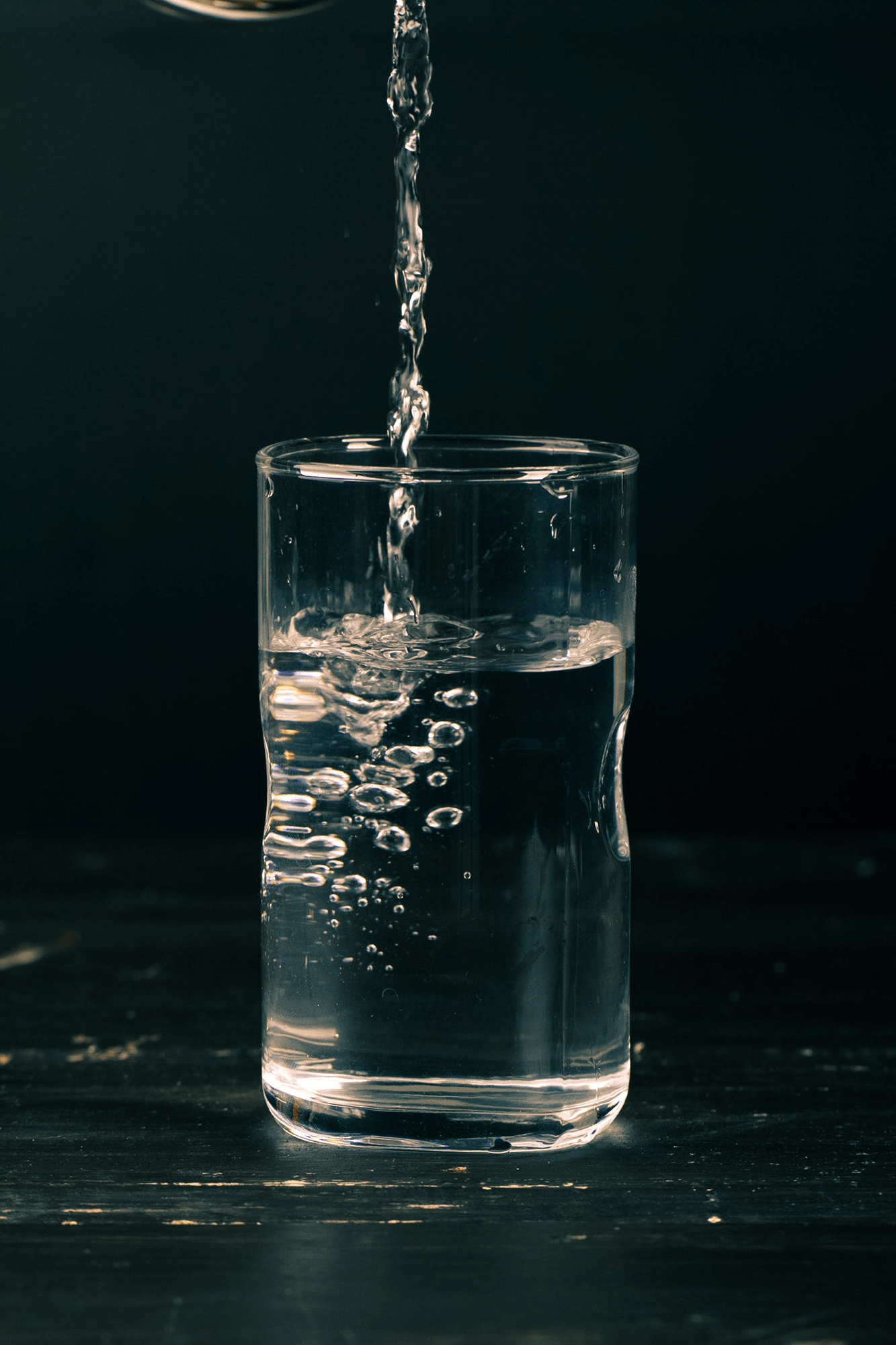In fluid dynamics, researchers are often challenged with complicated, messy flows. With so much going on at once, it’s hard to work out a way to keep track of it all. Here, researchers are looking at the break-up of two colliding liquid jets. This setup is often used to break rocket fuel into droplets prior to combustion. This video shows off a new data analysis tool that lets researchers break the flow into different parts, track them in time, and extract data about the changes that happen along the way. (Video and image credit: E. Pruitt et al.)
Tag: instability

