh instability. Small disturbances can create a wavy perturbation in the falling jet. Under the right conditions, the pressure caused by surface tension will be larger in the narrower regions and smaller in the wider ones. This imbalance will drive flow toward the wider regions and away from the narrower ones, thereby increasing the waviness in the jet. Eventually, the wavy jet breaks into droplets, which enclose the same volume of water with less surface area than the perturbed jet did. The instability is named for Joseph Plateau and Lord Rayleigh, who studied it in the late 19th century and showed that a falling jet of a non-viscous fluid would break into droplets if the wavelength of its disturbance was larger than the jet’s circumference. (Image credit: N. Morberg)

Spinning Paint
Fluid dynamical behaviors are often the result of competing forces. Here paint flung from a spinning rod illustrates the effects of adhesion, surface tension, and centrifugal force. In general, surface tension tries to hold a fluid together, and adhesion allows it to stay attached to a surface. Centrifugal force, on the other hand, tends to push the fluid outward. As the spinning rod accelerates, centrifugal force wins over adhesion and the paint spirals outward. For awhile, surface tension manages to hold the paint together, stretching it into spiraling ligaments of fluid. But when centrifugal force overpowers surface tension as well, the ligaments of paint snap into smaller droplets, still flying outward. Check out the full video for more great slow motion shots, and be sure to look at photographer Fabian Oefner’s “Black Hole“ series, which inspired the video. (Image credit: BBC Earth Unplugged, source video)

Bubble Rupture
Surface tension draws bubbles into spheres, but the balance of forces holding the sphere together is delicate. When pierced by a projectile, sometimes soap films can heal themselves, but often the film ruptures. Once a hole forms in the bubble, the film’s integrity is lost. Instead of holding the bubble together, surface tension pulls the soap film apart in a spray of thread-like ligaments that break into droplets. In the blink of an eye, the bubble is gone. (Image credit: W. Horton)

Sea Foam
Photographer Lloyd Meudell captures surrealistic images of breaking sea foam.
Interestingly, the sea foam is essentially a three-phase fluid made up of air, water, and sand. Yet despite the surrealism of its forms, the foam bears strong resemblance to other flows. The shapes the foam forms are reminiscent of vibrated non-Newtonian fluids like paint or oobleck. Momentum deforms the foam into sheets and ligaments smoothed and held together by surface tension until droplets snap free. You can find more of Meudell’s work at his site. (Image credits: L. Meudell; via freakingmindblowing; submitted by molecular-freedom)

Get Your Own Space Coffee Cup
A few weeks ago, we reported on the espresso machine NASA and the ESA sent to astronauts aboard ISS. The Capillary Beverage Experiment, known colloquially as the “Space Coffee Cup”, is an accompanying project that aims to use our understanding of fluid behavior in microgravity to design an open cup that simulates earthbound drinking experiences. As you can see above, astronauts are already enjoying drinks with it. The cup’s special shape is optimized so that surface tension can replace the role gravity plays in drinking on Earth. Where we pour drinks on Earth, the cup wicks liquid to the spout using surface-tension-driven capillary action. Right now there are only a handful of 3D printed cups on-orbit and here on Earth, but the company that designed them wants to manufacture glass versions for use here on the ground. So if you’d like your own space coffee cup, be sure to check out their Kickstarter campaign! (Video credit: IRPI LLC; image credit: NASA/IRPI LLC; Kickstarter project link)
ETA: To those who have been asking, that’s European astronaut Samantha Cristoforetti, who is (clearly) a Star Trek fan. I believe she’s doing a tribute to Captain Janeway’s coffee. (Black.)

Acoustic Levitation
Destin from Smarter Every Day has a great new video exploring acoustic levitation. With carefully placed speakers, you can create a standing wave with sound that’s capable of levitating lightweight objects against the force of gravity. Around 4:00, Destin demonstrates this with colored water droplets, which is where the real fireworks start. As he turns up the volume on the speakers, the big droplets explode. This happens when surface tension can no longer hold the drop together. But the high-speed footage offers other clues about what’s going on. Notice how the drops flatten out as the sound volume increases. If you look back to the standing wave animation at 1:33, you’ll notice that just to either side of the nodes (the spots that don’t move), the wave is still oscillating back and forth a little bit. As you increase the sound volume, that standing wave gets stretched to a larger amplitude, which means that those little oscillations just to either side of the node get stronger (and steeper), too. This change in acoustic pressure squishes the drops into pancakes as the fluid tries to stay right at the node. Eventually the droplet is just too flattened for surface tension to keep it together and it bursts into smaller droplets. (Video credit: Smarter Every Day; submitted by Matthew P.)

