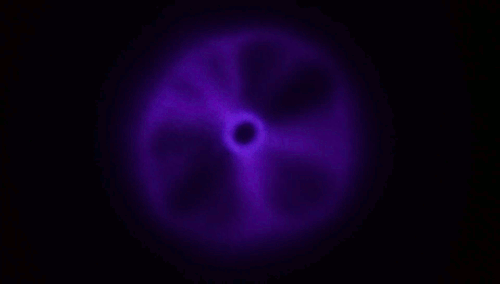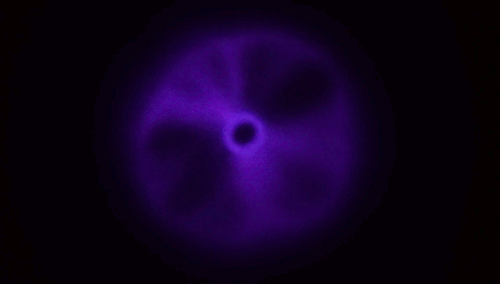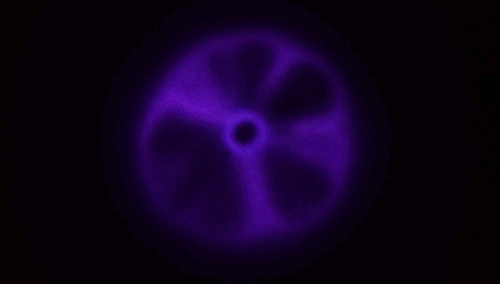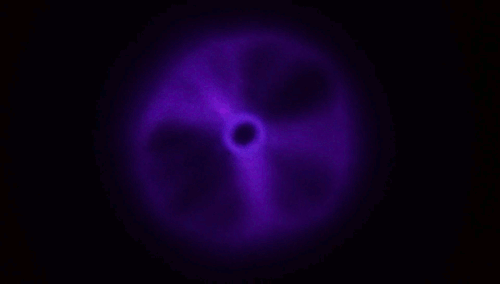
Although every child has experience blowing soap bubbles with a wand, only in recent years have scientists dedicated study to this problem. It turns out to be a remarkably complex one, with subtleties that can depend on the size of the wand relative to the jet a bubble-blower makes as well as the speed at which the air impacts the film. A recent study found that, at low or
moderate speeds, the film takes on a stable, curved shape (top image), but once you increase to a critical speed, the film will overinflate and burst. The key to forming a bubble, the authors suggest, is hitting that critical speed only briefly; if you slow down before the film ruptures, then the bubble has a chance to disconnect and form a sphere without breaking.
The work also suggests there are two reliable methods for bubble making in this way. One is to impulsively move the wand through the background fluid, as shown in the lower animation. The other is the one familiar to children: blow a jet just fast enough to overinflate the film, then let up so the bubble forms without breaking. (Image and research credit: L. Ganedi et al.; via Ars Technica; submitted by Kam-Yung Soh)