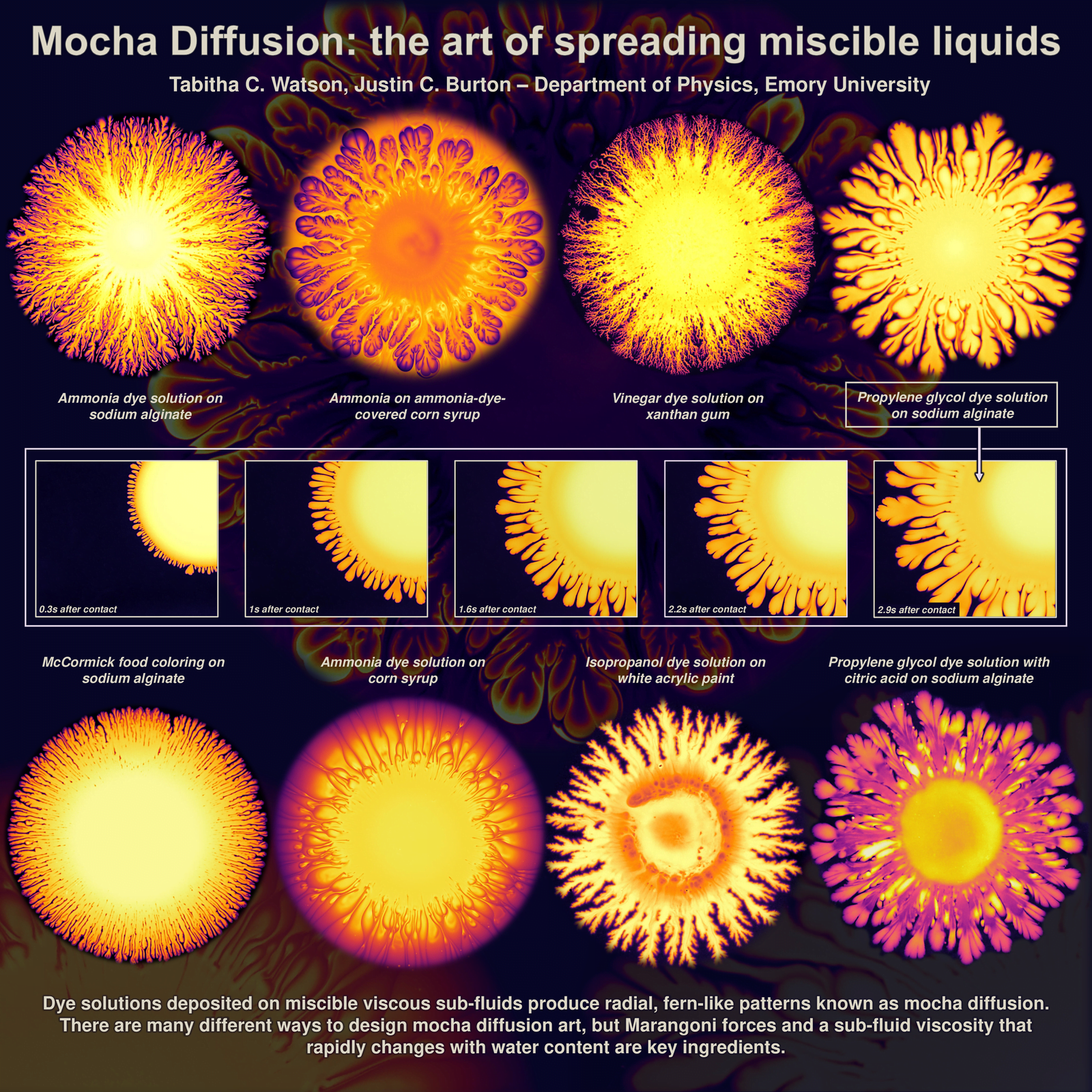
Mocha Diffusion
These firework-like patterns spread when dyes are added atop a viscous but miscible lower fluid layer. Here, researchers use lower layers like corn syrup and xanthan gum; then they spread dye mixtures including ammonia and vinegar atop those layers. Because the upper and lower layers of fluid are miscible and can diffuse into one another, they together form elaborate patterns. The mixing of the two layers creates gradients in surface tension that can drive the flow and create these mocha diffusion patterns. (Image credit: T. Watson and J. Burton)

The Sound of Bubbles
Every day I stand in front of my refrigerator and listen to the water dispenser pouring water into my glass. The skinny, fast-moving jet of water plunges into the pool, creating a flurry of bubbles. Those bubbles come from air the water jet pulls in with it, and the sound the water makes (minus the fridge’s noises) comes from those bubbles. A short, laminar jet will make fewer bubbles and, therefore, be quieter than a a jet that falls farther before hitting the water.
The reason? That tall jet falls for long enough that its walls start to wobble or even break up completely into separate droplets. Compared to a smooth jet, these wobbly or broken-up jets pull in more air and create more bubbles. That makes them louder. Researchers even suggest that listening to these bubbles can give a noninvasive method for finding how much fresh oxygen is in the water. (Image credit: R. Piedra; research credit: M. Boudina et al.; via APS Physics)

Liquid Lace
3D printers are a neat apparatus for exploring flow instabilities. If too much material is extruded compared to the speed of the printer head, coiling takes place. But under-extrusion creates patterns, too. Here, researchers show how under-extrusion can create a stable lace-like pattern. Once dried, the material can stretch, but only in certain directions, a bit like many textiles. (Video and image credit: L. Dreier et al.)

