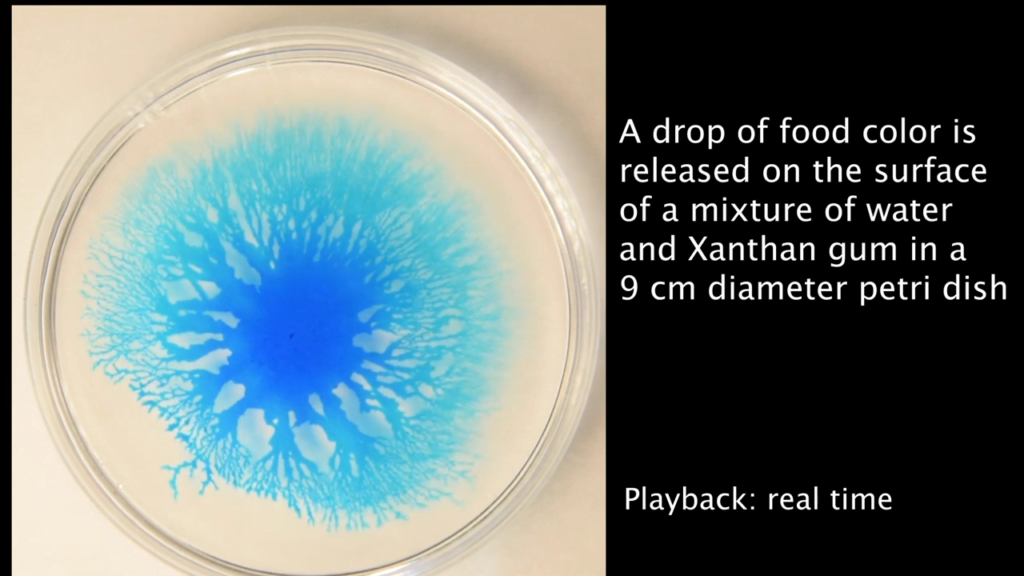
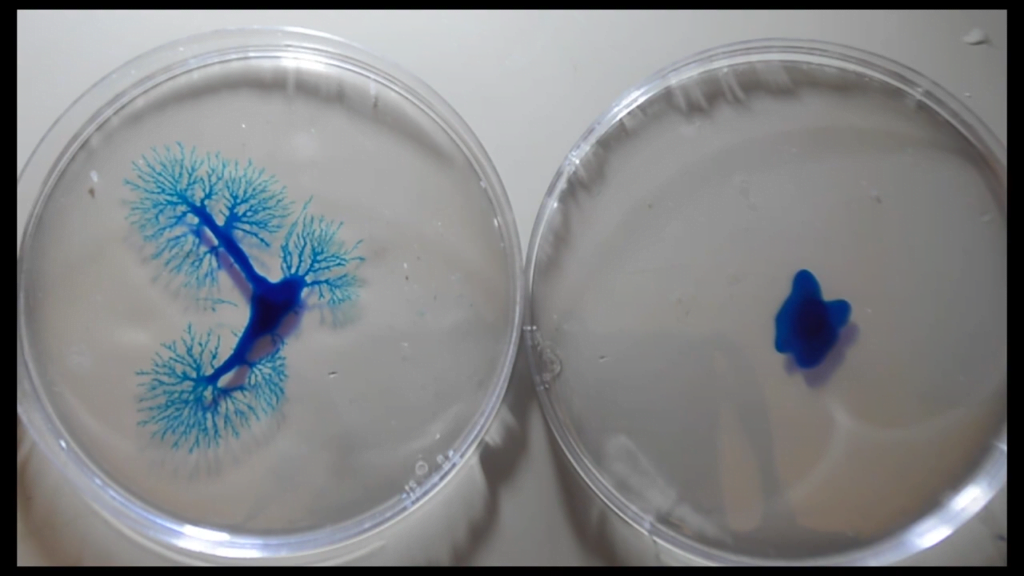
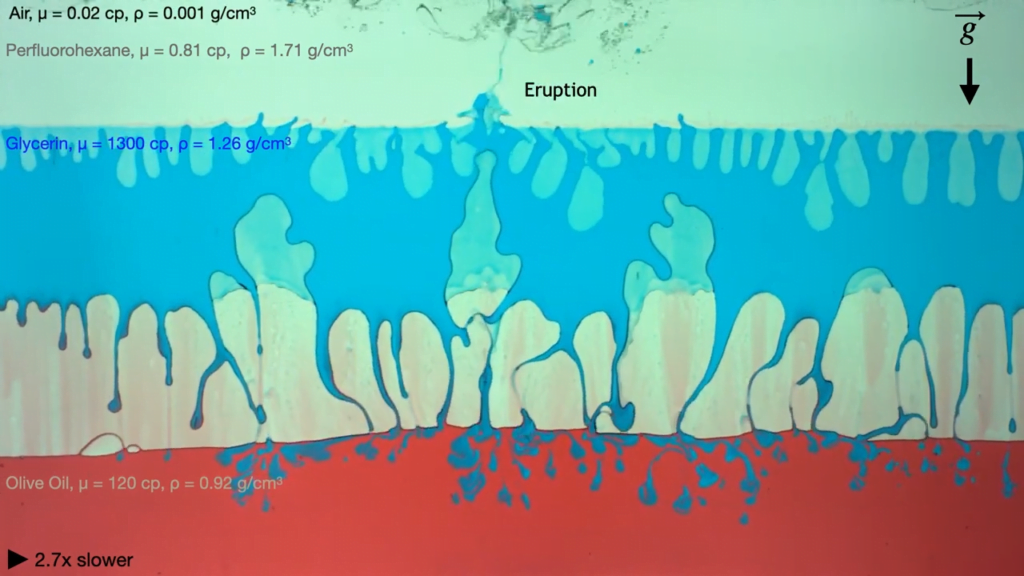
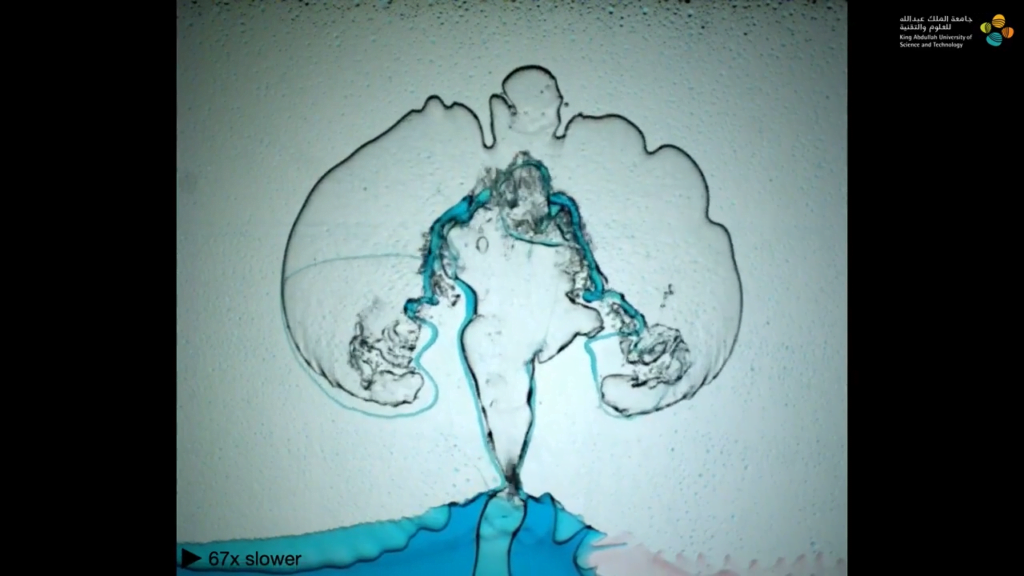
The mathematics of fluid dynamics still have many unknowns, which makes them an attractive playground for mathematicians of all stripes. One perennial area of interest is the Euler equations, which describe an ideal (i.e., zero viscosity), incompressible fluid. Mathematicians suspect that these equations may produce impossible answers — vortices with infinite velocities, for example — under just the right circumstances, but so far no one has been able to prove the existence of such singularities.
A recent Quanta article delves into this issue and the race between researchers using traditional methods and those using new deep learning techniques. Will the singularities be found and who will get there first? It’s well worth a read, whether theoretical mathematics is your thing or not. (Image credit: S. Wilkinson; see also Quanta; submitted by Jo V.)