Branching, tree-like structures are found throughout nature. Take a thin layer of a viscous fluid pressed between two glass plates and inject a less viscous fluid like air and you’ll get branch-like structures. These are the result of the Saffman-Taylor instability and usually result in a fairly random outcome because of the instability’s sensitivity to small variations. In a new study, researchers use multiple air injection ports to finely control the formation and growth of air fingers, allowing them to build well-ordered branching structures like the one above. By placing the air ports in an array, the same technique can be used to create fluid meshes. The authors suggest this new technique could have wide-ranging applications including the design of heat exchangers and the growth of artificial tissues. (Image and research credit: T. ul Islam and P. Gandhi, source)
Tag: science

Rolling Along
Leidenfrost drops – droplets deposited onto a surface much hotter than their boiling point – are known for their mobility. With the right surface, they can be propelled, trapped, and even guided through a maze, typically by directing the vapor layer that cushions them. But new work shows that these drops have internal dynamics that also contribute to their propulsion.
By adding tracer particles to each droplet, researchers can visualize flows inside the droplet. Large drops tend to have a flatter shape and contain two or more rotating vortices. Such drops won’t propel themselves without another force in play. But smaller droplets are more spherical and contain only a single rotating flow. Once these drops detach, they roll away! Despite the similarity to wheels, these liquid drops aren’t moving the same way. Remember that the drop is not actually in contact with the surface. To see what sets the drop’s direction, researchers examined the shape of the bottom of the drop. They found that it sits at a slant on its vapor cushion. That pushes evaporating gases out one side, propelling the drop the other way. (Image and video credit: A. Bouillant et al., source)

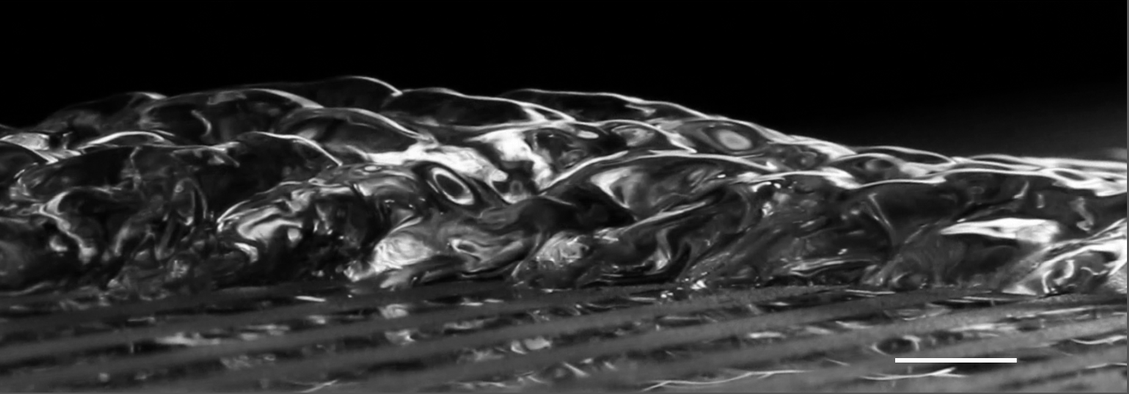
Layered Latte Physics
Latte lovers may be familiar with the layered latte, a beverage with distinctive horizontal layers mixing espresso and milk, but you may not have taken the time to wonder how these layers form. Like many layering phenomena in our oceans, the layered latte is the result of double-diffusive convection. This means that there are two variables that both affect density in the fluid mixture and that they act at different rates.
In the latte, those factors are 1) the different densities of the milk and espresso and 2) density changes caused as the latte cools to room temperature. A layered latte forms when the lighter espresso is poured into denser milk. If it’s poured quickly enough, the momentum of the pour forces some of the espresso down into the milk, despite the buoyant force that tries to keep the espresso on top. So that initial pour sets up a density gradient that runs from pure espresso at the top to pure milk on the bottom, with varying mixtures of the two in between.
The distinct layers won’t form until the latte begins cooling off. Along the walls of the container, heat is lost more quickly, causing fluid to cool and start sinking. But a specific bit of fluid can only sink until the fluid surrounding it is the same density. That can carry a cooler bit of latte to the bottom of a layer, but not into the denser layer below. At this point, our bit of latte moves inward, starts to warm up, and circulates up through the center of its layer. As when it sank, the fluid can only move up until it encounters a layer with equal or lesser density, at which point it must move horizontally instead. This thermal convection, combined with the density gradient formed by the initial pour, sets up the distinctive layers of the latte. The layers are quite stable – neither gentle stirring nor taking a sip will disrupt them for long – provided the drink remains warmer than the surrounding air. (Image credits: kopeattugu/Instagram, N. Xue et al.; research credit: N. Xue et al.; via NYTimes; submitted by Kam-Yung Soh)