“High Flow”
Roman De Giuli’s “High Flow” is vibrant and energetic. Colorful paints and inks flow across the page, creating complex patterns. I love the blossoming flows, feathery fronds, and spreading Marangoni effects. De Giuli’s films never disappoint! (Video and image credit: R. De Giuli)

“Alive”
In “Alive,” filmmaker Christopher Dormoy explores acrylic paints and the variety of ways in which the medium can be used. From a fluids perspective, there’s dripping, viscous flow, turbulent eddies, billowing plumes, and “accidental painting” due to density-driven instabilities. It’s a fun tour of fluid phenomena in art. What examples do you spot? (Video and image credit: C. Dormoy)

Controlling Finger Formation
When gas is injected into thin, liquid-filled gaps, the liquid-gas interface can destabilize, forming distinctive finger-like shapes. In laboratories, this mechanism is typically investigated in the gap between two transparent plates, a setup known as a Hele-Shaw cell. In the past, researchers looking to control the instability have explored how surface tension, viscosity, and the elasticity of the gap itself affect the flows. But a new set of studies look at the compressibility of the gas being injected.
The team found that viscous fingers formed later the higher the gas’s compressibility. That provides a potential control knob for people trying to exploit the mechanism, especially geologists. For geologists trying to extract oil, viscous fingering is detrimental, but, on the flip side, viscous fingers are desirable when injecting carbon dioxide for sequestration. With these results, users can tweak their injection characteristics to match their goals. (Image credit: C. Cuttle et al.; research credit: C. Cuttle et al. and L. Morrow et al.; via APS Physics)

Granular Gaps
Push air into a gap filled with a viscous fluid, and you’ll get the branching, dendritic pattern of a Saffman-Taylor instability. Here, researchers use a similar set-up: injection into a narrow gap between transparent planes to explore something quite different. In this experiment, the gap was initially filled with a mixture of air and tiny hydrophobic glass beads. When the team injected a viscous mixture of water and glycerol, new patterns emerged. At low injection rates, a single finger structure formed. But at high injection rates, a whole spoke-like pattern formed. (Image and research credit: D. Zhang et al.; via Physics Today)

Turbulent Thermal Convection
In the winter, warm air rises from our floor vents or radiators, creating a complex, invisible flow in the background of our lives. Buoyancy lifts warmer air upward while cooler, denser air sinks back down. This thermal convection is everywhere: in our buildings, the ocean, the sky overhead — even in the visible layer of our sun.
In nature, these systems are so large and complex that fully measuring or simulating them remains impossible. Instead, researchers focus on a simplified system — a Rayleigh-Bénard cell — that’s essentially an idealized version of a pot on a stovetop. The lower surface of the cell is heated — like the bottom of a pan on the burner — while the upper surface of the fluid cools. Even this idealized system is a challenge, though, and neither lab-scale versions nor simulations can reach the same conditions that we find in nature.
To bridge the gap, scientists rely on mathematical models — theories built on our best understanding of the physics — and physical analogies to similar systems — like flow over a flat plate — that are “easier” to measure. For a thorough overview of recent work in the area, check out this review in Physics Today. (Image credit: A. Blass; research credit: D. Lohse and O. Shishkina in Physics Today)

Imitating a Cough
Coughing and sneezing create violent air flows in and around our bodies. As that fast air rushes over mucus layers in our lungs, throat, and sinuses, the resulting flow breaks up the mucus into droplets. To explore the details of that process, researchers built a “cough machine” that sends a rush of air over a thin film of water mixed with glycerol. The setup allows them to observe the physics in a way that’s nearly impossible in a human cough or sneeze.

Imitating a cough: high-speed video shows how a thin film made of water and glycerol breaks down in a strong airflow. Parts of the film inflate into hollow bags that form thinner weak spots. When the film breaks in those places, it forms rims and ligaments that create a spray of droplets. As seen above, air flowing past shears the viscous fluid, stretching it out. The leading edge of the film destabilizes and breaks into large drops, but it’s what comes next that really gets things going. Areas of the film inflate to form hollow bags. When sections of the bag thin to about 1 micron, the film ruptures and the bags burst. This triggers a cascade of instabilities in the film’s rim that ultimately rip the film into a spray of tiny aerosol droplets. The researchers found that, despite their tiny size, these droplets collectively carry a large volume of liquid, making them all the more important for understanding transmission of respiratory illnesses. (Image credit: top – A. Piacquadio, experiment – P. Kant et al.; research credit: P. Kant et al.)