Make Your Own Dancing Droplets
As a follow-up to last week’s “dancing droplet” post, here’s a video that describes how to recreate the experiment yourself at home. The droplet motion is driven by the two-component structure of the droplets, where differing evaporation rates and surface tension values between the two fluids in the drop cause the attractions and chasing behavior you see. To demonstrate this at home, you’ll need glass, fire (for sterilization), tweezers, a pipette, water, and food coloring. Looks like a fun way to spend a weekend afternoon! (Video credit: M. Prakash et al.; via io9)

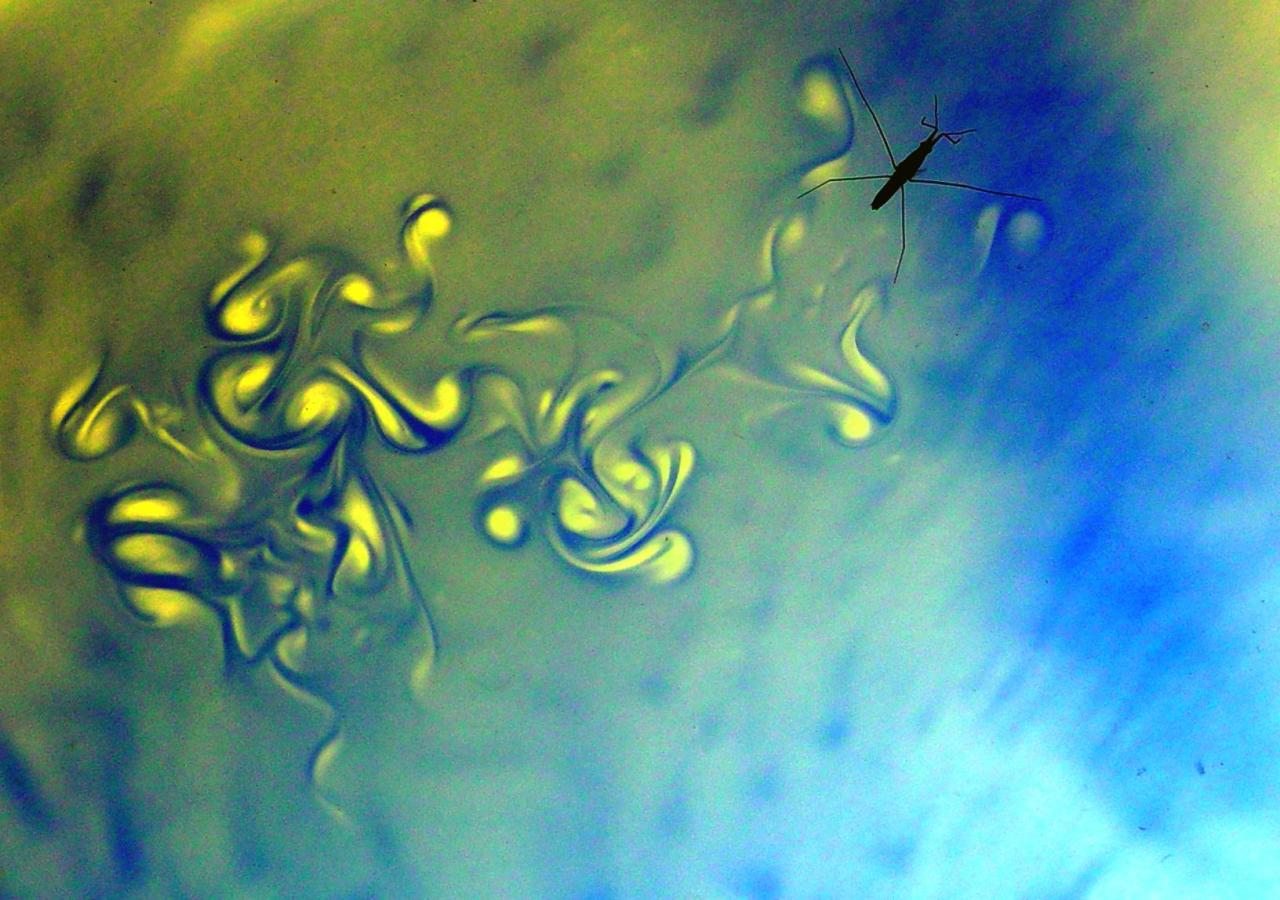
Rowing Water Striders

Water strider insects are light enough that their weight can be supported by surface tension. For some time, they were thought to propel themselves by using their long middle legs to generate capillary waves–ripples– that pushed them forward, but juvenile water striders are too small for this technique to work. Instead researchers found that water striders move by using their middle legs like oars. The leg motion creates vortices about 4 mm below the water surface, and this water moving backward propels the insect forward. In the photos above, the scientists visualized the flow by sprinkling thymol blue on the water and letting the striders move freely. You can learn more about the work here or in this Science Friday episode. (Photo credits: J. Bush et al.)