Liquid Sunbursts
Liquid sunbursts and swirling aquatic roses abound in photographer Mark Mawson’s work. Images like these are created from dropping ink into water and photographing it as it diffuses. For the roses, the tank is additionally stirred or spinning to create the vortex-like appearance. Check out his website for more striking images, including more billowing ink, some great splashes and beautiful turbulent mixing between coffee and milk. (Image credit: M. Mawson; submitted by clogwog)

A Hot Tub, Turned Fluidized Bed
Fluidized beds continue to be all the rage among science YouTubers, but Mark Rober supersizes his by turning a broken hot tub into a massive bath of bubbling sand. His video includes a nice explanation of how a granular material like sand gets fluidized as well as how to make your own miniature bed. One of my favorite moments is shown in the animation below. When Mark drops a bowling ball into the fluidized bed, it creates a remarkably liquid-like splash. The ball sprays a splash curtain of sand up on impact and sinks into its own cavity. When the cavity seals behind the ball, it shoots up a tall jet of sand, just like a Worthington jet in water. Even with air fluidizing it, the sand doesn’t have surface tension, though, so the jet breaks up quite differently than water! (Video and image credit: M. Rober; submitted by clogwog)


Cloud Flows
When viewed at the right pace, clouds can flow. This timelapse of fog over Mt. Tamalpais State Park near San Francisco shows clouds moving over the hills there. Physically, this flow is an example of a familiar phenomenon known as a hydraulic jump. It happens when a fast-moving flow moves into a region of slower flow. The kinetic energy of the incoming flow gets transferred into potential energy, causing the flow to suddenly rise in height. It can also trigger turbulence, as seen on the right side of the animation. Watch carefully along a river, and you’ll see the same thing happening. Or, if your kitchen sink has a flat bottom, you can create a circular hydraulic jump just by turning on the faucet. You’ll get a region of fast flow right where the water impacts the basin, and a little ways out, you’ll see a circular jump where the water is suddenly taller and slower. That’s a hydraulic jump, too! (Image credit: Nicholas Steinberg Photography, source; submitted by Madi R.)

Corrugating Water
The characteristics of a surface can have a major impact on the form a flow takes. The photo above shows a corrugated, almost pinecone-like water surface. It’s the result of a sheet of water flowing over a surface with alternating bands of hydrophobic (water-repelling) and hydrophilic (water-loving) properties. The water sheet narrows over hydrophobic sections and expands over hydrophilic ones. Gravity, inertia, and surface tension compete to create the overall braided appearance. You can see a top-down view of the flow in the original poster. (Image credit: M. Grivel et al., source)

Flow Inside the Heart
Inside each of us is a remarkable and constant flow, driven by a muscle that’s always at work. As blood circulates through our bodies, it goes through a surprisingly varied journey. In the heart, as seen above, blood flow is very unsteady and quite turbulent, due to the beating pulse of the heart. As valves open and close and the muscle walls constrict and relax, the rushing blood moves in eddy-filled spurts. In the outer reaches of our capillaries, however, the nature of the flow is quite different. Thanks to smaller vessel sizes and other factors, capillary blood flow is much steadier and more laminar. Viscosity becomes more important, as do the non-Newtonian properties of components in our blood. (Image credit: mushin111/YouTube, source; via Science; submitted by Gary N.)

Water Music of Vanuatu
In the Pacific Island nation of Vanuatu, women have a tradition of water music, accompanying their singing with a percussive use of water. This video explores the physics behind this music. Performers use three basic motions – a slap, a plunge, and a plow – that each have distinctive acoustics thanks to the interaction of hand, water, and air. High pitches come from the initial impact on the water, whereas lower pitches come mostly from the collapse of the air cavity in the hand’s wake. By altering the rhythms and patterns of these three building blocks, the musicians create a rich harmony to accompany their singing. (Video credit: R. Hurd et al.)

Atmospheric Aerosols
Recently, NASA Goddard released a visualization of aerosols in the Atlantic region. The simulation uses real data from satellite imagery taken between August and October 2017 to seed a simulation of atmospheric physics. The color scales in the visualization show concentrations of three major aerosol particles: smoke (gray), sea salt (blue), and dust (brown). One of the interesting outcomes of the simulation is a visualization of the fall Atlantic hurricane season. The high winds from hurricanes help pick up sea salt from the ocean surface and throw it high in the atmosphere, making the hurricanes visible here. Fires in the western United States provide most of the smoke aerosols, whereas dust comes mostly from the Sahara. Tiny aerosol particles serve as a major nucleation source for water droplets, affecting both cloud formation and rainfall. With simulations like these, scientists hope to better understand how aerosols move in the atmosphere and how they affect our weather. (Image credit: NASA Goddard Research Center, source; submitted by Paul vdB)